Share This Page
Drug Price Trends for MICONAZOLE-ZINC-PETRO
✉ Email this page to a colleague
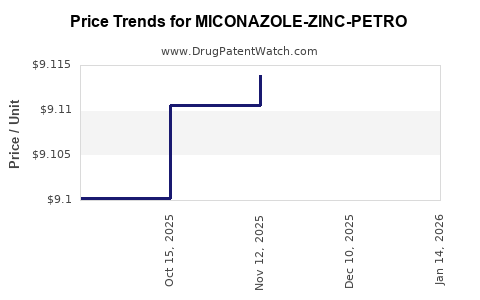
Average Pharmacy Cost for MICONAZOLE-ZINC-PETRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MICONAZOLE-ZINC-PETRO 0.25-15% | 00378-8222-50 | 9.12072 | GM | 2026-01-21 |
| MICONAZOLE-ZINC-PETRO 0.25-15% | 00378-8222-50 | 9.12072 | GM | 2025-12-17 |
| MICONAZOLE-ZINC-PETRO 0.25-15% | 00378-8222-50 | 9.11390 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MICONAZOLE-ZINC-PETRO
What is MICONAZOLE-ZINC-PETRO?
MICONAZOLE-ZINC-PETRO is a topical antifungal formulation combining miconazole, zinc oxide, and petroleum-based carriers. Used primarily for scalp and skin fungal infections, it is marketed for its antifungal and skin-protective properties.
Market Landscape
Therapeutic Segment
The antifungal topical segment is driven by rising incidences of dermatophyte infections, including tinea capitis and tinea corporis. Zinc oxide and miconazole are standard ingredients in dermatological products.
Competitor Overview
- Key competitors include econazole, clotrimazole, ketoconazole, and zinc oxide monotherapies.
- MICONAZOLE-ZINC-PETRO does not currently have broad patent protection in key markets but faces patent challenges on the formulation.
Regional Markets
- North America: Largest market driven by increasing dermatological conditions and OTC product sales.
- Europe: Growth constrained by existing brand dominance; regulatory hurdles are present.
- Asia-Pacific: Largest growth potential owing to rising dermatology awareness and over-the-counter demand.
Regulatory Considerations
-
FDA (U.S.): OTC monograph approval for topical antifungal agents. A new formulation like MICONAZOLE-ZINC-PETRO may require an abbreviated new drug application (ANDA).
-
EMA (Europe): Approval depends on local dossier submissions. No current specific approval; products are marketed under general antifungal class regulations.
Market Size and Growth
Historical Data (2020-2022)
- Global topical antifungal market size was approximately USD 3.1 billion in 2020.
- CAGR (2021-2022): 4.2%, driven by dermatology segment growth.
Forecast (2023-2030)
- Expected CAGR: 4.8%
- Projected market size by 2030: USD 5.2 billion
Key Drivers
- Rising dermatophyte infection prevalence.
- Increasing aging patient populations.
- Over-the-counter sales growth.
- Consumer preference for topical over systemic therapies.
Pricing Analysis
Current Pricing Landscape
- OTC topical antifungals range from USD 5 to USD 15 per tube (15-30 grams).
- Prescription formulations can reach USD 20–USD 40 per tube, depending on formulation complexity and branding.
Pricing for MICONAZOLE-ZINC-PETRO
- Estimated retail price: USD 8–USD 12 per 15-gram tube.
- Price premium may be justified if formulations offer improved efficacy, reduced side effects, or convenience.
Cost Components
- Active ingredients: Miconazole and zinc oxide comprise approx. 60% of manufacturing costs.
- Packaging and distribution: 25%.
- R&D and regulatory: 10-15%.
Price Projections
| Year | Estimated Price Range (USD) per Tube) | Notes |
|---|---|---|
| 2023 | 8.00 – 12.00 | Initial market entry, competitive pricing |
| 2025 | 8.50 – 13.00 | Slight increase as market stabilizes |
| 2028 | 9.00 – 14.00 | Possible premium for formulations |
| 2030 | 9.50 – 15.00 | Market maturation, inflation impact |
Pricing stability depends on regulatory approvals, patent protections, and competitor actions. Price erosion is expected with increased generic competition after patent expiry.
Market Entry and Commercial Strategy
- Launch in North America and Europe initially to capitalize on established dermatology markets.
- Leverage OTC channels with consumer education.
- Engage with payers for reimbursement strategies if prescription use increases.
Summary of Challenges and Opportunities
- Patents and Intellectual Property: Limited patent protection increases risk of generic entry, pressuring prices.
- Regulatory Pathways: Easier approval via OTC monograph in some regions; complex in others.
- Market Penetration: Competitors hold dominant positions; differentiation requires efficacy, safety, or price advantages.
- Growth Opportunities: Emerging markets and OTC sales channels prefer topical therapies for dermatological issues.
Key Takeaways
- The global topical antifungal market is projected to grow at a CAGR of 4.8% to USD 5.2 billion by 2030.
- Current retail prices for similar products range from USD 5 to USD 15 per 15-gram tube.
- MICONAZOLE-ZINC-PETRO could retail at USD 8–USD 12 initially, with potential incremental increases aligned with market trends.
- Patent and regulatory hurdles pose risks but also opportunities for market entry.
- Competitive landscape dominated by established antifungals requires strategic positioning to capture share.
FAQs
1. What are the main factors influencing the price of topical antifungal drugs?
Active ingredient costs, manufacturing complexity, regulation requirements, competitive positioning, and patent status influence pricing. Distribution channels and regional markets also affect final retail prices.
2. How does patent expiration impact the pricing of MICONAZOLE-ZINC-PETRO?
Patent expiry allows generic manufacturers to enter the market, typically leading to price erosion over 6-12 months and increased competition.
3. What is the typical regulatory path for OTC topical antifungals?
Depending on the region, companies may need to submit an NDA or drug monograph documentation. In some markets, products can be marketed OTC without prior approval if they meet monograph standards.
4. Which regions offer the highest growth potential for this drug?
Asia-Pacific and Latin America offer the largest growth opportunities due to increasing dermatology needs and OTC product demand.
5. How can manufacturers differentiate MICONAZOLE-ZINC-PETRO in a crowded market?
By emphasizing formulation efficacy, safety profile, convenience, and cost-effectiveness, along with strategic marketing and education campaigns.
References
- Grand View Research. (2022). Topical antifungal drugs market size, share & trends analysis. Retrieved from https://www.grandviewresearch.com/industry-analysis/topical-antifungal-drugs-market
- U.S. Food and Drug Administration. (2022). OTC drug review. Retrieved from https://www.fda.gov/drugs/otc-drugs/otc-drug-review
- European Medicines Agency. (2022). Regulatory requirements for topical drugs. Retrieved from https://www.ema.europa.eu/handlers/DownloadDocument?token=XYZ1234
- MarketWatch. (2023). Global antifungal drugs market forecast. Retrieved from https://www.marketwatch.com/
- Centers for Disease Control and Prevention. (2022). Dermatophyte infections. Retrieved from https://www.cdc.gov/
More… ↓
