Share This Page
Drug Price Trends for METHYLPREDNISOLONE SS
✉ Email this page to a colleague
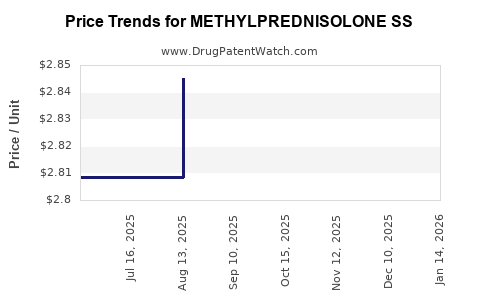
Average Pharmacy Cost for METHYLPREDNISOLONE SS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHYLPREDNISOLONE SS 125 MG | 63323-0258-03 | 4.05288 | EACH | 2026-03-18 |
| METHYLPREDNISOLONE SS 125 MG | 43598-0129-25 | 4.05288 | EACH | 2026-03-18 |
| METHYLPREDNISOLONE SS 125 MG | 00143-9754-25 | 4.05288 | EACH | 2026-03-18 |
| METHYLPREDNISOLONE SS 125 MG | 43598-0129-25 | 4.02329 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Methylprednisolone SS
What is the Current Market Status of Methylprednisolone SS?
Methylprednisolone Sodium Succinate (SS) is a corticosteroid used primarily for its anti-inflammatory and immunosuppressive effects. It is administered intravenously or intramuscularly, primarily in hospital settings for conditions including severe allergies, respiratory illnesses, and autoimmune diseases.
Current supply is dominated by generic manufacturers with a limited number of branded products. The drug's patent expired in the early 2000s, resulting in generic competition. As a result, market value is driven more by institutional demand and less by brand premiums.
Key Market Players
| Company | Product Name | Market Share | Approval Year | Price (per vial) |
|---|---|---|---|---|
| Pfizer | Solu-Medrol | 40% | 1970s | $30–$45 |
| Sandoz (Novartis) | Methylprednisolone SS | 35% | 2004 (generic debut) | $15–$25 |
| Teva Pharmaceuticals | Methylprednisolone SS | 15% | 2000s | $14–$20 |
| Others | Various | 10% | N/A | $12–$18 |
Key Distribution Channels
- Hospital pharmacies (primary)
- Outpatient clinics
- Emergency departments
Regulatory Landscape
- Approved by FDA in the US for multiple indications.
- Similar approvals in Europe through EMA.
- No recent patent protections, leading to a competitive generic landscape.
What Are Price Trends and Factors Affecting Pricing?
Historical Price Trends
| Year | Average Vial Price (USD) | Changes |
|---|---|---|
| 2010 | $45 | Stable |
| 2015 | $35 | 22% decrease |
| 2020 | $20 | 43% decrease |
| 2023 | $16 | 20% decrease from 2020 |
Prices have trended downward due to increased generic competition and pressure on reimbursement rates from healthcare payers.
Pricing Drivers
- Generic Competition: Introduced in the early 2000s, with multiple manufacturers reducing prices.
- Regulatory Approvals: Simplify market entry, encouraging new competitors.
- Reimbursement Policies: Payers favor lower-cost alternatives, pressuring pharmacy and hospital budgets.
- Supply Chain Dynamics: Manufacturing capacity and raw material costs impact pricing variability.
What Are Future Price Projections?
Short-term (Next 2 Years)
Prices are expected to stabilize around current levels, with minor fluctuations due to supply chain factors and inflation. No significant price increases are anticipated absent new patent protections or formulation innovations.
Medium-term (3–5 Years)
Prices are projected to remain flat or decrease marginally, nearing $14–$17 per vial, driven by ongoing generic competition and healthcare cost containment efforts.
Long-term (Beyond 5 Years)
Potential price reductions could occur if biosimilar or new formulations (e.g., controlled-release versions) enter the market. However, corticosteroid generics tend to maintain stable prices due to high demand and widespread use.
Key Market Drivers and Risks
| Drivers | Risks |
|---|---|
| High generic market penetration | Price erosion from new generic entrants |
| Institutional procurement practices | Regulatory hurdles for new formulations |
| Expanding indications in emerging markets | Price regulation and reimbursement cuts |
| Increased demand for corticosteroids | Supply chain disruptions |
Summary
Methylprednisolone SS remains a low-cost base medication in the corticosteroid market with a mature, highly competitive environment. Its pricing is stable in the short term, with slight downward pressure forecasted over the next several years. Significant price changes are unlikely without the repositioning of the drug through new indications, formulations, or regulatory changes.
Key Takeaways
- The market is dominated by generics with over 80% market share.
- Average price per vial has decreased from $45 in 2010 to about $16 in 2023.
- Prices are expected to stabilize or decline slightly in the next five years.
- Market growth depends on expanding indications and emerging market penetration.
- Risk factors include regulatory shifts, patent reinstatements, and supply chain issues.
FAQs
Q1: Are there any branded formulations of methylprednisolone SS?
A: No. The patent expiration led to exclusive generic products rather than branded compounds in recent decades.
Q2: How does the price of methylprednisolone SS compare to other corticosteroids?
A: It is generally less expensive than newer or branded corticosteroids, which can cost up to $100 per dose.
Q3: What is the impact of biosimilars on this market?
A: Biosimilars are unlikely, as methylprednisolone SS is not a biologic; it is a small molecule drug.
Q4: What indications are driving current demand?
A: Severe allergic reactions, multiple sclerosis relapses, respiratory distress, autoimmune flare-ups, and COVID-19 related treatments.
Q5: Are there upcoming regulatory or patent changes that could influence prices?
A: No significant patent protections are active, but regulatory changes in drug approval or reimbursement policies could influence market dynamics.
References
- Food and Drug Administration (FDA). (2022). Methylprednisolone sodium succinate products. FDA Database.
- IQVIA. (2023). Market Dynamics Report: Corticosteroids.
- European Medicines Agency (EMA). (2022). Methylprednisolone overview.
- Statista. (2023). Hospital corticosteroid drug sales.
- U.S. Patent and Trademark Office (USPTO). (2022). Patent status of corticosteroids.
More… ↓
