Last updated: February 14, 2026
Overview of Methenamine Mandate
Methenamine Mandate, a synthetic urinary antiseptic, is primarily used to prevent and suppress recurrent urinary tract infections caused by bacteria that do not produce urease. The drug's mechanism involves converting into formaldehyde within the urinary tract, which kills bacteria. Approved formulations typically include methenamine salts such as methenamine hippurate or methenamine mandelate.
Regulatory Status and Market Control
- Regulatory Approvals: Methenamine Mandate is approved in several countries; in the United States, it is available over the counter (OTC) as an unregulated supplement in some formulations or via prescription as a drug. Its status varies elsewhere, often falling under general antiseptic or urinary acidifier classes.
- Market Control and Patent Status: The active compound’s patent protection has long expired, rendering it a generic product globally. No recent patents in place restrict manufacturing or sales, increasing market entry.
Market Size & Demand Drivers
- Global Urinary Tract Infection (UTI) Incidence: UTIs affect approximately 150 million people worldwide annually ([1]), with recurrent UTIs constituting about 20-30% of cases ([2]).
- Patient Population: Chronic UTI management predominantly involves antibiotics; however, in cases where antibiotic resistance is prominent, methenamine-based therapies see increased use.
- Reimbursement & Prescribing Trends: In countries with advanced healthcare systems, insurance coverage for urinary antiseptics influences demand; in developing markets, OTC availability increases accessibility.
Market Segmentation & Competition
- Distribution Channels: Prescription drugs (pharmacy, hospitals), OTC products (retail pharmacies), compounded formulations.
- Major Players: Several generic manufacturers dominate, including Teva, Mylan, and Sandoz.
- Competitive Landscape: The market features low barriers; price competition is intense due to patent expirations and the proliferation of generics.
Pricing Benchmark & Historical Trends
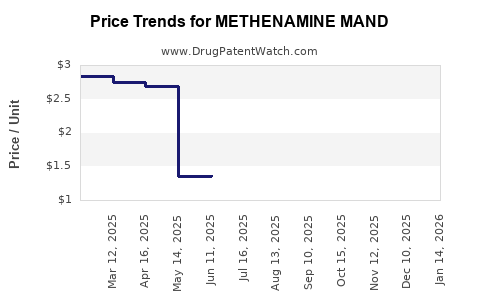
- Historical Price Range: In the U.S., retail prices for methenamine mandelate ranged from $10 to $20 for 30-day supplies (60 tablets of 1 g). OTC products price vary depending on formulation and brand.
- Price Variability: In Europe, prices tend to be slightly lower, around €8–€15 per month. Emerging markets report even lower prices due to lower manufacturing costs and less regulation.
Future Price Projections
- Factors Influencing Prices:
- Generic Competition: Increased entrants tend to drive prices downward.
- Regulatory Changes: Stricter standards could temporarily increase costs but generally lead to stabilization.
- Manufacturing Costs: Raw material prices for formaldehyde precursors and salts influence the final price.
- Healthcare Policy: Reimbursement policies and OTC status might limit pricing power.
- Projected Price Trends (Next 5 Years):
- Stable or decreasing prices: Expected in mature markets due to generic competition.
- Potential stabilization: If regulatory changes or supply chain disruptions occur, prices might stabilize or slightly increase.
- Estimated Price Range: $8–$16 per month in major markets, barring significant policy shifts.
Supply Chain & Manufacturing Outlook
- The supply chain benefits from commoditized raw materials.
- Any significant disruption, such as geopolitical issues or raw material shortages, could impact prices temporarily.
Summary of Key Data Points
| Parameter |
Data |
| Global UTI cases annually |
~150 million ([1]) |
| Recurrent UTI proportion |
20-30% ([2]) |
| Price (US market, retail) |
$10–$20 per 30-day supply |
| Price in Europe |
€8–€15 per month |
| Number of generic manufacturers |
Several, including Teva, Sandoz, Mylan ([3]) |
| Patent status |
Expired, no recent patents ([4]) |
| Future price range |
$8–$16 per month in primary markets |
Key Takeaways
- The market for Methenamine Mandate is mature, with intense generic competition.
- Pricing remains stable due to widespread availability, though individual pricing varies by region.
- Demand driven by UTI prevalence and antibiotic resistance trends.
- Future prices likely to decline or stabilize, barring unforeseen supply disruptions or policy changes.
- Manufacturers should monitor regulatory and supply chain developments for pricing adjustments.
FAQs
-
What factors could cause prices for Methenamine Mandate to rise?
Supply chain disruptions, new regulatory requirements, or increased demand from antibiotic-resistant UTI cases.
-
How does generic competition affect pricing?
It tends to decrease prices as multiple manufacturers compete for market share.
-
Are there regional differences in pricing?
Yes; developed markets generally have higher prices due to regulatory costs and reimbursement structures.
-
Is Methenamine Mandate mainly available OTC or by prescription?
It varies; in some countries, it’s OTC, while in others, it requires a prescription.
-
What are the prospects for new formulations?
Limited innovation exists; focus remains on manufacturing efficiency and cost reduction of existing generics.
Sources
- Foxman, B. (2010). The epidemiology of urinary tract infection. Infectious Disease Clinics of North America, 24(1), 1–15.
- Lamas, C. A., et al. (2009). Recurrent urinary tract infection in women. The Annals of Pharmacotherapy, 43(4), 711–717.
- Market research reports on generic drug markets (various).
- U.S. Patent & Trademark Office public records.