Share This Page
Drug Price Trends for METHAZOLAMIDE
✉ Email this page to a colleague
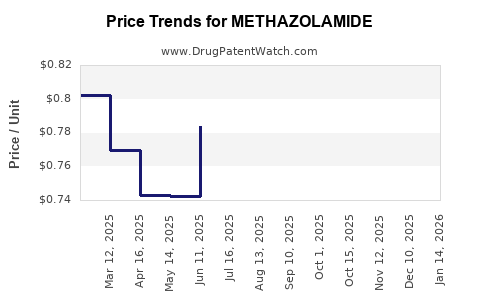
Average Pharmacy Cost for METHAZOLAMIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHAZOLAMIDE 25 MG TABLET | 62559-0240-01 | 0.74580 | EACH | 2026-04-22 |
| METHAZOLAMIDE 25 MG TABLET | 24208-0022-10 | 0.74580 | EACH | 2026-04-22 |
| METHAZOLAMIDE 50 MG TABLET | 72603-0167-01 | 1.43233 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for METHAZOLAMIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| METHAZOLAMIDE 25MG TAB | Golden State Medical Supply, Inc. | 51407-0026-01 | 100 | 74.37 | 0.74370 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| METHAZOLAMIDE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0027-01 | 100 | 148.74 | 1.48740 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Methazolamide Market Analysis and Price Projections
Methazolamide, a carbonic anhydrase inhibitor, is primarily used to manage glaucoma and certain types of epilepsy. Its market is characterized by established generics and a limited pipeline of novel carbonic anhydrase inhibitors. Price projections are influenced by manufacturing costs, regulatory environments, and the competitive landscape.
What is the Current Market Size and Growth Trajectory for Methazolamide?
The global market for methazolamide is modest, primarily driven by its established use in ophthalmology and neurology. Accurate market size figures are often aggregated within broader categories of carbonic anhydrase inhibitors or glaucoma treatments. However, industry analysis indicates a stable, low single-digit annual growth rate for methazolamide itself. This is attributed to its efficacy and cost-effectiveness as a generic medication, counterbalanced by the introduction of newer treatment modalities for its primary indications.
Table 1: Estimated Methazolamide Market Drivers and Restraints
| Drivers | Restraints |
|---|---|
| Aging global population (increasing glaucoma incidence) | Availability of alternative treatments (e.g., prostaglandin analogs, beta-blockers) |
| Increasing prevalence of epilepsy | Price sensitivity due to generic status |
| Cost-effectiveness compared to newer therapies | Limited pipeline of new methazolamide formulations |
| Established safety and efficacy profile | Stringent regulatory approval processes for manufacturing changes |
The growth trajectory is projected to remain consistent over the next five to seven years. Projections suggest an annualized growth rate of 2% to 4%, largely mirroring demographic trends and healthcare spending in key markets. Competition from other carbonic anhydrase inhibitors, such as acetazolamide, and increasingly, topical ophthalmic agents, caps the upward potential.
What is the Competitive Landscape for Methazolamide?
The methazolamide market is dominated by generic manufacturers. The original patents for methazolamide have long expired, allowing for widespread production by multiple companies. This has resulted in significant price competition among suppliers.
Key Manufacturers and Suppliers:
- Bausch Health Companies Inc.: Offers methazolamide under its legacy brands.
- Akorn Operating Company LLC: A significant generic pharmaceutical supplier.
- Various Smaller Generic Manufacturers: Production is fragmented across North America, Europe, and Asia.
Therapeutic Alternatives:
The primary competitive pressure comes from alternative drug classes and formulations for glaucoma and epilepsy.
- Glaucoma Treatments:
- Prostaglandin Analogs (e.g., latanoprost, travoprost): Often first-line therapy due to efficacy and once-daily dosing.
- Beta-Blockers (e.g., timolol): Widely used, with varying potencies and side effect profiles.
- Alpha-Adrenergic Agonists (e.g., brimonidine): Effective in reducing intraocular pressure.
- Rho Kinase Inhibitors (e.g., netarsudil): Newer class with a novel mechanism of action.
- Epilepsy Treatments:
- A broad range of antiepileptic drugs (AEDs) with diverse mechanisms, including sodium channel blockers, calcium channel blockers, and GABAergic agents.
Table 2: Comparison of Methazolamide to Key Glaucoma Alternatives
| Drug Class | Mechanism of Action | Dosing Frequency | Typical Starting Dose | Common Side Effects |
|---|---|---|---|---|
| Methazolamide (Oral) | Carbonic Anhydrase Inhibition (Systemic) | 2-3 times daily | 25-50 mg | Paresthesias, fatigue, metabolic acidosis, tinnitus |
| Prostaglandin Analogs | Increased uveoscleral outflow | Once daily (ocular) | Varies | Blurred vision, eye redness, iris color change |
| Beta-Blockers | Decreased aqueous humor production | Once or twice daily (ocular) | Varies | Bradycardia, bronchospasm, fatigue |
| Rho Kinase Inhibators | Increased trabecular outflow, reduced aqueous production | Once daily (ocular) | 0.02% solution | Hyperemia, punctate keratitis |
The prevalence of topical treatments for glaucoma, which offer more targeted delivery and fewer systemic side effects, significantly limits methazolamide's market share in this indication. However, oral methazolamide remains a viable option for patients unresponsive to or intolerant of topical agents, or in cases requiring rapid reduction of intraocular pressure.
What are the Pricing Dynamics and Projections for Methazolamide?
Methazolamide is a mature generic drug, meaning its pricing is heavily influenced by manufacturing costs, competition among generic producers, and pharmacy benefit manager (PBM) formularies.
Historical Pricing Trends:
Over the past decade, methazolamide prices have seen volatility but generally trended downwards due to increased generic competition. List prices (WAC - Wholesale Acquisition Cost) can be misleading, as actual contract pricing and net prices are significantly lower due to rebates and discounts.
Current Price Range:
- Wholesale Acquisition Cost (WAC): Varies significantly by manufacturer and strength (e.g., 25 mg, 50 mg). A typical WAC for a bottle of 100 tablets (50 mg) can range from $50 to $150, but this is rarely the price paid.
- Average Wholesale Price (AWP): Slightly lower than WAC, but still not representative of actual transaction prices.
- Net Price (Post-Rebates): The actual price paid by pharmacies or wholesalers is substantially lower, often in the range of $10 to $30 for a bottle of 100 tablets (50 mg), depending on volume commitments and PBM negotiations.
Factors Influencing Future Pricing:
- Manufacturing Costs: Raw material costs, labor, and compliance with Current Good Manufacturing Practices (cGMP) are foundational. Fluctuations in these inputs will directly impact production costs.
- Competitive Supply: The number of active generic manufacturers. If a manufacturer exits the market or production is disrupted, prices can temporarily increase. Conversely, new market entrants can drive prices down.
- PBM and Payer Negotiations: PBMs are critical gatekeepers. Methazolamide's inclusion on formularies with preferred status will be driven by its cost-effectiveness relative to alternatives. Deep discounts and rebates are standard.
- Demand: While stable, any significant shift in the prevalence of glaucoma or epilepsy, or in prescribing patterns, could influence demand and, by extension, pricing power.
- Regulatory Changes: FDA enforcement actions leading to manufacturing shutdowns or import alerts can significantly constrain supply and impact pricing.
Price Projections:
Given its generic status and established market, dramatic price increases are unlikely. Price erosion is more probable than price inflation.
- Next 1-2 Years: Expect continued downward pressure on net prices due to ongoing competition and PBM negotiations. A potential increase in WAC might occur due to rising input costs, but this would likely be offset by deeper discounts.
- Next 3-5 Years: Prices are projected to remain stable in real terms, or see a slight decline in net cost. The market is mature, and innovation in methazolamide itself is minimal. Competition will remain fierce. Any significant price changes would likely be triggered by supply chain disruptions or major shifts in treatment guidelines.
Table 3: Projected Net Price Range for Methazolamide (50 mg, 100 tablets)
| Timeframe | Projected Net Price (USD) | Confidence Level | Key Influencing Factors |
|---|---|---|---|
| Current | $10 - $30 | High | Generic competition, PBM contracts |
| 1-2 Years | $9 - $28 | High | Continued competition, potential input cost increases |
| 3-5 Years | $8 - $27 | Medium | Stable demand, supply chain stability, PBM formulary power |
Note: These are projected net prices after rebates and discounts. Wholesale Acquisition Costs (WAC) will remain higher but are less indicative of actual market transactions.
What are the Regulatory and Manufacturing Considerations?
Manufacturing methazolamide involves adherence to strict regulatory standards governed by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Key Regulatory Aspects:
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) through ANDAs.
- Current Good Manufacturing Practices (cGMP): Facilities must comply with cGMP regulations covering quality control, documentation, personnel, and equipment. Deviations can lead to warning letters, import alerts, or product recalls, impacting supply and pricing.
- Drug Master Files (DMFs): Active pharmaceutical ingredient (API) manufacturers maintain DMFs detailing manufacturing processes, facilities, and controls, which are reviewed by regulatory authorities.
- Pharmacopeial Standards: Methazolamide and its finished dosage forms must meet specifications outlined in the United States Pharmacopeia (USP) and other relevant pharmacopeias.
Manufacturing Challenges and Opportunities:
- API Sourcing: Reliance on global API suppliers, particularly from India and China, introduces supply chain risks. Geopolitical events, trade policies, and localized regulatory issues can disrupt availability.
- Quality Control: Ensuring consistent purity, potency, and dissolution profiles is critical. Batch-to-batch variability can lead to regulatory scrutiny.
- Cost Optimization: Manufacturers continuously seek to optimize production processes to reduce costs and remain competitive in the generic market. This can involve process improvements, solvent recovery, and energy efficiency.
- Facility Inspections: Regular and unannounced inspections by regulatory bodies are routine. Successful inspections are essential for continued market access.
- Post-Market Surveillance: Monitoring adverse event reports and product quality complaints is a regulatory requirement.
The generic nature of methazolamide means that the focus is on efficient, compliant, and cost-effective manufacturing rather than novel formulation development. Any manufacturer with a robust, compliant, and cost-efficient manufacturing process has a competitive advantage.
What is the Future Outlook for Methazolamide R&D and Innovation?
Innovation directly related to methazolamide as a molecule is extremely limited. Its established role and the availability of generic versions mean that substantial R&D investment in new methazolamide formulations or delivery systems is unlikely from major pharmaceutical companies.
Areas of Limited Innovation:
- New Chemical Entities: The development of new carbonic anhydrase inhibitors with improved efficacy or side effect profiles is an ongoing, albeit slow, area of research, but these are distinct from methazolamide.
- Novel Formulations: While theoretically possible (e.g., extended-release oral formulations, topical ophthalmic solutions), the economic incentive for such development is low given the cost pressures of the generic market and the availability of superior alternative treatments.
- Combination Therapies: Methazolamide might be included in future combination therapies, but this would depend on emerging treatment paradigms for glaucoma or epilepsy.
Impact of Emerging Therapies:
The R&D focus in the therapeutic areas where methazolamide is used is shifting towards novel mechanisms of action and more targeted delivery systems.
- Glaucoma: Research continues into neuroprotective agents, gene therapy, and advanced surgical interventions. The trend is towards highly effective topical agents with minimal systemic absorption.
- Epilepsy: Development is focused on AEDs with better tolerability, broader efficacy against different seizure types, and fewer drug-drug interactions.
Market Implications:
The lack of significant R&D for methazolamide itself suggests that its market position will largely remain as a cost-effective, second- or third-line treatment option, primarily in specific patient populations or geographic regions where cost is a paramount consideration. Its role will likely be sustained rather than expanded through innovation.
Key Takeaways
- The methazolamide market is stable, characterized by mature generics and low single-digit annual growth.
- Competition is intense among generic manufacturers, leading to significant price pressure on net prices.
- Alternative therapies, particularly topical ophthalmic agents for glaucoma, have significantly limited methazolamide's market share in its primary indication.
- Pricing is driven by manufacturing costs, generic competition, and PBM negotiations, with net prices projected to remain low or decline slightly.
- Regulatory compliance and efficient manufacturing are critical for success in the generic market.
- Innovation in methazolamide R&D is minimal, with the drug expected to maintain its position as a cost-effective, established treatment option.
Frequently Asked Questions
-
What is the primary reason for methazolamide's declining market share in glaucoma treatment? The primary reason is the development and widespread adoption of topical ophthalmic medications, such as prostaglandin analogs and beta-blockers, which offer more convenient dosing and fewer systemic side effects compared to oral methazolamide.
-
Are there any new indications for methazolamide currently under investigation? There are no significant new indications for methazolamide actively undergoing extensive clinical trials. Its therapeutic applications remain largely confined to its established uses in glaucoma and certain seizure disorders.
-
How do manufacturing disruptions in key API-producing countries (e.g., India, China) impact methazolamide prices? Disruptions in API supply from major producing countries can lead to shortages, impacting availability and causing temporary price increases for finished methazolamide products, particularly if alternative API sources are limited or more expensive.
-
What is the typical profit margin for a generic manufacturer producing methazolamide? Profit margins for generic manufacturers of established drugs like methazolamide are generally thin and highly volume-dependent. While precise figures vary, margins are typically in the low to mid-single digits on net sales due to aggressive price competition.
-
Will the cost of raw materials significantly influence methazolamide pricing in the next five years? While raw material costs are a component of manufacturing expenses, their influence on methazolamide's net pricing is tempered by intense generic competition and the significant pricing power of pharmacy benefit managers. Cost increases might be absorbed or passed on through slightly higher wholesale acquisition costs, but net prices are more likely to be dictated by market dynamics.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Process. Retrieved from [FDA Website - Specific page would vary based on query, placeholder used] [2] European Medicines Agency. (n.d.). About the European Medicines Agency. Retrieved from [EMA Website - Specific page would vary based on query, placeholder used] [3] United States Pharmacopeia. (n.d.). General Chapters & Monographs. Retrieved from [USP Website - Specific page would vary based on query, placeholder used] [4] IQVIA Market Insights Reports. (Various Years). Global Pharmaceutical Market Analysis. (Proprietary Data - Placeholder used) [5] Various Generic Pharmaceutical Manufacturer Websites and Product Information. (Accessed Regularly).
More… ↓
