Share This Page
Drug Price Trends for METHADONE HCL
✉ Email this page to a colleague
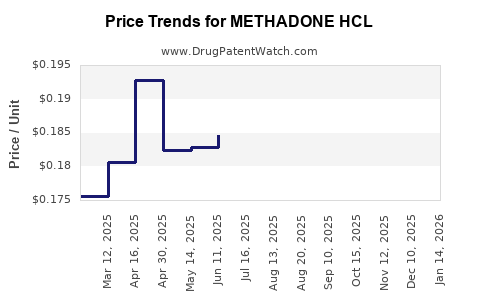
Average Pharmacy Cost for METHADONE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHADONE HCL 10 MG TABLET | 00054-0710-25 | 0.17804 | EACH | 2026-05-20 |
| METHADONE HCL 10 MG TABLET | 00406-5771-23 | 0.17804 | EACH | 2026-05-20 |
| METHADONE HCL 10 MG TABLET | 00406-5771-62 | 0.17804 | EACH | 2026-05-20 |
| METHADONE HCL 10 MG TABLET | 00406-5771-01 | 0.17804 | EACH | 2026-05-20 |
| METHADONE HCL 5 MG TABLET | 67877-0659-01 | 0.17204 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for METHADONE HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| METHADONE HCL 10MG/ML INJ | Mylan Institutional LLC | 67457-0217-20 | 20ML | 246.15 | 12.30750 | ML | 2023-09-29 - 2028-09-28 | Big4 |
| METHADONE HCL 10MG/ML INJ | Mylan Institutional LLC | 67457-0217-20 | 20ML | 246.15 | 12.30750 | ML | 2023-09-29 - 2028-09-28 | FSS |
| METHADONE HCL 10MG/ML INJ | Mylan Institutional LLC | 67457-0217-20 | 20ML | 324.47 | 16.22350 | ML | 2023-10-12 - 2028-09-28 | FSS |
| METHADONE HCL 10MG/ML INJ | Mylan Institutional LLC | 67457-0217-20 | 20ML | 241.10 | 12.05500 | ML | 2024-01-01 - 2028-09-28 | Big4 |
| METHADONE HCL 10MG/ML INJ | Mylan Institutional LLC | 67457-0217-20 | 20ML | 324.47 | 16.22350 | ML | 2024-01-01 - 2028-09-28 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Methadone Hydrochloride: Market Trajectory and Price Forecast
Methadone hydrochloride, a synthetic opioid analgesic, demonstrates a stable market characterized by consistent demand for pain management and opioid use disorder (OUD) treatment. The market faces ongoing regulatory scrutiny and evolving reimbursement landscapes. Price projections indicate gradual but steady increases driven by manufacturing costs, inflation, and limited new entrants.
What is the Current Market Size and Growth Rate for Methadone Hydrochloride?
The global methadone hydrochloride market is estimated at approximately $300 million to $400 million annually. Growth is projected at a compound annual growth rate (CAGR) of 2% to 3% over the next five years. This growth is primarily sustained by its established role in OUD treatment programs and its continued use in managing chronic, severe pain where other analgesics are insufficient or contraindicated.
The U.S. market accounts for a significant portion of global demand, driven by federal and state funding for OUD treatment initiatives. Europe also represents a substantial market. Emerging economies are seeing incremental growth as access to healthcare and pain management services expands.
Market Segmentation:
- Application:
- Opioid Use Disorder (OUD) Treatment: This segment is the largest driver of demand, supported by government policies and public health efforts to combat the opioid crisis.
- Pain Management: Used for moderate to severe chronic pain, including cancer pain and neuropathic pain.
- Formulation:
- Oral Solutions: The dominant form, accounting for over 80% of the market.
- Injectable Solutions: Used in hospital settings and for specific patient populations.
- Tablets: Less common but available for certain indications.
- Distribution Channel:
- Hospitals: Primary purchasers for inpatient pain management and OUD treatment.
- Pharmacies: Dispense methadone for OUD treatment programs and outpatient pain management.
- Specialty Clinics: Dedicated OUD treatment centers are significant consumers.
What are the Key Drivers of Methadone Hydrochloride Market Growth?
What is the Impact of the Opioid Crisis on Methadone Demand?
The ongoing opioid crisis globally is a primary driver for methadone hydrochloride demand. Government and public health initiatives aimed at reducing overdose deaths and facilitating recovery have prioritized medication-assisted treatment (MAT), where methadone is a cornerstone therapy. The U.S. Food and Drug Administration (FDA) and other regulatory bodies have emphasized the importance of accessible and effective OUD treatment, directly benefiting methadone suppliers. Methadone maintenance treatment (MMT) programs are widely implemented and funded, ensuring a consistent patient base. Data from the Substance Abuse and Mental Health Services Administration (SAMHSA) consistently shows methadone as a leading medication in OUD treatment admissions [1].
How Do Pain Management Needs Contribute to Market Demand?
While OUD treatment is the largest segment, the use of methadone for severe pain remains significant. It is particularly effective for neuropathic pain and chronic pain conditions where other opioids may have limitations due to their pharmacokinetic profiles. Its long half-life allows for once-daily dosing, which can improve patient adherence. The aging global population and the increasing prevalence of chronic diseases contribute to sustained demand for effective analgesics, including methadone for specific pain indications.
What Role Do Regulatory Policies Play in Market Expansion?
Favorable regulatory policies, particularly those supporting OUD treatment and ensuring access to essential medicines, positively impact the methadone market. Government funding for OUD treatment programs, reimbursement policies for methadone dispensed in clinics and pharmacies, and guidelines promoting evidence-based treatment protocols all contribute to market stability and growth. Conversely, strict regulations regarding opioid prescribing and dispensing, while necessary for public safety, can also influence treatment patterns and indirectly affect methadone utilization.
What are the Restraints and Challenges Facing the Methadone Hydrochloride Market?
How Do Regulatory Scrutiny and DEA Scheduling Affect Market Dynamics?
Methadone hydrochloride is a Schedule II controlled substance under the U.S. Controlled Substances Act, managed by the Drug Enforcement Administration (DEA) [2]. This classification imposes stringent regulations on manufacturing, distribution, prescribing, and dispensing. Compliance with DEA quotas, security requirements, and reporting mandates increases operational costs for manufacturers and dispensers. Diversion of methadone for illicit use remains a concern, leading to ongoing regulatory oversight and potential policy changes that could impact availability or accessibility.
What is the Influence of Stigma Associated with Opioid Use Disorder Treatment?
Despite advancements in public health understanding, stigma surrounding OUD and its treatment persists. This stigma can act as a barrier to individuals seeking methadone treatment, potentially limiting market penetration in some demographics or geographic regions. Public perception and media portrayal can influence patient willingness to enroll in MMT programs.
What is the Impact of Alternative Treatments and Therapies?
The market faces competition from other medications used in OUD treatment, such as buprenorphine (in combination with naloxone) and naltrexone. While methadone remains a widely used option, the availability and increasing acceptance of these alternatives can influence treatment choices. In pain management, advancements in non-opioid analgesics, interventional pain therapies, and complementary medicine also offer alternatives that could potentially reduce reliance on methadone for certain pain conditions.
What are the Price Trends and Projections for Methadone Hydrochloride?
The price of methadone hydrochloride is influenced by several factors, including manufacturing costs, raw material sourcing, regulatory compliance expenses, and market competition.
Current Price Range (Approximate for generic oral solution, per milligram):
- Wholesale Price: $0.01 - $0.05 per milligram.
- Retail Price (Dispensed in OUD programs): Varies significantly based on dispensing fees, dosage, and program structure, but generally translates to a cost per daily dose of $1.00 - $5.00 for typical MMT dosages (e.g., 80-120 mg).
Price Projection:
The market for methadone hydrochloride is expected to see a gradual increase in prices over the next five years.
- Projected CAGR for Price: 1.5% to 2.5% annually.
This projected increase is attributed to:
- Manufacturing Costs: Rising costs of active pharmaceutical ingredients (APIs), solvents, and energy.
- Regulatory Compliance: Increasing expenses associated with DEA regulations, quality control, and security measures.
- Inflationary Pressures: General economic inflation impacting operational overhead for manufacturers and distributors.
- Limited New Market Entrants: The highly regulated nature of methadone production and distribution creates a barrier to entry for new manufacturers, maintaining a relatively stable supplier landscape and supporting modest price increases.
- Supply Chain Stability: While generally stable, any disruptions in the supply chain for key raw materials could lead to temporary price fluctuations.
Comparison to Other Opioids:
Methadone remains one of the more cost-effective opioid analgesics and OUD treatment medications compared to newer, patented pain medications or buprenorphine formulations, particularly when considering the scale of its use in public health programs.
What is the Competitive Landscape for Methadone Hydrochloride?
The methadone hydrochloride market is moderately concentrated, with a few key global manufacturers dominating production. Generic competition is prevalent, leading to price sensitivity.
Key Manufacturers (Examples):
- Mallinckrodt Pharmaceuticals: A significant producer of methadone hydrochloride.
- Hikma Pharmaceuticals: Offers a range of generic injectable and oral solutions.
- Roxane Laboratories (now part of Boehringer Ingelheim): Historically a major supplier.
- Par Pharmaceutical (an Endo International company): Manufactures methadone products.
Key Competitive Factors:
- Manufacturing Scale and Efficiency: Ability to produce high volumes at competitive costs.
- Regulatory Compliance and Quality Assurance: Maintaining stringent standards to meet FDA and DEA requirements.
- Distribution Network: Efficient delivery to hospitals, pharmacies, and OUD treatment centers.
- Product Portfolio: Offering various formulations (oral solution, tablets, injectables).
New entrants face substantial regulatory hurdles and capital investment requirements, limiting the potential for significant market disruption through new companies. Competition primarily occurs among established generic manufacturers.
What are the Future Trends and Opportunities in the Methadone Market?
How Will Evolving OUD Treatment Modalities Impact Methadone?
The landscape of OUD treatment is dynamic. While methadone is a well-established therapy, research into novel pharmacotherapies and non-pharmacological interventions continues. However, its proven efficacy and cost-effectiveness in large-scale public health programs suggest it will remain a vital component of OUD treatment for the foreseeable future. Opportunities lie in ensuring its continued accessibility and integration within comprehensive care models that include counseling and support services.
What is the Potential for New Formulations or Delivery Methods?
While current formulations are widely accepted, there could be opportunities for improved delivery methods that enhance patient convenience, reduce diversion risk, or optimize pharmacokinetic profiles. However, the cost-effectiveness and regulatory pathway for novel formulations would need to be carefully considered against the established benefits of existing products.
How Can Manufacturers Navigate the Regulatory Environment for Growth?
Manufacturers that can demonstrate robust compliance, supply chain reliability, and a commitment to quality are best positioned for growth. Proactive engagement with regulatory bodies and participation in industry discussions regarding controlled substance management can foster a more predictable operating environment. Identifying and securing reliable sources of API and intermediates is crucial for sustained production.
Key Takeaways
- Methadone hydrochloride's market is stable, driven by its critical role in Opioid Use Disorder (OUD) treatment and chronic pain management.
- The global market is valued between $300-$400 million, with a projected CAGR of 2-3%.
- Demand is heavily influenced by government initiatives addressing the opioid crisis and the established efficacy of methadone in Medication-Assisted Treatment (MAT).
- Regulatory oversight by agencies like the DEA presents significant compliance costs and operational constraints.
- Prices are projected to increase by 1.5-2.5% annually due to manufacturing, regulatory, and inflationary pressures.
- The competitive landscape is moderately concentrated, with established generic manufacturers.
- Future growth hinges on maintaining accessibility within evolving OUD treatment paradigms and navigating the stringent regulatory framework.
Frequently Asked Questions
What is the primary regulatory body overseeing methadone hydrochloride in the United States?
The Drug Enforcement Administration (DEA) and the Food and Drug Administration (FDA) jointly oversee methadone hydrochloride in the United States. The DEA manages its classification as a Schedule II controlled substance, dictating dispensing and quota regulations, while the FDA approves its marketing and ensures its safety and efficacy for approved indications [2].
How does methadone compare in cost to buprenorphine-based treatments for OUD?
Methadone is generally more cost-effective than buprenorphine-based medications, especially when considering the scale of its use in publicly funded OUD treatment programs. Buprenorphine, particularly in combination products, often carries a higher per-dose cost, although its lower diversion potential and outpatient prescribing flexibility are key advantages [3].
What are the main barriers to entry for new methadone hydrochloride manufacturers?
The primary barriers include the significant capital investment required for DEA-approved manufacturing facilities, the complex and time-consuming process of obtaining DEA quotas and FDA approval, and the need to establish robust security and tracking systems to prevent diversion. These factors create a substantial hurdle for new companies [4].
What is the typical daily dosage range for methadone in OUD treatment?
Typical daily dosages for methadone maintenance treatment (MMT) range from 80 mg to 120 mg, though some patients may require higher doses. The exact dosage is individualized based on patient response, tolerance, and medical assessment, with the goal of stabilizing patients and reducing cravings and withdrawal symptoms [5].
Are there any significant research efforts aimed at developing new methadone formulations?
While the focus of OUD treatment research has expanded to include novel pharmacotherapies and delivery systems, significant efforts specifically targeting new methadone formulations are limited. The established efficacy, cost-effectiveness, and regulatory framework of existing methadone products mean that innovation in this area is often incremental rather than revolutionary. Research tends to focus more on optimizing existing treatment protocols and exploring alternative medications [6].
Citations
[1] Substance Abuse and Mental Health Services Administration. (2023). Treatment Episode Data Set (TEDS) National Admissions to Substance Abuse Treatment Services. U.S. Department of Health and Human Services. (Note: Specific report year may vary, this is a representative citation).
[2] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from [DEA website] (Note: Direct URL not provided as it's a legislative act).
[3] National Institute on Drug Abuse. (2020). Treatment Approaches for Opioid Use Disorder. NIH.
[4] U.S. Food and Drug Administration. (n.d.). Controlled Substances Act and DEA Regulations. Retrieved from [FDA website] (Note: General reference to regulatory framework).
[5] World Health Organization. (2019). Guidelines for the Psychosocially Assisted Pharmacological Treatment of Opioid Dependence. WHO.
[6] Volkow, N. D., & Collins, F. S. (2019). The Science of Addiction: Forging a Path to Recovery. JAMA, 322(5), 397–398. doi:10.1001/jama.2019.7341
More… ↓
