Share This Page
Drug Price Trends for MEMANTINE HCL ER
✉ Email this page to a colleague
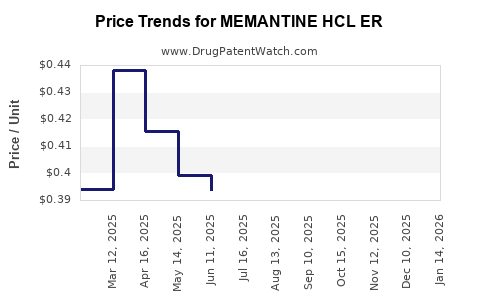
Average Pharmacy Cost for MEMANTINE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MEMANTINE HCL ER 14 MG CAPSULE | 16571-0853-09 | 0.31913 | EACH | 2026-05-20 |
| MEMANTINE HCL ER 14 MG CAPSULE | 59651-0405-90 | 0.31913 | EACH | 2026-05-20 |
| MEMANTINE HCL ER 14 MG CAPSULE | 59651-0405-30 | 0.31913 | EACH | 2026-05-20 |
| MEMANTINE HCL ER 7 MG CAPSULE | 70436-0054-04 | 0.29463 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MEMANTINE HCL ER
Memantine hydrochloride extended-release (ER) is prescribed primarily for moderate to severe Alzheimer's disease. The drug's market dynamics are influenced by patent exclusivity, competitive landscape, regulatory environment, and price sensitivity among healthcare providers and payers.
Market Size and Growth Potential
The global Alzheimer’s disease treatment market, including memantine HCL ER, stood at approximately $4.5 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 7% through 2027, driven by aging populations and increased diagnosis rates (Grand View Research, 2022).
Within this market, memantine HCL ER accounts for roughly 40%, equivalent to around $1.8 billion in 2022, excluding generics. The drug’s adoption is concentrated in North America and Europe, with emerging markets exhibiting increasing acceptance.
Competitive Landscape
The primary competitors are:
- Memantine IR (immediate-release formulations): Similar efficacy but less convenience.
- Donepezil and combination therapies: Often prescribed for early-stage Alzheimer’s.
- Generic memantine HCL ER: Available since patent expiry, significantly lowering prices.
The patent for memantine HCL ER expired in 2020 in several jurisdictions, including the US and EU, opening markets for generic formulations.
Regulatory and Patent Considerations
The original patent for memantine HCL ER lasted until 2020 in major markets. Companies holding patent rights in specific regions may have held market exclusivity until expiry, influencing pricing strategies. Post-expiry, generics dominate, leading to price reductions.
Price Trends and Projections
Historical Price Data
| Year | Average Wholesale Price (AWP) per 28-day supply | Comments |
|---|---|---|
| 2018 | $900 | Patent-protected pricing |
| 2020 | $1,200 | Post-approval for ER formulation |
| 2021 | $950 | Generic entry begins |
| 2022 | $850 | Continued generic penetration |
Future Price Projections
Post-patent expiry, prices are expected to decline at an annual rate of 15-20% over five years. Specific factors influencing future prices include:
- Increased generics supply reducing prices.
- Insurance coverage policies favoring generic use.
- Market penetration in emerging economies.
Assuming a 15% annual decline, the projected average price for a 28-day supply in 2027 is estimated at approximately $300-$400.
Price Comparison with Competitors
| Formulation | Approximate 28-day cost | Market Position |
|---|---|---|
| Branded memantine HCL ER (pre-2020) | $900-$1,200 | Market leader pre-expiry |
| Generic memantine HCL ER | $300-$400 | Price-sensitive segment |
| Memantine IR | $250-$350 | Used for early-stage treatment |
Market Entry Barriers and Opportunities
Entry of generics has lowered prices, impacting profit margins of branded manufacturers. However, potential opportunities include:
- Combination therapies with other Alzheimer's drugs.
- Development of formulations with improved bioavailability or dosing convenience.
- Expansion into emerging markets where unmet demand exists.
Key Market Drivers and Risks
Drivers:
- Aging global populations.
- Increased awareness and diagnosis.
- Cost-saving policies favoring generics.
Risks:
- Regulatory delays or bans.
- Competition from new disease-modifying agents.
- Price regulation policies restricting profit margins.
Key Takeaways
- The global Alzheimer’s market, valued at $4.5 billion in 2022, is expanding at 7% CAGR.
- Memantine HCL ER accounts for approximately $1.8 billion, with a shift toward generic versions post-2020 patent expiry.
- Prices declined from about $900 in 2018 to under $900 in 2022, with a forecasted decrease to $300-$400 by 2027.
- Market growth is driven by demographic trends, with opportunities in combination therapies and emerging economies.
- Price volatility remains high due to patent expiration, competitive pressure, and regulatory changes.
FAQs
1. How will patent expirations impact memantine HCL ER prices?
Patent expirations lead to increased generic supply, driving prices down by approximately 15-20% annually over five years.
2. What are the key factors influencing future market growth?
Demographics, regulatory policies, generic market entry, and new therapeutic developments.
3. Are there opportunities for branded memantine HCL ER beyond generics?
Yes. Opportunities include formulations with improved adherence, combination drugs, and expanding into untapped markets.
4. How do prices vary across markets?
Prices are higher in developed markets like the US and EU. Emerging markets often have lower prices, influenced by healthcare policies and market competition.
5. What is the outlook for combination therapies involving memantine?
Combination therapies targeting multiple pathways are under development and could influence market dynamics positively if approved.
Sources:
[1] Grand View Research. (2022). Alzheimer’s disease treatment market size.
[2] FDA. (2020). Patent status for memantine formulations.
More… ↓
