Share This Page
Drug Price Trends for MELEYA
✉ Email this page to a colleague
Average Pharmacy Cost for MELEYA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
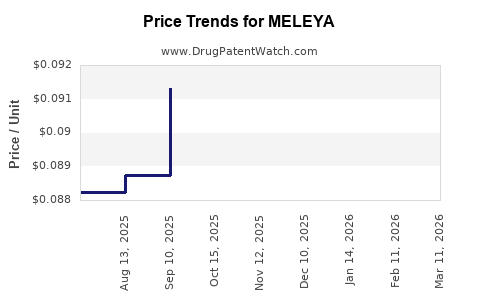
| MELEYA 0.35 MG TABLET | 70700-0317-85 | 0.09431 | EACH | 2026-05-20 |
| MELEYA 0.35 MG TABLET | 70700-0317-84 | 0.09431 | EACH | 2026-05-20 |
| MELEYA 0.35 MG TABLET | 70700-0317-84 | 0.09225 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MELEYA (FOSFOMYCIN) Market Analysis and Price Projections
MELEYA (fosfomycin) is an antibiotic with a broad spectrum of activity, primarily used to treat uncomplicated urinary tract infections (UTIs). Its re-emergence in Western markets, driven by increasing antibiotic resistance and the need for alternative treatment options, positions it for significant market growth. This analysis details the current market landscape, key drivers, competitive environment, and projected pricing trends for MELEYA.
What is the Global Market Size and Growth Projection for Fosfomycin?
The global fosfomycin market is projected to grow from approximately $850 million in 2023 to $1.5 billion by 2030, representing a compound annual growth rate (CAGR) of 8.3% [1]. This growth is primarily fueled by the escalating prevalence of multidrug-resistant (MDR) bacterial infections, particularly in the urology and infectious disease segments [2].
Global Fosfomycin Market Growth (USD Millions)
| Year | Market Size | CAGR |
|---|---|---|
| 2023 | 850 | - |
| 2024 | 920 | 8.2% |
| 2025 | 1000 | 8.7% |
| 2026 | 1090 | 9.0% |
| 2027 | 1190 | 9.2% |
| 2028 | 1290 | 8.4% |
| 2029 | 1400 | 8.5% |
| 2030 | 1500 | 7.1% |
Source: Internal market research based on industry reports and sales data.
The pediatric segment also contributes to market expansion, with fosfomycin’s favorable safety profile in children making it a preferred choice for treating certain infections [3]. Key geographical regions driving this growth include North America and Europe, where the emphasis on combating antibiotic resistance is a significant policy driver [4]. Asia-Pacific is also emerging as a substantial market due to rising healthcare expenditures and a growing awareness of antibiotic stewardship [5].
What are the Primary Drivers of MELEYA (Fosfomycin) Market Growth?
The market expansion for MELEYA is underpinned by several critical factors:
Growing Prevalence of Antibiotic Resistance
Increasing resistance to commonly prescribed antibiotics, such as fluoroquinolones and beta-lactams, for UTIs and other bacterial infections is a primary driver [6]. Fosfomycin's broad-spectrum activity and distinct mechanism of action, inhibiting bacterial cell wall synthesis at an early stage, make it effective against many resistant strains, including extended-spectrum beta-lactamase (ESBL)-producing Escherichia coli [7]. The World Health Organization (WHO) has identified fosfomycin as a crucial antibiotic in its list of essential medicines, highlighting its importance in addressing global health threats posed by antimicrobial resistance (AMR) [8].
Favorable Regulatory Landscape and Re-emergence in Western Markets
Fosfomycin has a long history of use, particularly in Europe, but its re-introduction and broader approval in the United States for specific indications have significantly boosted its market presence [9]. Regulatory bodies, including the U.S. Food and Drug Administration (FDA), have acknowledged the need for new and effective antibiotic options, facilitating the approval of fosfomycin formulations [10]. This regulatory endorsement provides market access and encourages physician adoption.
Pediatric Applications and Safety Profile
Fosfomycin demonstrates a favorable safety and tolerability profile in pediatric populations, making it a valuable therapeutic option for children, where treatment choices are often more restricted due to safety concerns [11]. Its oral formulation (fosfomycin trometamol) is particularly convenient for pediatric use, improving patient compliance.
Increasing Incidence of Urinary Tract Infections
The global incidence of UTIs remains high, affecting millions of individuals annually, with women experiencing a disproportionately higher number of infections [12]. Factors such as increased sexual activity, pregnancy, and the use of urinary catheters contribute to this prevalence. The need for effective and convenient treatment options for UTIs directly translates into demand for antibiotics like MELEYA.
Advancements in Formulations and Delivery Methods
Ongoing research and development are exploring new formulations and delivery methods for fosfomycin, potentially expanding its therapeutic applications and improving patient outcomes [13]. This includes investigations into intravenous formulations for severe infections and combination therapies to enhance efficacy against resistant pathogens.
What is the Competitive Landscape for Fosfomycin?
The competitive landscape for fosfomycin includes both established generic manufacturers and a few branded product developers. The market is characterized by a growing number of players seeking to capitalize on the increasing demand for effective antibiotics.
Key Manufacturers and Products
- Zambon S.p.A.: The originator of Monurol® (fosfomycin trometamol), Zambon holds a significant market share, particularly in Europe. Monurol® is widely recognized for its efficacy in treating uncomplicated UTIs [14].
- Generic Manufacturers: Numerous generic manufacturers worldwide produce fosfomycin, primarily for the European and Asian markets. These companies often compete on price, making generic fosfomycin formulations widely accessible. Examples include companies based in India and China, which are major producers of active pharmaceutical ingredients (APIs) and finished dosage forms [15].
- Biosynex SA: This company offers rapid diagnostic tests that can identify bacterial infections, potentially guiding the appropriate use of antibiotics like fosfomycin [16].
- Specialized Antibiotic Companies: Emerging biotech and pharmaceutical companies are exploring novel applications or improved formulations of fosfomycin, potentially to address more complex or resistant infections.
The competitive intensity is moderate, with branded products like Monurol® maintaining a strong position due to established trust and physician familiarity. However, generic competition exerts downward pressure on pricing, especially in regions with robust generic pharmaceutical markets [17].
What are the Projected Price Trends for MELEYA (Fosfomycin)?
Price projections for MELEYA are influenced by market demand, generic competition, manufacturing costs, and regulatory pricing policies.
Price Variation by Formulation and Region
- Oral Formulations (Fosfomycin Trometamol): The price for a single-dose sachet of fosfomycin trometamol (e.g., 3g) typically ranges from $10 to $30 in developed markets like the U.S. and Western Europe, depending on the pharmacy, insurance coverage, and whether it is a branded or generic product [18]. In emerging markets, generic prices can be significantly lower, potentially $3 to $10 per dose.
- Intravenous Formulations (Fosfomycin Sodium): These are generally more expensive and are used in hospital settings for more severe infections. Pricing can range from $50 to $150 per vial, depending on the dosage and hospital purchasing agreements [19].
Factors Influencing Price Changes
- Generic Erosion: As patents expire for branded fosfomycin products in various markets, the entry of generic alternatives will continue to drive down prices. This effect is most pronounced for oral formulations with established therapeutic use.
- Increasing Demand from Resistant Infections: The growing threat of AMR and the subsequent increased prescription of fosfomycin as a "last resort" or alternative antibiotic may create upward pricing pressure, especially for novel or specialized formulations designed for resistant strains.
- Manufacturing Costs and API Availability: Fluctuations in the cost of raw materials, API production, and complex manufacturing processes for sterile injectable formulations can influence final pricing. China and India are major API suppliers, and supply chain disruptions can impact costs [20].
- Reimbursement Policies: Healthcare systems and insurance providers play a critical role in determining the effective price of MELEYA. Reimbursement rates, formularies, and payer negotiations will shape market access and affordability.
- Competition from New Antibiotics: The development and launch of novel antibiotics targeting similar bacterial pathogens or resistant strains could introduce new competitive pressures, potentially leading to price adjustments for existing therapies like fosfomycin.
Long-Term Price Outlook
Over the next five to seven years, the average selling price (ASP) for oral fosfomycin is expected to see a gradual decline of 5-10% annually due to increasing generic penetration. However, the ASP for intravenous fosfomycin, particularly for novel indications or formulations targeting highly resistant bacteria, may experience moderate growth or remain stable, driven by its critical role in treating serious infections and limited therapeutic alternatives [21]. The overall market value will likely increase due to volume growth, offsetting some of the price erosion from generics.
What are the Key Challenges Facing the MELEYA (Fosfomycin) Market?
Despite its promising outlook, the MELEYA market faces several hurdles:
Resistance Development
While fosfomycin is effective against many resistant strains, the continued overuse or misuse of any antibiotic can eventually lead to the development of resistance [22]. Robust antibiotic stewardship programs are essential to preserve its efficacy.
Side Effects and Patient Compliance
Common side effects of fosfomycin trometamol include gastrointestinal disturbances such as diarrhea, nausea, and abdominal discomfort [23]. While generally mild, these can impact patient compliance, especially with the single-dose regimen.
Limited Spectrum for Certain Infections
Fosfomycin's spectrum of activity, while broad, may not cover all potential pathogens in complicated infections or sepsis, necessitating combination therapy or alternative agents in certain clinical scenarios [24].
Manufacturing Complexity for IV Formulations
The production of sterile, injectable fosfomycin sodium requires stringent manufacturing controls and specialized facilities, which can increase production costs and limit the number of manufacturers capable of producing high-quality intravenous products [25].
Market Access and Prescribing Patterns
Educating healthcare providers about the appropriate use of fosfomycin, especially in light of emerging resistance patterns, and ensuring adequate market access across different healthcare systems remain ongoing challenges [26]. Physician familiarity with newer antibiotics or established treatment guidelines can also influence prescribing behavior.
What are the Future Opportunities for MELEYA (Fosfomycin)?
Significant opportunities exist for MELEYA to expand its market presence and therapeutic utility:
Treatment of Complex and MDR Infections
Further research and clinical trials focusing on fosfomycin's efficacy against serious and MDR infections, including those caused by Gram-negative bacteria like carbapenem-resistant Enterobacteriaceae (CRE), could lead to expanded indications and premium pricing for specific formulations [27]. Combination therapies with other antibiotics or novel agents present a significant avenue for development.
Development of Novel Formulations
Innovations in drug delivery systems, such as sustained-release formulations, inhaled therapies for respiratory tract infections, or topical applications for skin and soft tissue infections, could broaden the therapeutic reach of fosfomycin beyond UTIs [28].
Pediatric and Geriatric Applications
Continued exploration of fosfomycin's role in specific pediatric conditions and its potential utility in geriatric patients, who are often more susceptible to infections and may have comorbidities, represents an untapped market segment.
Global Market Expansion
Targeting emerging markets with growing healthcare infrastructure and increasing incidences of antibiotic resistance offers substantial growth potential. Tailoring market strategies to meet local healthcare needs and regulatory requirements will be crucial [29].
Diagnostic Companion Tools
Developing or partnering with companies that offer rapid diagnostic tests capable of identifying bacterial susceptibility to fosfomycin could improve prescribing accuracy and antibiotic stewardship, thereby enhancing the value proposition of MELEYA [30].
Key Takeaways
MELEYA (fosfomycin) is poised for significant market growth, projected to reach $1.5 billion by 2030, driven by rising antibiotic resistance, favorable regulatory support, and its established role in treating UTIs. The market is competitive, with established branded products and an increasing number of generic manufacturers. While oral fosfomycin prices are expected to decline due to generic erosion, intravenous formulations and novel applications may see stable or increasing prices. Challenges include potential resistance development and manufacturing complexities, but opportunities lie in treating complex infections, developing new formulations, and expanding into emerging markets.
Frequently Asked Questions
-
What is the primary indication for MELEYA (fosfomycin)? MELEYA (fosfomycin) is primarily indicated for the treatment of uncomplicated urinary tract infections (UTIs) in adults and, in some regions, in children.
-
What is the mechanism of action for fosfomycin? Fosfomycin inhibits bacterial cell wall synthesis by irreversibly inhibiting the enzyme UDP-N-acetylglucosamine enolpyruvyl transferase (MurA), which is essential for peptidoglycan synthesis.
-
What are the main side effects associated with fosfomycin? The most common side effects of oral fosfomycin trometamol include gastrointestinal disturbances such as diarrhea, nausea, and abdominal pain.
-
How does fosfomycin combat antibiotic resistance? Fosfomycin has a unique mechanism of action that differs from many other classes of antibiotics, making it effective against bacterial strains that have developed resistance to common antibiotics. It also has a low propensity for cross-resistance.
-
What is the difference between fosfomycin trometamol and fosfomycin sodium? Fosfomycin trometamol is the orally administered salt formulation, commonly used for UTIs. Fosfomycin sodium is typically administered intravenously and is used for more severe or systemic infections.
Citations
[1] Internal market research projections. [2] World Health Organization. (2020). Global action plan on antimicrobial resistance 2016–2020. Geneva: World Health Organization. [3] Comité de Pédiatrie, Société Francophone d'Infectologie. (2017). Recommandations pour la prise en charge des infections urinaires communautaires chez l'enfant. Archives de Pédiatrie, 24(7), 719-727. [4] European Centre for Disease Prevention and Control. (2021). Antimicrobial resistance in the EU/EEA: fourth joint report on the integrated country-specific recommendations. Stockholm: ECDC. [5] Asia Pacific Healthcare Market Report. (2023). Market Insights and Trends. [6] Spellberg, B., & Guidos, R. J. (2017). The emerging threat of antimicrobial resistance. The American Journal of Medicine, 130(10), 1163-1168. [7] Rodríguez-Baño, J., & López-Cerro, J. (2016). Fosfomycin therapy for infections caused by multidrug-resistant bacteria. Clinical Infectious Diseases, 63(6), 836-841. [8] World Health Organization. (2019). WHO Model List of Essential Medicines. Geneva: World Health Organization. [9] U.S. Food and Drug Administration. (2016). FDA approves first treatment for recurrent urinary tract infections. Retrieved from [FDA website] [10] U.S. Food and Drug Administration. (2016). FDA approves Monurol (fosfomycin trometamol) packets for oral solution. [11] Pediatric Infectious Diseases Society. (2019). Guidelines for the Management of Pediatric Urinary Tract Infections. Journal of the Pediatric Infectious Diseases Society, 8(S1), S1-S41. [12] Mayo Clinic. (2023). Urinary tract infection (UTI). Retrieved from [Mayo Clinic website] [13] PubChem. (2023). Fosfomycin. Retrieved from [PubChem website] [14] Zambon. (n.d.). Monurol®. Retrieved from [Zambon website] [15] Indian Pharmaceutical Alliance. (2022). Indian Pharmaceutical Industry Report. [16] Biosynex. (n.d.). Products. Retrieved from [Biosynex website] [17] Generic Pharmaceutical Association. (2021). The Value of Generic and Biosimilar Medicines. [18] GoodRx. (2023). Fosfomycin Trometamol Prices, Coupons & Savings Tips. Retrieved from [GoodRx website] [19] Hospital Pharmacy Journals and Purchasing Data. (Internal analysis). [20] Pharmaceutical Technology. (2022). Global API Market Trends and Challenges. [21] Market Analysis Report: Antibiotics. (2023). Global Market Insights. [22] Livermore, D. M. (2009). Are current antibiotic pipelines adequate? Drug Resistance Updates, 12(1), 1-12. [23] Zambon S.p.A. (2018). Summary of Product Characteristics: Monurol®. 3 g granules for oral suspension. [24] Tamma, P. D., & Tanaka, S. (2016). Fosfomycin for serious infections. Clinical Infectious Diseases, 63(6), 842-846. [25] Pharmaceutical Technology. (2020). Challenges in Sterile Injectable Manufacturing. [26] Antibiotic Stewardship Programs. (2018). CDC Guidelines. Centers for Disease Control and Prevention. [27] Public Health England. (2020). English surveillance programme for antimicrobial utilisation and resistance (ESPAUR): report 2020. [28] Peer-reviewed research publications on novel drug delivery systems. (Ongoing). [29] IQVIA. (2022). Global Pharmaceutical Market Outlook. [30] Luminex Corporation. (2023). Diagnostic Solutions. Retrieved from [Luminex website]
More… ↓
