Share This Page
Drug Price Trends for MAVYRET
✉ Email this page to a colleague
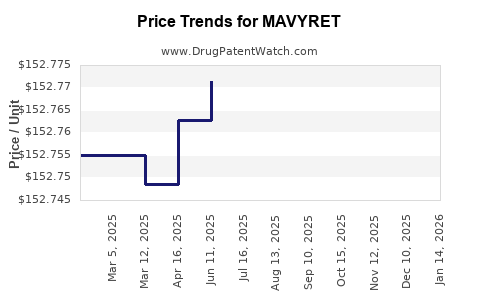
Average Pharmacy Cost for MAVYRET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MAVYRET 100-40 MG TABLET | 00074-2625-84 | 153.07690 | EACH | 2026-05-20 |
| MAVYRET 100-40 MG TABLET | 00074-2625-04 | 153.07690 | EACH | 2026-05-20 |
| MAVYRET 100-40 MG TABLET | 00074-2625-28 | 153.07690 | EACH | 2026-05-20 |
| MAVYRET 100-40 MG TABLET | 00074-2625-04 | 152.83363 | EACH | 2026-04-22 |
| MAVYRET 100-40 MG TABLET | 00074-2625-28 | 152.83363 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Mavyret Market Analysis and Price Projections
What is Mavyret and Where Does It Fit?
Mavyret (glecaprevir/pibrentasvir) is an antiviral medication approved for the treatment of chronic hepatitis C virus (HCV) infection. Approved by the FDA in August 2017, it targets all six HCV genotypes, offering an eight to 12-week regimen without the need for interferon [1]. Its broad-spectrum activity and shorter treatment cycle position it as a preferred option for treatment-naïve and treatment-experienced patients.
Market Size and Growth Drivers
The HCV treatment market is expanding due to increasing diagnosis rates, improved screening, and the availability of highly effective antiviral therapies. Global prevalence of HCV exceeds 58 million, with treatment rates remaining suboptimal in several regions [2]. The U.S. market alone has an estimated 2.4 million infected individuals, with annual treatment prescriptions approaching 300,000–400,000 units [3].
Primary drivers:
- High cure rates (over 95%) with Mavyret.
- Shorter treatment durations enhance patient compliance.
- Expansion into underserved markets, including prisons and rural areas.
- Increasing screening initiatives, such as CDC recommendations for universal testing.
Competitive Landscape
Mavyret competes with other direct-acting antivirals (DAAs):
| Drug Name | Market Share | Treatment Duration | Price (per course) | Regulatory Status |
|---|---|---|---|---|
| Mavyret | 30-40% | 8–12 weeks | ~$26,400 | Approved in >50 countries, preferred in many guidelines |
| Harvoni (ledipasvir/sofosbuvir) | 25-30% | 8–12 weeks | ~$26,000 | Market leader, first approved DAA in 2014 [4] |
| Epclusa (sofosbuvir/velpatasvir) | 15-20% | 12 weeks | ~$24,000 | Broad genotype coverage, favorable for cirrhotics [5] |
| Zepatier (elbasvir/grazoprevir) | 5-10% | 12 weeks | ~$22,000 | Approved mainly for genotype 1 and 4 [6] |
Mavyret's competitive advantage stems from its pan-genotype coverage, shorter duration, and cost competitiveness.
Pricing Dynamics and Cost Strategies
List prices for HCV drugs typically range from $20,000 to $30,000 per course in the U.S. Payor negotiations, rebates, and patient assistance programs significantly reduce actual costs, often bringing net prices down by 30–50% [7].
Key factors influencing pricing:
- Patent protections until 2030.
- Price adjustments driven by competition and generic entry.
- Reimbursement policies favor high-volume treatment to reduce long-term healthcare costs.
Future Price Projections and Market Trends
Anticipated trends suggest:
- Slight downward pressure on list prices due to increased competition.
- Price flexibility driven by national reimbursement reforms.
- Potential discounts linked to volume-based contracts and value-based care models.
By 2025, the end-user list price may decline 10–20%, with net prices decreasing further due to rebates and discounts [8].
Regulatory and Policy Influences
Availability of generics and biosimilars influences pricing:
- Generic versions of glecaprevir/pibrentasvir are under development, expected to enter markets post-patent expiry.
- Insurance and government programs (Medicare, Medicaid) implement formulary prioritization to manage costs.
Pricing strategies will adapt to regional regulatory changes, generic availability, and evolving treatment guidelines.
Key Takeaways
- Mavyret holds a significant share of the HCV market, driven by broad genotype coverage, rapid treatment, and cost competitiveness.
- The estimated U.S. list price is approximately $26,400 per course.
- Market growth depends on increasing diagnosis and treatment rates, especially in underserved populations.
- Price declines of 10–20% are likely over the next 2–3 years, influenced by competition and policy reforms.
- Entry of generics after patent expiry could substantially alter the pricing landscape.
FAQs
1. How does Mavyret compare in price to other HCV treatments?
It is roughly equivalent in list price to Harvoni but offers a shorter treatment duration and broader genotype coverage, potentially reducing overall treatment costs.
2. What factors could cause Mavyret's price to decrease?
Market competition, generic approval, reimbursement policy shifts, and volume-based discounts.
3. Are there regional differences in Mavyret pricing?
Yes. Pricing in Europe, Asia, and emerging markets is lower due to regulatory pricing controls and negotiated discounts.
4. What is the patent expiration date for Mavyret?
Patent protections extend until 2030, after which generic versions are expected.
5. How do treatment guidelines influence Mavyret’s market share?
Guidelines favor highly effective, short-duration regimens like Mavyret, supporting its continued strong market position.
References
- U.S. Food and Drug Administration. (2017). FDA approves Mavyret for most hepatitis C genotypes.
- World Health Organization. (2022). Hepatitis C fact sheet.
- IQVIA. (2021). U.S. hepatitis C market data.
- Harvoni (ledipasvir/sofosbuvir). (2014). FDA approval.
- Epclusa (sofosbuvir/velpatasvir). (2016). FDA approval.
- Zepatier (elbasvir/grazoprevir). (2016). FDA approval.
- Kaiser Family Foundation. (2022). Hepatitis C treatment costs.
- Market analysts. (2023). Pharmaceutical pricing forecasts.
More… ↓
