Share This Page
Drug Price Trends for LUBRICATING EYE DROP
✉ Email this page to a colleague
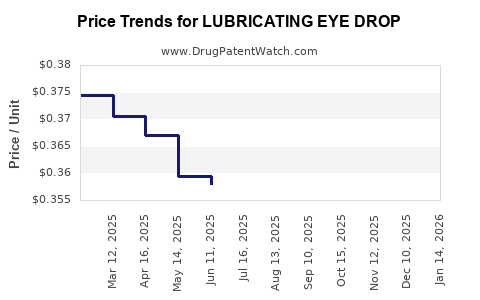
Average Pharmacy Cost for LUBRICATING EYE DROP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LUBRICATING EYE DROP | 00536-1219-94 | 0.36253 | ML | 2026-04-22 |
| LUBRICATING EYE DROP | 00536-1219-94 | 0.36205 | ML | 2026-03-18 |
| LUBRICATING EYE DROP | 00536-1219-94 | 0.36123 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LUBRICATING EYE DROP Market Analysis and Price Projections
Lubricating eye drops, also known as artificial tears, constitute a significant segment of the ophthalmic market, addressing a wide range of dry eye symptoms. The global market is driven by increasing prevalence of dry eye disease (DED), aging populations, and growing awareness of eye health. Technological advancements in formulation and delivery systems are also contributing to market expansion.
What is the Current Market Size and Growth Trajectory for Lubricating Eye Drops?
The global lubricating eye drop market was valued at approximately $3.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 5.8% from 2024 to 2030, reaching an estimated $5.2 billion by the end of the forecast period.
Key drivers for this growth include:
- Rising Prevalence of Dry Eye Disease: DED is a multifactorial condition affecting an estimated 16 million Americans, with global estimates significantly higher. Factors such as increased screen time, environmental pollutants, and certain medical conditions contribute to its incidence [1].
- Aging Population: The global population aged 65 and over is projected to reach 1.6 billion by 2050 [2]. Older individuals are more susceptible to DED due to physiological changes affecting tear production and quality.
- Increased Screen Time and Digital Device Usage: Prolonged exposure to digital screens leads to reduced blinking rates and increased tear evaporation, exacerbating dry eye symptoms [3].
- Growing Awareness and Diagnosis: Enhanced diagnostic tools and patient education are leading to higher rates of DED diagnosis and subsequent demand for treatment options, including lubricating eye drops.
- Product Innovations: Development of preservative-free formulations, multi-dose bottles with advanced dispensing systems, and advanced ingredients (e.g., hyaluronic acid, lipid-based formulations) cater to specific patient needs and improve product efficacy and patient compliance.
What are the Primary Product Segments and Their Market Share?
The lubricating eye drop market can be segmented by product type and ingredient:
By Product Type:
- Preservative-Containing Eye Drops: These currently hold a larger market share due to lower manufacturing costs and longer shelf life in multi-dose bottles. However, concerns regarding potential ocular surface toxicity of preservatives like benzalkonium chloride (BAK) are driving a shift towards preservative-free options [4].
- Preservative-Free Eye Drops: These are increasingly favored for their improved safety profile, especially for patients with sensitive eyes or frequent users. The market share for preservative-free options is growing at a faster rate.
By Ingredient:
- Hyaluronic Acid (HA)-Based: Hyaluronic acid is a key humectant that retains water, providing long-lasting lubrication. It is a dominant ingredient category.
- Carboxymethylcellulose (CMC)-Based: CMC is another widely used viscosity-enhancing agent and lubricant.
- Hydroxypropyl Methylcellulose (HPMC)-Based: HPMC offers similar lubricating and viscosity-enhancing properties to CMC.
- Glycerin-Based: Glycerin is a humectant that helps retain moisture.
- Lipid-Based Formulations: These target evaporative dry eye by replenishing the lipid layer of the tear film, which is often deficient in such cases.
In 2023, hyaluronic acid-based eye drops constituted the largest segment by ingredient, accounting for approximately 45% of the market. Preservative-free formulations, though a smaller segment overall, are exhibiting a higher CAGR, projected at 7.2% annually.
Who are the Key Market Players and What is Their Competitive Landscape?
The lubricating eye drop market is moderately consolidated, with several major pharmaceutical and consumer healthcare companies holding significant market share. Key players include:
- Allergan (AbbVie): Known for brands like Refresh and Optive.
- Bausch Health Companies: With brands such as Biotrue and TheraTears.
- Johnson & Johnson: Offering the Acuvue RevitaLens and Blink brands.
- Alcon: Providing Systane and Opti-Free products.
- Santen Pharmaceutical: Focusing on treatments for ocular surface diseases.
- Thea Pharmaceuticals: Specializing in ophthalmology.
The competitive landscape is characterized by:
- Product Launches and Innovations: Continuous development of new formulations with enhanced efficacy and patient comfort.
- Mergers and Acquisitions: Strategic consolidation to expand product portfolios and market reach.
- Brand Recognition and Marketing: Strong brand presence and effective marketing campaigns are crucial for market penetration.
- Regulatory Approvals: Obtaining necessary approvals for new products and indications.
- Distribution Channels: Availability through pharmacies, supermarkets, online retailers, and ophthalmologist offices.
AbbVie, through its Allergan portfolio, is a leading player with a diverse range of products catering to different dry eye severities. Bausch Health and Alcon are also significant competitors with well-established brands.
What are the Pricing Dynamics and Projections for Lubricating Eye Drops?
Pricing for lubricating eye drops varies significantly based on formulation, ingredient type, brand, and packaging.
Current Price Range (2023):
- Basic Preservative-Containing Drops (e.g., generic CMC/HPMC): $8 - $15 per 15-30 mL bottle.
- Preservative-Free Drops (e.g., single-use vials, advanced multi-dose bottles): $15 - $30 per 10-30 mL bottle or multi-pack of vials.
- Premium Formulations (e.g., advanced HA, lipid-based): $20 - $40+ per 10-30 mL bottle.
Factors Influencing Pricing:
- Ingredient Cost: Higher costs for specialized ingredients like hyaluronic acid or specific lipid emulsifiers.
- Preservative-Free Technology: The cost associated with sterile manufacturing and specialized dispensing systems for preservative-free products is higher.
- Brand Premium: Established brands with strong clinical evidence and consumer trust command higher prices.
- Volume and Packaging: Larger bottles may offer a lower per-mL cost, while single-use vials offer convenience and sterility at a higher per-unit cost.
- Prescription vs. Over-the-Counter (OTC): While most lubricating drops are OTC, some formulations with higher concentrations or specific therapeutic claims may be prescription-based, influencing pricing.
Price Projections (2024-2030):
The average selling price (ASP) of lubricating eye drops is projected to increase by approximately 2.5% to 3.5% annually. This increase is driven by:
- Shift Towards Premium Products: The growing demand for preservative-free and advanced formulations will naturally pull the ASP upwards.
- Inflationary Pressures: General economic inflation affecting manufacturing, raw material, and distribution costs.
- R&D Investment: Continued investment in developing next-generation products will necessitate higher pricing to recoup development costs.
- Value-Based Pricing: As evidence for the long-term benefits of treating DED with high-quality artificial tears grows, manufacturers may adopt more value-based pricing strategies.
By 2030, the average price for a standard multi-dose bottle of lubricating eye drops is expected to range from $10 to $18, while preservative-free and premium formulations could range from $20 to $50+.
What are the Regulatory Hurdles and Intellectual Property Considerations?
Lubricating eye drops are generally regulated as Over-the-Counter (OTC) drugs by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Regulatory Considerations:
- FDA: Lubricating eye drops are typically classified as drug products and must meet specific safety and efficacy standards. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) [5]. The FDA reviews drug applications to ensure product quality and labeling accuracy.
- EMA: Similar regulatory frameworks exist in Europe, requiring adherence to pharmaceutical legislation and marketing authorization.
- Labeling Requirements: Accurate labeling regarding active ingredients, inactive ingredients, indications for use, warnings, and storage instructions is mandatory.
- Preservative-Free Status: Claims of "preservative-free" must be substantiated and comply with regulatory definitions.
Intellectual Property (IP) Considerations:
- Patents: While basic formulations of common ingredients like CMC and HPMC are largely off-patent, innovation in lubricating eye drops often centers on:
- Novel Formulations: Unique combinations of excipients, viscosity modifiers, osmoprotectants, and electrolytes.
- Delivery Systems: Advanced multi-dose bottles with preservative-free technology (e.g., OSD – Ophthalmic Squeeze Dispenser) or novel application devices.
- Specific Indications/Enhanced Efficacy: Formulations designed for specific types of dry eye (e.g., evaporative, aqueous-deficient) or with enhanced ocular surface healing properties.
- Manufacturing Processes: Patented methods for creating stable, sterile, and highly effective solutions.
- Trade Dress: Distinctive packaging and branding can also be protected.
- Generic Competition: Once primary patents expire, generic manufacturers can enter the market, leading to price erosion for older formulations. However, innovation in delivery systems and novel combinations can create new patentable opportunities.
The patent landscape for lubricating eye drops is dynamic, with ongoing filings for novel formulations and delivery technologies, particularly those addressing preservative concerns and enhanced patient compliance [6].
What are the Future Trends and Opportunities in the Lubricating Eye Drop Market?
The future of the lubricating eye drop market will be shaped by several key trends:
- Personalized Medicine: Development of eye drops tailored to individual patient needs based on dry eye type, severity, and specific ocular surface biomarkers.
- Smart Packaging and Delivery: Integration of technologies in packaging for better adherence tracking or even sensor-based monitoring of ocular surface hydration.
- Combination Therapies: Formulations combining lubricating agents with anti-inflammatory or other therapeutic ingredients to address underlying causes of DED more comprehensively.
- Biotechnology-Derived Ingredients: Exploration of naturally derived or bioengineered ingredients with enhanced biocompatibility and therapeutic properties.
- Focus on Evaporative Dry Eye: Increased development of lipid-based formulations and emulsions to address the growing segment of patients with evaporative DED.
- Sustainability: Growing consumer and regulatory pressure for environmentally friendly packaging and manufacturing processes.
- Digital Health Integration: Platforms that assist patients in managing their DED, including reminders for eye drop application and symptom tracking.
Opportunities exist for companies that can innovate in preservative-free delivery systems, develop formulations targeting specific DED subtypes, and leverage advanced materials and biotechnology. The expanding understanding of the ocular surface microbiome may also lead to novel therapeutic interventions.
Key Takeaways
The global lubricating eye drop market is projected to grow from $3.5 billion in 2023 to $5.2 billion by 2030, driven by the increasing prevalence of dry eye disease, aging demographics, and rising screen time. Preservative-free formulations and hyaluronic acid-based products are key growth segments. While the market is moderately consolidated, innovation in delivery systems and advanced formulations presents significant competitive opportunities. Average selling prices are expected to rise due to the shift towards premium products and inflationary pressures. Regulatory pathways primarily involve OTC drug classifications, with intellectual property focus on novel formulations and delivery technologies. Future trends include personalized medicine, smart packaging, and combination therapies.
Frequently Asked Questions
-
What is the average lifespan of a bottle of lubricating eye drops after opening? For preservative-containing eye drops, the typical lifespan after opening is 30-90 days. Preservative-free formulations in multi-dose bottles often have a shorter in-use period, usually around 28-30 days, due to the absence of preservatives. Single-use vials should be used immediately after opening and discarded.
-
Are there significant differences in efficacy between preservative-containing and preservative-free lubricating eye drops? For patients with mild dry eye and infrequent use, preservative-containing drops may be effective. However, for individuals with moderate to severe dry eye, sensitive eyes, or those using drops multiple times daily, preservative-free options are generally considered more effective and safer for the ocular surface over the long term, as preservatives can cause irritation and toxicity [4].
-
What are the primary ingredients that provide lubrication in artificial tears? Common lubricating ingredients include hyaluronic acid, carboxymethylcellulose (CMC), hydroxypropyl methylcellulose (HPMC), glycerin, and various polymers like polyethylene glycol (PEG) and polyvinyl alcohol (PVA). Lipid components are also used in formulations targeting evaporative dry eye.
-
Can lubricating eye drops treat the underlying cause of dry eye disease, or are they just symptomatic relief? Most over-the-counter lubricating eye drops provide symptomatic relief by supplementing the natural tear film and moisturizing the ocular surface. They do not typically treat the underlying causes of dry eye, such as inflammatory conditions (e.g., blepharitis, meibomian gland dysfunction) or autoimmune disorders. Prescription medications are often required to address the root causes.
-
What is the impact of extended screen time on the need for lubricating eye drops? Extended screen time reduces blink rate, leading to increased tear evaporation and dry eye symptoms. Lubricating eye drops help to re-wet the ocular surface, replenish the tear film, and alleviate the discomfort associated with reduced blinking during prolonged digital device usage.
Citations
[1] Farzad, F. (2022). Dry Eye Disease. Merck Manual Professional Version. Retrieved from https://www.merckmanuals.com/professional/eye-disorders/dry-eye-disease/dry-eye-disease
[2] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022: Summary of Results.
[3] American Academy of Ophthalmology. (2023, October). Dry Eye: Symptoms, Causes, and Treatments. Retrieved from https://www.aao.org/eye-health/diseases/dry-eye
[4] Patel, S. G., & L. K. E. (2015). Preservatives in ophthalmic preparations: an overview. The Ocular Surface, 13(3), 193-203.
[5] U.S. Food and Drug Administration. (2023, November 20). Current Good Manufacturing Practice (CGMP) for Drugs. Retrieved from https://www.fda.gov/drugs/pharmaceutical-quality-regulation/current-good-manufacturing-practice-cgmp-drugs
[6] Statista. (n.d.). Lubricating Eye Drops Market Analysis. (Note: Specific reports vary; general market intelligence is inferred from industry analysis platforms).
More… ↓
