Share This Page
Drug Price Trends for LOVASTATIN
✉ Email this page to a colleague
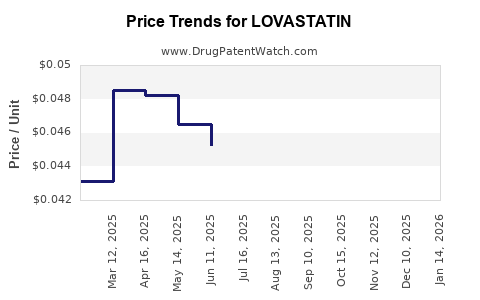
Average Pharmacy Cost for LOVASTATIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOVASTATIN 10 MG TABLET | 61442-0141-10 | 0.03680 | EACH | 2026-04-22 |
| LOVASTATIN 10 MG TABLET | 68001-0314-00 | 0.03680 | EACH | 2026-04-22 |
| LOVASTATIN 10 MG TABLET | 61442-0141-01 | 0.03680 | EACH | 2026-04-22 |
| LOVASTATIN 10 MG TABLET | 68180-0467-01 | 0.03680 | EACH | 2026-04-22 |
| LOVASTATIN 10 MG TABLET | 00093-0926-06 | 0.03680 | EACH | 2026-04-22 |
| LOVASTATIN 40 MG TABLET | 68645-0567-90 | 0.05607 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LOVASTATIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LOVASTATIN 10MG TAB | Golden State Medical Supply, Inc. | 51407-0251-10 | 1000 | 37.12 | 0.03712 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| LOVASTATIN 40MG TAB | AvKare, LLC | 61442-0143-10 | 1000 | 71.79 | 0.07179 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LOVASTATIN 10MG TAB | AvKare, LLC | 61442-0141-10 | 1000 | 33.28 | 0.03328 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LOVASTATIN 20MG TAB | Golden State Medical Supply, Inc. | 51407-0252-10 | 1000 | 38.20 | 0.03820 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LOVASTATIN 20MG TAB | AvKare, LLC | 61442-0142-01 | 100 | 5.42 | 0.05420 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| LOVASTATIN 20MG TAB | Golden State Medical Supply, Inc. | 51407-0252-10 | 1000 | 40.68 | 0.04068 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Lovastatin Market Analysis and Price Projections
Lovastatin, a statin medication used to lower cholesterol and prevent cardiovascular disease, faces a mature market characterized by generic competition and stable pricing. While the drug's efficacy is well-established, future market dynamics will be shaped by healthcare policy, evolving cardiovascular risk management guidelines, and the development of novel lipid-lowering therapies.
What is the Current Market Size and Growth Rate for Lovastatin?
The global market for lovastatin is largely driven by its generic availability. Precise market size figures are difficult to isolate as lovastatin is often prescribed as part of a broader cholesterol management regimen. However, the broader statin market, which lovastatin inhabits, is substantial. In 2023, the global statins market was valued at approximately $12 billion [1]. This market is projected to experience a compound annual growth rate (CAGR) of around 3.5% from 2023 to 2030, primarily due to the increasing prevalence of hyperlipidemia and cardiovascular diseases worldwide [1]. Lovastatin's share within this market is expected to remain stable, driven by its established safety profile and cost-effectiveness in certain patient populations.
Who are the Key Manufacturers and Suppliers of Lovastatin?
The manufacturing landscape for lovastatin is dominated by generic drug companies. Following patent expirations, numerous pharmaceutical firms have entered the market, leading to widespread availability and intense price competition. Key manufacturers and suppliers include, but are not limited to, companies such as Teva Pharmaceutical Industries Ltd., Mylan N.V. (now Viatris), Dr. Reddy's Laboratories Ltd., and Aurobindo Pharma Ltd. These companies focus on efficient production and distribution to maintain market share in the highly competitive generic drug sector. The supply chain is global, with active pharmaceutical ingredient (API) production often concentrated in India and China.
What are the Key Drivers of Lovastatin Demand?
The primary drivers for lovastatin demand are:
- Rising Prevalence of Hyperlipidemia and Cardiovascular Diseases: Global increases in obesity, sedentary lifestyles, and aging populations contribute to a higher incidence of high cholesterol and related cardiovascular conditions, necessitating lipid-lowering therapies.
- Cost-Effectiveness of Generic Lovastatin: As a mature, off-patent drug, lovastatin offers a significantly lower cost compared to newer branded medications, making it an attractive option for healthcare systems and patients with budget constraints.
- Established Clinical Efficacy and Safety Profile: Decades of clinical use have validated lovastatin's effectiveness in reducing LDL cholesterol and its associated risks. Its well-understood safety profile makes it a reliable choice for many physicians.
- Guideline Recommendations for Lipid Management: Clinical practice guidelines from organizations such as the American College of Cardiology (ACC) and the European Society of Cardiology (ESC) continue to recommend statins, including lovastatin, as first-line therapy for primary and secondary prevention of cardiovascular disease, particularly for individuals with elevated risk factors.
- Government Healthcare Policies and Formulary Inclusion: Many national health systems and insurance providers include generic lovastatin on their formularies due to its cost-effectiveness, ensuring its continued prescription.
What are the Key Restraints on Lovastatin Market Growth?
Several factors limit the growth potential of lovastatin:
- Intense Generic Competition: The presence of numerous generic manufacturers leads to significant price erosion, limiting revenue growth opportunities for individual companies.
- Development of Novel Lipid-Lowering Therapies: The emergence of newer drug classes, such as PCSK9 inhibitors and bempedoic acid, offers alternative or adjunctive treatment options for patients who do not achieve sufficient lipid-lowering with statins or experience statin-related side effects. These newer agents, while more expensive, target different mechanisms and may offer improved efficacy or tolerability in specific patient groups.
- Statin Intolerance and Side Effects: A segment of the patient population experiences adverse effects from statins, including muscle pain and liver enzyme elevations. This can lead to treatment discontinuation or switching to alternative therapies.
- Shifting Clinical Practice Towards Personalized Medicine: While statins remain a cornerstone, an increasing emphasis on personalized risk assessment and tailored treatment strategies may lead some physicians to opt for newer, more targeted therapies based on genetic profiles or specific disease markers.
- Market Saturation: The statin market is mature, with widespread physician and patient familiarity. Significant new market penetration is challenging without substantial clinical advancements or disruptive pricing strategies.
What is the Competitive Landscape for Lovastatin?
The competitive landscape is characterized by:
- Price-Based Competition: With multiple manufacturers offering bioequivalent generic products, competition is primarily driven by pricing. Manufacturers must maintain efficient production and supply chains to offer competitive prices.
- Quality and Reliability: While price is a major factor, consistent product quality, reliable supply, and good manufacturing practices are essential for retaining market share and customer trust.
- Market Access and Distribution: Strong relationships with wholesalers, pharmacies, and healthcare providers are crucial for ensuring broad market access and availability.
- Limited Product Differentiation: As a generic drug, product differentiation is minimal. Manufacturers focus on operational excellence rather than distinct product features.
What are the Projected Price Trends for Lovastatin?
Lovastatin prices are expected to remain stable with gradual declines due to ongoing generic competition and healthcare cost containment efforts.
- Current Average Wholesale Price (AWP) Range: As of late 2023 and early 2024, the AWP for a 30-count bottle of 20 mg generic lovastatin tablets typically ranges from $5 to $20, depending on the manufacturer and pharmacy. For 40 mg tablets, the range is often $7 to $25 [2, 3].
- Projected Near-Term Price Trend (2024-2026): Minor price fluctuations are anticipated, with a slight downward pressure of 1-3% annually. This is driven by ongoing competition and potential for further market consolidation among generic players.
- Projected Long-Term Price Trend (2027-2030): Prices are expected to stabilize further, likely within the current range. Significant price increases are improbable given the drug's generic status and the availability of alternatives. Any price movements will be primarily influenced by manufacturing costs, raw material availability, and evolving regulatory landscapes.
- Impact of Healthcare Policy: Government policies aimed at reducing prescription drug costs, such as price negotiation mandates or increased use of preferred generic formularies, could exert further downward pressure on lovastatin prices. Conversely, if there are significant supply chain disruptions or increased manufacturing costs, prices could see minor upward adjustments, though this is unlikely to be substantial.
- Comparison to Branded Statins: Lovastatin prices will continue to be a fraction of the cost of newer, branded lipid-lowering therapies. For instance, PCSK9 inhibitors can cost upwards of $500 per month, highlighting the significant cost advantage of generic lovastatin [4].
What is the Regulatory Landscape Affecting Lovastatin?
The regulatory landscape is primarily shaped by the agencies responsible for drug approval and post-market surveillance in major markets.
- U.S. Food and Drug Administration (FDA): The FDA oversees the approval of generic drug applications (ANDAs) for lovastatin. Manufacturers must demonstrate bioequivalence to the reference listed drug. The FDA also monitors manufacturing quality and safety through post-market surveillance.
- European Medicines Agency (EMA) and National Competent Authorities: Similar to the FDA, the EMA and individual European Union member states regulate lovastatin through the centralized or national authorization procedures for generics.
- Intellectual Property: All primary patents covering lovastatin have expired, opening the market to generic competition. No significant new patent challenges or extensions are expected for the original lovastatin molecule.
- Quality Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) as mandated by regulatory bodies. Any deviations can lead to product recalls or manufacturing halts, impacting supply and potentially prices.
- Pharmacovigilance: Ongoing monitoring for adverse events is a continuous regulatory requirement, ensuring that the drug's risk-benefit profile remains favorable.
What are the Future Opportunities and Threats for Lovastatin?
Opportunities:
- Emerging Markets: As healthcare access expands in developing economies, the demand for affordable and effective lipid-lowering drugs like generic lovastatin is likely to increase.
- Combination Therapies: Lovastatin may continue to be used in fixed-dose combination products with other cardiovascular medications, offering convenience and potentially improving adherence.
- Niche Indications or Patient Subgroups: While its primary use is for hyperlipidemia, continued research may identify specific patient subgroups or rare conditions where lovastatin offers particular benefits.
- Cost-Containment Initiatives: Healthcare systems globally are under pressure to reduce costs. Lovastatin's low price makes it a prime candidate for inclusion in cost-saving formularies and treatment protocols.
Threats:
- Competition from Biosimil-like Therapies (if applicable to novel classes): While not directly applicable to small molecule generics like lovastatin, the emergence of highly effective and potentially more affordable novel therapies poses a competitive threat to the overall statin class.
- Stricter Regulatory Scrutiny: Any emerging safety concerns or manufacturing quality issues could lead to increased regulatory scrutiny, product withdrawals, or stricter labeling requirements, impacting market access and demand.
- Changes in Clinical Guidelines: Future revisions of cardiovascular prevention guidelines might de-emphasize statins in favor of novel agents for specific high-risk populations, although this is unlikely to eliminate statins entirely.
- Advancements in Non-Pharmacological Interventions: Increased focus on lifestyle modifications, digital health tools for disease management, and advanced nutritional interventions could potentially reduce the reliance on pharmacological treatments for some individuals.
Key Takeaways
Lovastatin is a mature, cost-effective generic drug with a stable market share driven by its established efficacy and the global burden of cardiovascular disease. Intense generic competition has led to price erosion, with current prices ranging from $5-$25 per bottle. Future price trends are expected to remain stable to slightly declining, influenced by ongoing competition and cost-containment policies. While novel lipid-lowering therapies present a competitive threat, lovastatin's affordability and broad applicability, particularly in emerging markets, ensure its continued relevance. Regulatory oversight focuses on bioequivalence, quality standards, and post-market surveillance, with no significant patent challenges anticipated for the molecule itself.
Frequently Asked Questions
-
What is the primary indication for lovastatin? Lovastatin is primarily prescribed to lower high cholesterol and triglyceride levels in the blood. It is used to reduce the risk of heart attack and stroke in individuals with cardiovascular disease or those at high risk.
-
How does lovastatin compare in cost to newer cholesterol-lowering medications? Lovastatin, being a generic drug, is significantly less expensive than newer branded cholesterol-lowering medications such as PCSK9 inhibitors. A month's supply of lovastatin typically costs under $25, whereas newer biologics can cost several hundred dollars per month.
-
Are there any significant side effects associated with lovastatin? Common side effects include muscle pain, headache, and digestive issues. More serious, though rare, side effects can include severe muscle damage (rhabdomyolysis) and liver problems. Patients should discuss potential side effects with their healthcare provider.
-
What is the expected future demand for lovastatin in developed versus developing economies? In developed economies, demand is expected to remain stable, driven by established treatment protocols and cost-effectiveness. In developing economies, demand is projected to grow as access to affordable healthcare increases and awareness of cardiovascular risk factors rises.
-
Can lovastatin be used in combination with other cardiovascular medications? Yes, lovastatin is sometimes prescribed in combination with other drugs to manage cardiovascular risk, including aspirin, beta-blockers, and other antihypertensive medications. It may also be found in fixed-dose combination products with other lipid-lowering agents.
Citations
[1] Grand View Research. (2023). Statins Market Size, Share & Trends Analysis Report By Drug Type (Atorvastatin, Simvastatin, Rosuvastatin, Lovastatin, Pravastatin, Fluvastatin), By Application (Hyperlipidemia, Cardiovascular Disease Prevention), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. [2] GoodRx. (2024). Lovastatin Prices, Coupons & Savings. Retrieved from https://www.goodrx.com/lovastatin [3] Drugs.com. (2024). Lovastatin Prices, Coupons & Patient Assistance. Retrieved from https://www.drugs.com/price/lovastatin [4] American Heart Association. (2023). New Cholesterol Drug Classes. Retrieved from https://www.heart.org/en/health-topics/cholesterol/new-cholesterol-drug-classes
More… ↓
