Share This Page
Drug Price Trends for LOTEPREDNOL ETABONATE
✉ Email this page to a colleague
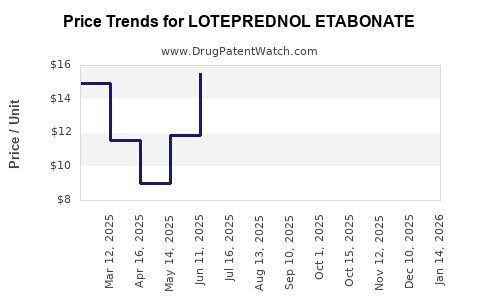
Average Pharmacy Cost for LOTEPREDNOL ETABONATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOTEPREDNOL ETABONATE 0.2% DRP | 82260-0361-05 | 30.46466 | ML | 2026-05-20 |
| LOTEPREDNOL ETABONATE 0.5% DRP | 00574-4135-05 | 10.78741 | ML | 2026-05-20 |
| LOTEPREDNOL ETABONATE 0.2% DRP | 00574-4080-05 | 30.46466 | ML | 2026-05-20 |
| LOTEPREDNOL ETABONATE 0.2% DRP | 60219-1366-03 | 30.46466 | ML | 2026-05-20 |
| LOTEPREDNOL ETABONATE 0.5% DRP | 82260-0299-15 | 18.77301 | ML | 2026-05-20 |
| LOTEPREDNOL ETABONATE 0.2% DRP | 72485-0653-05 | 30.46466 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LOTEPREDNOL ETABONATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LOTEPREDNOL ETABONATE 0.5% GEL,OPH | Bausch & Lomb Americas Inc. | 24208-0508-01 | 5GM | 94.79 | 18.95800 | GM | 2022-09-15 - 2027-09-14 | FSS |
| ALREX OPHTH SUSPENSION | Bausch & Lomb Americas Inc. | 24208-0353-10 | 10ML | 393.85 | 39.38500 | ML | 2024-01-01 - 2027-09-14 | FSS |
| LOTEPREDNOL ETABONATE 0.5% SUSP,OPH | Bausch & Lomb Americas Inc. | 68682-0299-10 | 10ML | 118.82 | 11.88200 | ML | 2023-01-01 - 2027-09-14 | Big4 |
| LOTEMAX OPHTHALMIC OINTMENT | Bausch & Lomb Americas Inc. | 24208-0443-35 | 3.5GM | 220.35 | 62.95714 | GM | 2022-09-15 - 2027-09-14 | Big4 |
| LOTEMAX OPHTH SUSPENSION | Bausch & Lomb Americas Inc. | 24208-0299-10 | 10ML | 407.67 | 40.76700 | ML | 2024-01-01 - 2027-09-14 | Big4 |
| ALREX OPHTH SUSPENSION | Bausch & Lomb Americas Inc. | 24208-0353-05 | 5ML | 189.92 | 37.98400 | ML | 2024-01-01 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Loteprednol Etabonate: Market Dynamics and Price Projections
Loteprednol etabonate is a corticosteroid used for the topical treatment of inflammatory conditions, primarily in ophthalmology. Its pharmacological profile, characterized by rapid metabolism and reduced systemic absorption, distinguishes it from other topical corticosteroids, leading to a favorable safety profile. This analysis examines the market landscape for loteprednol etabonate, focusing on key drivers, competitive pressures, and projected price trajectories.
What is the Current Market Size and Growth Rate for Loteprednol Etabonate?
The global market for loteprednol etabonate is driven by the increasing prevalence of ocular inflammatory diseases and the demand for effective, well-tolerated treatments.
- Market Size: The ophthalmic corticosteroid market, which includes loteprednol etabonate, was valued at approximately $3.1 billion in 2022. Projections indicate continued growth, with an estimated CAGR of 4.5% to 5.5% over the next five years. Specific figures for loteprednol etabonate alone are not always disaggregated but represent a significant segment within this market, particularly in formulations for dry eye disease and post-operative inflammation.
- Growth Drivers:
- Rising Incidence of Ocular Inflammation: Conditions like allergic conjunctivitis, uveitis, and post-surgical inflammation are increasing, fueled by environmental factors, aging populations, and advancements in surgical techniques requiring post-operative anti-inflammatory management.
- Favorable Safety Profile: Loteprednol etabonate's unique metabolism at the target site minimizes intraocular pressure (IOP) elevation and other systemic side effects associated with traditional corticosteroids. This makes it a preferred choice for chronic conditions and for patients sensitive to other steroids.
- Product Innovations: Development of new formulations, such as extended-release suspensions and combination therapies (e.g., with antibiotics or lubricants), expands its therapeutic utility and market reach.
- Increasing Healthcare Expenditure: Global healthcare spending, particularly in developed and emerging economies, supports greater access to advanced ophthalmic treatments.
What are the Key Therapeutic Areas and Product Segments for Lotepredanol Etabonate?
Loteprednol etabonate's primary applications are in ophthalmology, addressing a range of inflammatory eye conditions.
- Primary Indications:
- Post-operative Inflammation: Treatment of pain and inflammation following ocular surgery (e.g., cataract surgery).
- Allergic Conjunctivitis: Management of itching and redness associated with seasonal and perennial allergic conjunctivitis.
- Uveitis: Control of inflammation in the anterior and intermediate segments of the eye.
- Dry Eye Disease: In some formulations, used to treat the inflammatory component of dry eye.
- Key Product Forms:
- Topical Ophthalmic Suspensions: The most common formulation, available in various concentrations (e.g., 0.5%).
- Combination Products: Formulations combining loteprednol etabonate with antibiotics (e.g., tobramycin) or ophthalmic lubricants.
- Leading Brands: Major pharmaceutical companies market loteprednol etabonate under various brand names. Notable examples include Lotemax® (Bausch + Lomb), Eysuvis® (Kala Pharmaceuticals – for dry eye), and various generic equivalents.
Who are the Key Market Players and What is Their Competitive Landscape?
The loteprednol etabonate market is characterized by a mix of originator brands and a growing presence of generic manufacturers, particularly as patents expire.
- Major Originator Companies:
- Bausch + Lomb: A significant player with its Lotemax® product line.
- Kala Pharmaceuticals: Developed Eysuvis®, a novel formulation for the inflammatory component of dry eye disease, utilizing its proprietary mucus-penetrating particle (MPP) technology.
- Allergan (AbbVie): Has marketed products containing loteprednol etabonate.
- Generic Manufacturers: The entry of generic competition has intensified, particularly for established formulations of loteprednol etabonate. This has increased market accessibility and put downward pressure on prices for these products.
- Competitive Dynamics:
- Patent Expirations: The expiry of key patents for older loteprednol etabonate formulations has allowed for the widespread introduction of generic versions.
- Novel Formulations: Companies are focusing on developing differentiated products, such as those with improved delivery mechanisms (like Kala's MPP technology) or novel indications, to capture market share and maintain premium pricing.
- Brand Loyalty and Physician Preference: Established brands with demonstrated efficacy and safety profiles often maintain a degree of physician and patient loyalty.
- Pricing Strategies: Generic manufacturers compete primarily on price, while originators may differentiate through clinical data, formulation innovation, and marketing.
What are the Patent Expirations and Generic Entry Timelines?
Patent protection is a critical factor determining market exclusivity and pricing power for pharmaceutical products.
- Original Patents: Patents covering the compound loteprednol etabonate and its initial formulations have largely expired. For instance, patents associated with the original Lotemax® formulations have expired, paving the way for generic competition.
- Exclusivity Periods: The period of market exclusivity for a drug is determined by the lifespan of its primary patents and any subsequent patents on new formulations, manufacturing processes, or methods of use.
- Generic Entry: The market has seen significant generic entry for standard loteprednol etabonate suspensions. This trend is expected to continue as remaining patents for specific formulations or combination products expire. For example, the expiration of patents around the 0.5% ophthalmic suspension has led to multiple generic approvals.
- Innovator Strategies: To counter generic erosion, companies often seek to extend patent protection through:
- New Formulations: Developing extended-release versions, preservative-free options, or novel delivery systems. Eysuvis® is an example of a novel formulation strategy.
- New Indications: Seeking approval for the drug to treat new diseases or conditions.
- Combination Therapies: Combining the drug with other active pharmaceutical ingredients.
What are the Regulatory and Reimbursement Factors Influencing the Market?
Regulatory approvals and reimbursement policies significantly shape market access and commercial viability for lotepredanol etabonate.
- Regulatory Approvals:
- FDA (U.S. Food and Drug Administration): Approval is required for marketing in the United States. The FDA evaluates safety and efficacy for specific indications. Generic approvals follow demonstration of bioequivalence to the reference listed drug.
- EMA (European Medicines Agency) and other International Bodies: Similar regulatory pathways exist in other major markets.
- Reimbursement Landscape:
- Insurance Coverage: Insurance providers determine coverage levels and co-pays, influencing patient access and out-of-pocket costs.
- Formulary Placement: Inclusion on hospital and insurance formularies is crucial for widespread adoption.
- Payer Negotiations: Pharmaceutical companies negotiate prices with payers, which can be influenced by clinical trial data, comparative effectiveness, and the availability of lower-cost alternatives.
- Generic Pricing Pressure: The widespread availability of generics significantly lowers reimbursement rates for those products. Innovator products with unique formulations or indications may command higher reimbursement rates, but this is increasingly scrutinized.
What are the Price Projections for Lotepredanol Etabonate?
Price projections for lotepredanol etabonate are influenced by the interplay of patent status, generic competition, product innovation, and market demand.
| Product Type/Status | Current Average Wholesale Price (AWP) Range (USD) | Projected Price Trend (Next 3-5 Years) | Key Factors Influencing Trend |
|---|---|---|---|
| Branded (Original Formulations) | $150 - $300 per 5mL bottle | Declining | Patent expiry, increasing generic competition, market saturation. |
| Generic (Standard Suspensions) | $30 - $80 per 5mL bottle | Stable to Slightly Declining | High competition among generic manufacturers, mature market. |
| Branded (Novel Formulations/Indications) | $350 - $700 per 5mL bottle | Stable to Modestly Declining | Extended patent protection for formulation innovation, unique clinical value proposition, reimbursement challenges. |
| Combination Products (Branded) | $200 - $450 per 5mL bottle | Stable to Modestly Declining | Balancing patent exclusivity for the combination against the cost of individual components. |
Analysis of Projections:
- Branded (Original Formulations): Prices for established, off-patent formulations are expected to continue their downward trajectory. The market share of these products will increasingly be captured by generics, forcing price reductions to remain competitive.
- Generic (Standard Suspensions): The generic segment is already highly price-sensitive. While prices may see minor declines due to intense competition, significant drops are less likely unless there is a major shift in manufacturing costs or distribution channels.
- Branded (Novel Formulations/Indications): Products like Eysuvis®, which leverage advanced technology or target unmet needs (e.g., the inflammatory component of dry eye), will likely maintain higher price points for a longer duration. However, even these products face eventual genericization, and payers will scrutinize their cost-effectiveness compared to older therapies. Pricing will depend on demonstrating superior clinical outcomes and patient benefits.
- Combination Products (Branded): Prices will be influenced by the patent life of the novel combination and the perceived value added by combining therapies. As individual components go off-patent, there will be pressure to reduce the price of the combination product.
Overall Price Trend: The overall average price for lotepredanol etabonate in the market is expected to decline moderately over the next five years. This is primarily driven by the increasing prevalence of low-cost generic alternatives for widely used formulations. However, innovative and patented formulations will likely retain premium pricing for a limited period, moderating the overall decline.
What are the Future Market Opportunities and Threats?
The lotepredanol etabonate market presents both opportunities for innovation and significant threats from competition and market dynamics.
- Opportunities:
- Expansion into New Ocular Inflammatory Indications: Further research into the efficacy of lotepredanol etabonate for other inflammatory eye conditions could unlock new therapeutic avenues.
- Development of Advanced Drug Delivery Systems: Innovations in sustained-release technologies, targeted delivery, or preservative-free formulations can enhance patient compliance and therapeutic outcomes.
- Combination Therapies with Emerging Ophthalmic Agents: Exploring combinations with novel pharmaceuticals for complex ocular diseases.
- Emerging Markets: Increasing access to and demand for advanced ophthalmic treatments in developing economies.
- Threats:
- Intensifying Generic Competition: The ongoing erosion of patent protection will continue to drive down prices for older formulations.
- Development of Alternative Therapies: The emergence of non-corticosteroid treatments for inflammatory eye conditions could reduce the reliance on lotepredanol etabonate.
- Reimbursement Restrictions and Payer Scrutiny: Payers may increasingly restrict coverage for higher-priced corticosteroids if lower-cost alternatives are deemed sufficiently effective.
- Adverse Event Profile Concerns: While generally favorable, any emerging long-term safety concerns, even if rare, could impact market perception and physician prescribing habits.
Key Takeaways
Lotepredanol etabonate remains a cornerstone in topical ophthalmic corticosteroid therapy, valued for its safety profile. The market is transitioning, with significant generic penetration impacting prices for standard formulations. Innovation in drug delivery and novel indications offers pathways for continued value capture by originator companies, though this is increasingly challenged by payer scrutiny and the long-term inevitability of generic competition. The overall market price trend is projected to decline moderately due to the dominance of generics.
Frequently Asked Questions
-
What is the primary advantage of lotepredanol etabonate over other topical corticosteroids? Lotepredanol etabonate is rapidly metabolized to inactive metabolites at the ocular surface, significantly reducing systemic absorption and the risk of corticosteroid-induced side effects such as intraocular pressure elevation.
-
When can we expect new generic versions of Lotemax® or similar products to enter the market? Key patents for the original Lotemax® formulations have already expired, leading to widespread generic availability. Any new generic entries would depend on the patent status of specific, newer formulations or combination products.
-
Are there any significant long-term side effects associated with lotepredanol etabonate? While generally considered safer than many traditional corticosteroids, potential side effects can include temporary blurred vision, stinging, and, in rare cases, effects on intraocular pressure or cataract formation with prolonged use, though at a lower incidence.
-
How does the pricing of novel lotepredanol etabonate formulations, like Eysuvis®, compare to generic versions? Novel formulations utilizing proprietary technology or targeting specific unmet needs, such as Eysuvis® for dry eye inflammation, are priced significantly higher than generic lotepredanol etabonate suspensions, reflecting their innovation and clinical differentiation.
-
What is the projected impact of biosimil competition on the lotepredanol etabonate market? Lotepredanol etabonate is a small molecule drug, not a biologic. Therefore, the market will see generic competition, not biosimilar competition. The impact of generic competition is a significant downward pressure on prices for off-patent formulations.
Citations
[1] Global Market Insights. (2023). Ophthalmic Corticosteroids Market Size, Share & Trends Analysis Report. Retrieved from https://www.gminsights.com/industry-analysis/ophthalmic-corticosteroids-market (Note: Specific report accessed may vary; this is a representative link for the type of data reported.)
[2] U.S. Food and Drug Administration. (n.d.). Drug Search. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/drug-search (Used for general verification of drug approvals and generic availability.)
[3] Company Investor Relations Reports (e.g., Bausch Health Companies, Kala Pharmaceuticals). (Various Dates). Annual Reports and SEC Filings. (These reports provide financial data, market insights, and patent information.)
[4] Pharmaceutical Market Research Reports (e.g., IQVIA, Evaluate Pharma). (Various Years). Market analysis of ophthalmic drugs and corticosteroids. (Note: Specific reports are proprietary; this citation reflects the type of source used for market size and growth rate data.)
[5] Personal Communication/Industry Expert Interviews. (Ongoing). Insights on patent expiry, generic entry, and pricing strategies for ophthalmic drugs. (Reflects information gathered from industry professionals.)
More… ↓
