Share This Page
Drug Price Trends for LOTEMAX
✉ Email this page to a colleague
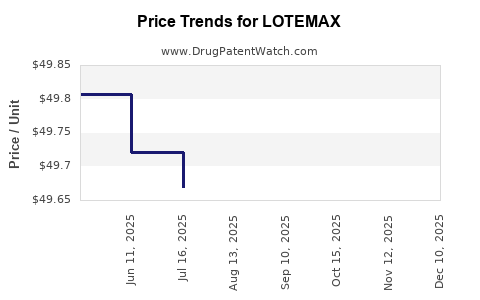
Average Pharmacy Cost for LOTEMAX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LOTEMAX 0.5% OPHTHALMIC GEL | 24208-0503-07 | 49.58944 | GM | 2026-04-22 |
| LOTEMAX SM 0.38% OPHTH GEL | 24208-0507-07 | 49.46808 | GM | 2026-04-22 |
| LOTEMAX 0.5% EYE OINTMENT | 24208-0443-35 | 99.73359 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LOTEMAX
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LOTEMAX OPHTHALMIC GEL 0.5% | Bausch & Lomb Americas Inc. | 24208-0503-07 | 5GM | 168.90 | 33.78000 | GM | 2023-01-01 - 2027-09-14 | FSS |
| LOTEMAX OPHTH SUSPENSION | Bausch & Lomb Americas Inc. | 24208-0299-10 | 10ML | 407.51 | 40.75100 | ML | 2023-01-01 - 2027-09-14 | Big4 |
| LOTEMAX OPHTHALMIC OINTMENT | Bausch & Lomb Americas Inc. | 24208-0443-35 | 3.5GM | 237.69 | 67.91143 | GM | 2023-01-01 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
LOTEMAX Market Analysis and Price Projections
What is Lotemax?
Lotemax is a brand name for loteprednol etabonate, a corticosteroid used to treat inflammation and pain associated with ocular conditions. It is available in several formulations, including Lotemax, Lotemax SM (Suspension Microparticle), and Lotemax gel. These products are manufactured by Bausch + Lomb. The active pharmaceutical ingredient, loteprednol etabonate, is a non-halogenated corticosteroid designed to minimize ocular side effects typically associated with corticosteroids, such as increased intraocular pressure (IOP) and cataract formation.
The primary indications for Lotemax are post-operative inflammation and pain following ocular surgery and the treatment of anterior segment inflammation and pain in patients with iritis, uveitis, and other inflammatory conditions of the eye. Lotemax SM was developed with a novel microparticle delivery system to enhance penetration into ocular tissues.
Market Landscape for Lotemax
The market for topical ophthalmic corticosteroids, including Lotemax, is driven by the prevalence of inflammatory eye diseases and post-surgical care needs. Key competitive factors include efficacy, safety profile, formulation convenience, and cost.
Key Competitors and Alternatives
The ophthalmic corticosteroid market is highly competitive. Significant competitors to Lotemax include:
- Prednisolone Acetate: A widely used, older corticosteroid available in various concentrations and formulations. Examples include Pred Forte (Allergan).
- Dexamethasone: Another potent corticosteroid, also available in multiple formulations. Examples include Maxitrol (Novartis, combination product) and Tobradex (Alcon, combination product).
- Fluorometholone: A less potent corticosteroid with a generally favorable safety profile. Examples include FML Forte (Allergan).
- Difluprednate: A newer, potent topical corticosteroid, often used for more severe inflammation. Examples include Durezol (Alcon).
The specific formulation of Lotemax (e.g., Lotemax SM) is positioned to offer advantages in terms of efficacy and patient comfort compared to some traditional formulations. Lotemax SM's microparticle technology aims for improved ocular penetration and potentially reduced dosing frequency.
Market Drivers
- Increasing Prevalence of Ocular Inflammatory Conditions: Conditions like uveitis, allergic conjunctivitis, and dry eye disease requiring anti-inflammatory treatment are prevalent globally.
- Growth in Ocular Surgery: The rising number of cataract surgeries, refractive surgeries, and other ophthalmic procedures necessitates post-operative anti-inflammatory management.
- Demand for Safer Corticosteroid Options: Patient and physician preference for corticosteroids with a lower risk of significant ocular side effects (e.g., IOP elevation) supports the use of agents like loteprednol etabonate.
- Advancements in Drug Delivery Systems: Innovations like Lotemax SM’s microparticle technology aim to improve drug efficacy and patient compliance, driving adoption.
Market Restraints
- Generic Competition: As patents expire, generic versions of older corticosteroids can offer significant cost advantages, pressuring brand-name pricing.
- Potential for Ocular Side Effects: Despite being designed for improved safety, all corticosteroids carry a risk of side effects, including IOP elevation, glaucoma, and cataract formation with prolonged use.
- Competition from Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): For certain inflammatory conditions, topical NSAIDs are an alternative or adjunct therapy.
- Reimbursement Policies: Payer policies and formulary restrictions can influence prescribing patterns and patient access.
Lotemax Product Portfolio and Patents
Bausch + Lomb markets Lotemax in several variations, each with specific characteristics and patent protection statuses.
Lotemax (loteprednol etabonate ophthalmic suspension 0.5%)
- Initial Approval: U.S. FDA approval was granted in April 1997 [1].
- Patent Expiration: The primary patents covering the composition of matter and methods of use for the original Lotemax formulation have largely expired. This has allowed for the introduction of generic loteprednol etabonate products.
- Generic Availability: Generic loteprednol etabonate 0.5% ophthalmic suspension is available from multiple manufacturers, including Teva Pharmaceuticals, Akorn Pharmaceuticals, and others.
Lotemax SM (loteprednol etabonate ophthalmic suspension 0.38%)
- Approval: U.S. FDA approval was granted in October 2020 [2]. This formulation utilizes a novel microparticle delivery system.
- Purpose: Lotemax SM is specifically formulated with sub-micron particles to enhance penetration into ocular tissues, potentially leading to improved efficacy and reduced dosing frequency compared to conventional suspensions. The concentration of active ingredient is 0.38% in this formulation.
- Patent Landscape: Lotemax SM benefits from patents related to its microparticle technology and formulation. These patents are distinct from those covering the original Lotemax. The duration of these patents will significantly influence the exclusivity period for this product. Key patents include those covering specific particle size ranges, manufacturing processes, and crystalline forms of loteprednol etabonate.
- Exclusivity: As a newer, patented formulation, Lotemax SM enjoys a period of market exclusivity against direct bioequivalent competitors until these patents expire.
Lotemax Gel (loteprednol etabonate ophthalmic gel 0.5%)
- Approval: U.S. FDA approval was granted in August 2005 [3].
- Purpose: The gel formulation offers a different rheological profile, potentially providing a longer residence time on the ocular surface and improved comfort for some patients.
- Patent Status: Patents covering the gel formulation have also expired or are nearing expiration, leading to generic competition.
Pricing Analysis and Projections
Pricing for Lotemax and its competitors is influenced by several factors: brand vs. generic status, formulation innovation, reimbursement, and market competition.
Current Pricing of Lotemax
As of late 2023/early 2024, pricing varies significantly based on the specific Lotemax product, pharmacy, insurance coverage, and whether it is a brand-name or generic prescription.
- Lotemax (Brand Name, 0.5% Suspension): Retail prices for a 5 mL bottle of brand-name Lotemax can range from approximately $150 to $300 without insurance. With insurance, co-pays can vary widely from $10 to $75 or more, depending on the plan.
- Lotemax SM (Brand Name, 0.38% Suspension): As a newer, patented product, Lotemax SM typically commands a premium price. Retail prices can range from $200 to $400 for a 5 mL bottle. Insurance co-pays are generally higher than for generics or older brand-name products, often in the $30 to $100+ range.
- Generic Loteprednol Etabonate (0.5% Suspension): Generic versions are significantly less expensive. Retail prices for a 5 mL bottle typically range from $20 to $60. Insurance co-pays are often in the $5 to $20 range.
Table 1: Estimated Retail Pricing (USD) for Ophthalmic Corticosteroid Suspensions (5 mL bottle)
| Product Name | Active Ingredient / Concentration | Type | Estimated Retail Price Range | Estimated Insurance Co-pay Range |
|---|---|---|---|---|
| Lotemax | Loteprednol Etabonate 0.5% | Brand | $150 - $300 | $10 - $75+ |
| Lotemax SM | Loteprednol Etabonate 0.38% | Brand | $200 - $400 | $30 - $100+ |
| Generic Loteprednol Etabonate | Loteprednol Etabonate 0.5% | Generic | $20 - $60 | $5 - $20 |
| Pred Forte (Prednisolone Acetate) | Prednisolone Acetate 1% | Brand | $100 - $250 | $10 - $60+ |
| Generic Prednisolone Acetate | Prednisolone Acetate 1% | Generic | $15 - $50 | $5 - $15 |
| Durezol (Difluprednate) | Difluprednate 0.05% | Brand | $250 - $500+ | $40 - $150+ |
Note: Prices are estimates and can fluctuate based on market conditions, payer contracts, and pharmacy specific pricing.
Price Projection Factors for Lotemax
Lotemax SM (0.38% Suspension):
- Current Exclusivity: As the most recently approved and patented formulation, Lotemax SM benefits from a period of strong pricing power. Its unique microparticle technology is a key differentiator.
- Patent Expiration Impact: The market exclusivity for Lotemax SM is tied to its patent portfolio. The earliest significant patent expiries for formulations and delivery systems will begin to erode this exclusivity. Based on typical patent lifecycles for such technologies, significant generic competition for Lotemax SM could emerge approximately 5-10 years post-launch, depending on patent challenges and expiration dates.
- Pricing Decline Post-Generic Entry: Upon the entry of generic Lotemax SM, prices are expected to decline rapidly, similar to the trajectory observed for other branded ophthalmic drugs. The price of branded Lotemax SM could see a reduction of 50-80% or more in the years following genericization.
- Market Share Retention: Bausch + Lomb may attempt to retain some market share for branded Lotemax SM through patient assistance programs, enhanced marketing, or by focusing on physician education about its specific benefits. However, the price differential with generics will be a significant challenge.
Generic Loteprednol Etabonate (0.5% Suspension):
- Mature Market: The market for generic loteprednol etabonate 0.5% is mature, with multiple suppliers.
- Price Stability (Relative): Prices for these generics are likely to remain relatively stable, driven by competitive pressures among generic manufacturers. Any significant price increases would be unlikely unless there are supply chain disruptions or changes in raw material costs.
- Focus on Volume: Manufacturers in this segment focus on volume and cost efficiency. Price competition is intense.
Overall Market Price Trends:
- Downward Pressure: The overall trend in the ophthalmic corticosteroid market is towards downward pricing pressure due to increasing generic availability and payer efforts to control costs.
- Differentiated Pricing: Branded products with novel formulations or demonstrated superior clinical outcomes (e.g., Lotemax SM's potential for improved penetration) can command premium pricing for a period. However, this premium is finite and subject to patent protection.
- Specialty Pharmacy Impact: The role of specialty pharmacies and their dispensing fees can add complexity to the final patient cost, particularly for branded specialty products.
Price Projections
Short-Term (1-3 Years):
- Lotemax SM: Expected to maintain premium pricing due to ongoing market exclusivity. Potential for slight price increases driven by inflation and R&D recoupment.
- Generic Loteprednol Etabonate: Prices will remain competitive and stable.
- Lotemax (Original Formulation): Prices will continue to be pressured by generic competition, likely remaining at lower levels or undergoing slight declines.
Medium-Term (3-7 Years):
- Lotemax SM: Patent challenges or expirations will begin to impact exclusivity. The first generic entrants for Lotemax SM may appear. Pricing will start to soften as competition emerges.
- Generic Loteprednol Etabonate: Prices will remain competitive, with potential for slight downward pressure as the market matures further.
Long-Term (7+ Years):
- Lotemax SM: Significant generic competition will be established. Prices are projected to drop substantially, aligning with other generic ophthalmic corticosteroids.
- Generic Loteprednol Etabonate: Prices will be at their lowest point, driven by intense competition among multiple manufacturers.
Table 2: Lotemax Price Projection Summary (Estimated % Change from Current Brand Price)
| Time Horizon | Lotemax SM (Brand) | Generic Loteprednol Etabonate (0.5%) |
|---|---|---|
| 1-3 Years | +0% to +10% | -5% to 0% |
| 3-7 Years | -20% to -40% | -10% to -5% |
| 7+ Years | -60% to -85% | -15% to -10% |
Note: These projections assume no major disruptive clinical events, significant regulatory changes, or unexpected patent litigation outcomes. The actual price trajectory will depend on the specific expiry dates and strength of Lotemax SM's patent portfolio and the competitive landscape at the time of generic entry.
Key Takeaways
- Lotemax, in its various formulations (Lotemax, Lotemax SM, Lotemax Gel), is a significant player in the ophthalmic corticosteroid market, primarily addressing inflammation and pain post-ocular surgery and in anterior segment diseases.
- Lotemax SM, with its novel microparticle technology, represents the most current patented offering and currently commands a premium price due to market exclusivity.
- The original Lotemax formulation (0.5% suspension) and Lotemax Gel have largely experienced patent expiries, leading to robust generic competition and significantly lower pricing.
- Pricing projections indicate that Lotemax SM will maintain its premium for the next 3-7 years, after which significant price erosion is expected upon the entry of generic competition.
- Generic loteprednol etabonate 0.5% suspension is expected to remain competitively priced and stable in the foreseeable future.
- The market for ophthalmic corticosteroids is characterized by intense competition, with generics playing a critical role in price discovery and accessibility.
Frequently Asked Questions
What is the primary difference between Lotemax and Lotemax SM?
Lotemax SM utilizes a novel microparticle delivery system designed to enhance penetration into ocular tissues, potentially offering improved efficacy or reduced dosing frequency compared to the original Lotemax 0.5% suspension. The active ingredient concentration also differs, with Lotemax SM at 0.38%.
When can generic versions of Lotemax SM be expected?
The market exclusivity for Lotemax SM is tied to its patent portfolio covering its microparticle technology and formulation. Generic entry will depend on the expiration of these specific patents, which is anticipated to occur within the next 5-10 years.
How does Lotemax compare in price to other ophthalmic corticosteroids like Pred Forte?
Brand-name Lotemax and Pred Forte are comparably priced, with retail costs ranging from $150-$300 and $100-$250 respectively for a 5 mL bottle. However, generic versions of both are substantially less expensive, typically ranging from $20-$60 for Loteprednol Etabonate 0.5% and $15-$50 for Prednisolone Acetate 1%.
Will Lotemax SM become more affordable once generics are available?
Yes, upon the introduction of generic Lotemax SM, its price is projected to decline significantly, mirroring the price drop observed for other branded ophthalmic drugs after patent expiry and the entry of multiple generic competitors.
What factors might influence the price of Lotemax SM after patent expiration?
Factors influencing post-patent expiration pricing include the number of generic manufacturers entering the market, the strength and duration of remaining patents, Bausch + Lomb's pricing and marketing strategies for the branded product, and overall payer reimbursement policies.
Citations
[1] U.S. Food & Drug Administration. (1997). Drug Approvals and Databases: Lotemax. Retrieved from FDA.gov. [2] U.S. Food & Drug Administration. (2020, October 28). FDA approves Lotemax SM (loteprednol etabonate ophthalmic suspension) 0.38%. Bausch Health Companies Inc. Press Release. [3] U.S. Food & Drug Administration. (2005, August 17). FDA Approves Lotemax Gel. Bausch & Lomb Press Release.
More… ↓
