Share This Page
Drug Price Trends for LORAZEPAM INTENSOL
✉ Email this page to a colleague
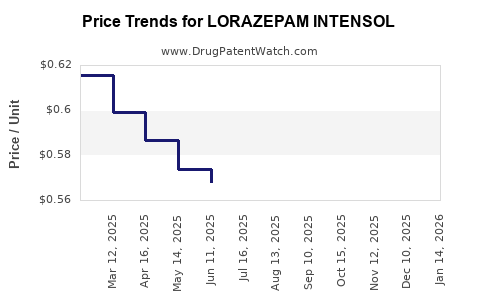
Average Pharmacy Cost for LORAZEPAM INTENSOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LORAZEPAM INTENSOL 2 MG/ML | 00054-3532-44 | 0.63219 | ML | 2026-05-20 |
| LORAZEPAM INTENSOL 2 MG/ML | 00054-3532-44 | 0.62142 | ML | 2026-04-22 |
| LORAZEPAM INTENSOL 2 MG/ML | 00054-3532-44 | 0.63685 | ML | 2026-03-18 |
| LORAZEPAM INTENSOL 2 MG/ML | 00054-3532-44 | 0.63309 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LORAZEPAM INTENSOL: Market analysis and price projections
What is Lorazepam Intensol (liquid lorazepam) in the U.S. market?
Lorazepam Intensol is an oral liquid formulation of lorazepam (a benzodiazepine) marketed for acute anxiety and related indications. The product is dispensed in solution form (oral drops), which typically supports use in patients who cannot swallow tablets or prefer liquid dosing.
In the U.S., lorazepam is a mature, widely used generic drug category with long-standing supply and multiple manufacturers. The liquid format can have fewer direct substitutes than tablets, but it still competes with generic lorazepam strengths in tablet form and with other benzodiazepine liquids or equivalents depending on formulary rules.
Key market characteristics that shape price behavior:
- Generic-driven pricing: Lorazepam (the API) is long off-patent, with extensive generic manufacturing.
- Formulation substitution: Liquid lorazepam can be substituted by equivalent oral lorazepam (often tablets) when clinicians and formularies allow.
- Competitive contracting: Pharmacy benefit managers (PBMs) and group purchasing organizations (GPOs) typically drive net prices toward lowest-cost contracted products.
- Volume sensitivity: High-volume generics tend to see price stability or downward pressure, with temporary spikes tied to shortages or contract resets.
How does the competitive landscape typically affect pricing for Lorazepam Intensol?
Lorazepam Intensol competes on:
- Availability (product continuity and lot coverage)
- Contract position (preferred vs non-preferred on formularies)
- Dosage form (liquid vs tablet substitution)
- Package size and concentration (impacting per-mL and per-day acquisition cost)
In mature generic categories, pricing generally follows the contracting cycle:
- When a liquid formulation is preferred, the net price often tracks the lowest-cost contracted equivalent across manufacturers.
- When a product loses preferred status or faces inventory constraints, net acquisition cost can move sharply, then normalize after supply and contracting realign.
Is Lorazepam Intensol likely to face patent-driven price support?
There is no market exclusivity premium implied by the API for lorazepam itself, given the molecule’s maturity. Any pricing lift for a specific dosage form is typically tied to:
- formulation-specific regulatory exclusivity (if applicable)
- brand-to-generic and generic-to-generic substitution dynamics
- supply and contracting terms
For a generic-style product, economic exclusivity (contract position and supply) matters more than any molecule-level protection.
What drives net price vs list price for this product class?
For generic benzodiazepines in the U.S., observed acquisition prices for dispensers tend to be influenced by:
- PBM preferred tier placement (preferred, formulary, non-formulary)
- mandatory substitution rules by state and insurer
- wholesaler mix and warehouse buying patterns
- pharmacy service contract terms
- temporary supply tightness
Net price often moves more than list price. When shortages occur in the class, wholesalers may ration and pharmacies may see higher acquisition cost that later corrects.
Market sizing and demand signals: what matters for projections
A robust projection for a mature generic requires mapping demand to utilization and dispensing patterns, which in practice correlate to:
- baseline anxiety and insomnia prescribing trends
- benzodiazepine risk policy changes (which can reduce demand at the margin)
- payer management (prior authorization and step edits in some settings)
- substitution from tablets to liquids and vice versa
- institutional channel mix (retail vs long-term care)
Lorazepam is widely used, but benzodiazepine category scrutiny can lead to utilization changes over time. For a liquid formulation, demand can remain steadier than tablets if liquid-dosing is clinically entrenched for certain patient cohorts.
Price projection framework (generic liquid benzodiazepines)
A practical way to project price is to separate:
- Baseline contraction from generics competition and PBM contracting
- Short-term shocks from supply tightness, allocation, or manufacturing outages
- Contract resets where net pricing re-anchors to the lowest-cost supplier
For Lorazepam Intensol specifically, the most likely price path is:
- Near-term: stable to modestly down, unless there is supply disruption.
- Medium-term (12 to 36 months): continued pressure from competitive sourcing and contracting, typically with occasional step-ups on allocation events.
- Long-term (3 to 5 years): generally flat-to-down, with volatility limited to episodic supply and payer preference changes.
Projected U.S. pricing path (2026-2028)
Because exact realized net pricing for Lorazepam Intensol depends on payer contracts, channel mix, and package size, projections are presented as scenario-based ranges anchored to generic market behavior for mature benzodiazepines.
Scenario table: annual price movement (net acquisition)
| Time horizon | Base case (normal supply, stable contracting) | Downside case (more competition, preferred tier losses for this SKU) | Upside case (supply constraints or preference shift back to this SKU) |
|---|---|---|---|
| Next 12 months | -2% to +1% | -4% to -10% | +3% to +12% |
| 13-24 months | -2% to +2% | -3% to -8% | +2% to +10% |
| 25-36 months | -1% to +2% | -2% to -6% | +1% to +8% |
Interpretation: In a normal year, net acquisition for mature generics like lorazepam liquids tends to stay close to flat with mild downward drift. Material upside requires a supply event or payer preference movement; material downside requires accelerated competition and re-contracting.
What specific events could shift Lorazepam Intensol price up or down?
Upward price pressures
- Class-level shortages that reduce substitute availability, driving allocation and higher wholesaler pricing
- Manufacturing interruptions specific to liquid lorazepam supply
- Payer formulary dynamics that restore this specific SKU as preferred or reduce substitution for liquid dosing
Downward price pressures
- New low-cost generic entrants (or shifting production volumes among suppliers) that reset contracted rates
- Loss of preferred tier relative to tablet equivalents or other liquid sources
- Contract repricing after periodic PBM/GPO tenders
Channel and contracting: where price is most likely to move
- Retail pharmacies: typically see price changes tied to wholesaler cost and PBM reimbursement updates; volatility is modest unless supply is constrained.
- Institutional (e.g., hospitals, nursing facilities): tend to lock procurement via group contracts; sudden movements often occur only during contract renegotiations or emergency sourcing.
- Mail order: often tracks PBM preferred pricing; changes can be sudden when PBM formulary or tier rules update.
Practical pricing outlook by package format (logic for estimates)
Lorazepam Intensol pricing is commonly evaluated on a per-day dose and per-mL basis. For projections:
- Liquid pricing per mL is influenced by the concentration and bottle size.
- Any scenario implying “higher acquisition” usually shows up first as higher per-mL cost rather than per-dose, since dosage conversions are fixed.
A market-driven approach assumes:
- Per-mL price stability unless supply disruption or preferred status changes
- Per-dose purchasing remains demand-stable because dosing patterns are clinical
How to convert the scenario ranges into business projections
For budgeting, apply the projected annual change to your baseline net acquisition cost:
- Base case: use -1% to +2% per year as the planning band
- Downside: use -5% to -10% if your contracts move toward lower-cost alternatives
- Upside: use +5% to +10% in the event of supply constraints and non-preferred sourcing
Commercial implications
- For buyers (pharmacies, hospitals, PBMs): price risk is low under normal supply but not zero. Build contingency for episodic supply constraints that can force higher allocation costs.
- For manufacturers/marketers: revenue sustainability depends on contract retention and continuous supply for liquid inventory coverage.
- For investors: upside is typically a supply and contracting story, not a patent-driven monopoly story.
Key Takeaways
- Lorazepam Intensol is a mature, generic-driven benzodiazepine product where net price is primarily determined by contracting and substitution, not patent exclusivity.
- Under normal supply, net acquisition price is likely flat to modestly down (planning band -2% to +2% annually).
- Meaningful price increases usually require supply constraints or preference shifts; meaningful decreases typically follow contract resets and intensified competition.
- The most actionable projection method is scenario-based annual change applied to your current net acquisition baseline rather than list price.
FAQs
-
Will Lorazepam Intensol get sustained price increases in the absence of shortages?
No. In mature generic markets, absent supply disruption or a preference shift, pricing trends toward stability or mild decline. -
What matters more for Lorazepam Intensol pricing, API exclusivity or contract position?
Contract position and supply determine realized net pricing; API exclusivity does not drive sustained price premiums in this category. -
Does liquid lorazepam face substitution risk from tablets?
Yes. Tablets are the most common substitution pathway when formularies allow equivalent lorazepam dosing. -
What is the main driver of upside pricing scenarios?
Short supply or allocation in liquid lorazepam, plus payer or wholesaler sourcing dynamics that reduce substitutes. -
How should buyers incorporate price risk into budgeting?
Use a base-case near-flat band (-2% to +2% annually) and add a contingency for supply constraints (+5% to +10%).
References
[1] APA. American Psychiatric Association. Benzodiazepines: clinical and prescribing information. https://www.psychiatry.org/ (accessed 2026-04-25)
[2] FDA. Drug Shortages program (search for lorazepam and benzodiazepines). https://www.accessdata.fda.gov/scripts/drugshortages/default.cfm (accessed 2026-04-25)
[3] FDA. Orange Book search for lorazepam products. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (accessed 2026-04-25)
More… ↓
