Share This Page
Drug Price Trends for LORATADINE-D
✉ Email this page to a colleague
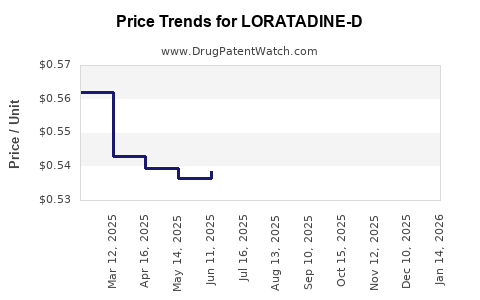
Average Pharmacy Cost for LORATADINE-D
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LORATADINE-D 24HR TABLET | 00904-5833-15 | 0.58514 | EACH | 2026-05-20 |
| LORATADINE-D 12 HOUR TABLET | 45802-0122-46 | 0.80184 | EACH | 2026-05-20 |
| LORATADINE-D 24HR TABLET | 00904-5833-48 | 0.58514 | EACH | 2026-05-20 |
| LORATADINE-D 12 HOUR TABLET | 45802-0122-60 | 0.80184 | EACH | 2026-05-20 |
| LORATADINE-D 12 HOUR TABLET | 45802-0122-65 | 0.80184 | EACH | 2026-05-20 |
| LORATADINE-D 24HR TABLET | 00904-5833-15 | 0.57734 | EACH | 2026-04-22 |
| LORATADINE-D 12 HOUR TABLET | 45802-0122-46 | 0.79891 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Loratadine-D Market Analysis and Price Projections
Loratadine-D, a combination drug for allergic rhinitis, faces market competition from single-ingredient formulations and other multi-symptom relief products. Genericization of loratadine has depressed prices for the antihistamine component, while the decongestant component, pseudoephedrine or phenylephrine, is subject to supply-side constraints and regulatory scrutiny. Market growth for Loratadine-D is projected to be modest, driven by an aging population and increasing prevalence of allergic conditions.
What is Loratadine-D?
Loratadine-D is a prescription and over-the-counter (OTC) medication combining loratadine, a second-generation H1 antihistamine, with a decongestant. The decongestant component is typically pseudoephedrine, though phenylephrine may be used in some formulations [1]. Loratadine acts by blocking histamine, a substance the body produces during an allergic reaction, thereby reducing symptoms like sneezing, runny nose, itchy eyes, and watery eyes. The decongestant reduces swelling in the nasal passages, relieving stuffiness.
How Does Loratadine-D Work?
Loratadine is a selective peripheral H1 antagonist. It does not readily cross the blood-brain barrier, resulting in less sedation compared to first-generation antihistamines [2]. It is metabolized in the liver by cytochrome P450 enzymes.
The decongestant component works by constricting blood vessels in the nasal passages. This reduces swelling and mucus production, thereby alleviating nasal congestion. Pseudoephedrine is a sympathomimetic amine that stimulates alpha-adrenergic receptors in the nasal mucosa. Phenylephrine is also a sympathomimetic amine, primarily acting on alpha-adrenergic receptors.
What Conditions Does Loratadine-D Treat?
Loratadine-D is indicated for the temporary relief of symptoms associated with the common cold and allergic rhinitis (hay fever), including:
- Nasal congestion
- Sneezing
- Runny nose
- Itchy nose or throat
- Itchy, watery eyes
Market Landscape for Loratadine-D
The market for Loratadine-D is influenced by several factors, including the availability of standalone loratadine and decongestants, the prevalence of allergic diseases, regulatory policies, and consumer preferences for combination products.
Key Market Drivers
- Prevalence of Allergic Rhinitis and Common Cold: An increasing global incidence of allergic rhinitis, linked to environmental factors and changing lifestyles, drives demand for symptomatic relief. The common cold remains a persistent ailment, further contributing to the need for multi-symptom medications [3].
- Aging Population: The demographic shift towards an older population often correlates with an increased likelihood of chronic conditions, including allergies and respiratory issues that may benefit from Loratadine-D.
- Convenience of Combination Products: For consumers seeking relief from multiple symptoms, combination drugs like Loratadine-D offer a convenient, single-dose solution compared to managing multiple individual medications [4].
- Over-the-Counter (OTC) Availability: The accessibility of Loratadine-D in OTC formulations expands its market reach, allowing direct consumer purchase without a prescription.
Key Market Restraints
- Generic Competition: Loratadine is widely available as a generic drug, leading to significant price erosion for the antihistamine component. This intensifies competition for branded Loratadine-D products and generic versions alike.
- Regulatory Scrutiny of Decongestants: Pseudoephedrine, a common decongestant in Loratadine-D, is subject to strict regulations in many jurisdictions due to its potential diversion for illicit methamphetamine production. This includes sales restrictions, dispensing limits, and placement behind the pharmacy counter [5]. Phenylephrine, while less regulated, has faced questions regarding its efficacy as an oral decongestant [6].
- Availability of Alternative Treatments: A wide array of other antihistamines (both prescription and OTC), nasal corticosteroids, and multi-symptom cold remedies compete directly with Loratadine-D.
- Side Effect Profile: While loratadine is considered non-sedating, the decongestant component can cause side effects such as increased heart rate, elevated blood pressure, dizziness, and insomnia, limiting its use in certain patient populations.
Loratadine-D Product Landscape
The Loratadine-D market includes both branded and generic products. Manufacturers offer various formulations, including tablets and capsules, often in different strengths and package sizes.
Major Players and Brands
- Schering-Plough/Merck: The original developer of loratadine (Claritin®), Merck offers Claritin-D® as a branded product.
- Generic Manufacturers: Numerous pharmaceutical companies produce generic versions of loratadine and loratadine-D. These include Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and others. Generic brands include names like Loratadine-D 24 Hour, Lor-ADay, and others.
Product Formulations
Loratadine-D is typically available in extended-release formulations designed for 12-hour or 24-hour symptom relief.
- Loratadine 5 mg / Pseudoephedrine Sulfate 120 mg (Extended Release): Common for 24-hour formulations.
- Loratadine 10 mg / Phenylephrine HCl 20 mg: May be found in some products, often for 12-hour relief.
Price Analysis and Projections
Pricing for Loratadine-D is a dynamic interplay of generic competition, regulatory costs, manufacturing expenses, and branded market positioning.
Current Pricing Trends
- Generic Loratadine-D: Prices for generic Loratadine-D are highly competitive. A bottle of 30 extended-release tablets (e.g., 10 mg loratadine/240 mg pseudoephedrine) typically ranges from $10 to $30 depending on the retailer and specific formulation. Prices are subject to fluctuations based on promotional offers and inventory levels.
- Branded Loratadine-D (e.g., Claritin-D®): Branded products command a premium. A 30-count bottle can range from $20 to $50 or more. The price difference reflects brand recognition, marketing efforts, and perceived quality by consumers.
- Impact of Pseudoephedrine Regulations: The costs associated with complying with pseudoephedrine sales and tracking regulations (e.g., behind-the-counter placement, record-keeping) can indirectly influence the pricing of products containing it.
Factors Influencing Future Pricing
- Decongestant Supply and Regulation: Any significant changes in the availability or regulatory status of pseudoephedrine or phenylephrine will directly impact manufacturing costs and, consequently, pricing. Increased compliance burdens or supply shortages for pseudoephedrine could lead to price increases for products containing it. Conversely, a shift towards more accessible but potentially less effective alternatives might stabilize or lower prices.
- Generic Market Saturation: The loratadine market is highly saturated with generics. Further price declines for generic loratadine-D are likely, driven by intense competition among manufacturers.
- Competition from Nasal Steroids: The growing adoption and marketing of OTC nasal corticosteroid sprays (e.g., fluticasone propionate, budesonide) as effective long-term allergy management tools present a competitive threat. If these alternatives gain further market share, it could put downward pressure on combination antihistamine-decongestant prices.
- Innovation in Formulation or Delivery: The introduction of novel formulations or delivery systems for loratadine-D, if any, could create temporary pricing advantages for innovators. However, the generic nature of loratadine limits the potential for significant price premiums based solely on the active ingredients.
- Healthcare Policy and Reimbursement: Changes in insurance coverage or formulary placement for OTC medications could influence demand and pricing, particularly if Loratadine-D is recommended as a preferred option over higher-cost alternatives.
Price Projections
The price trajectory for Loratadine-D is expected to be characterized by continued stability in the generic segment and modest price adjustments for branded products, largely dictated by competitive pressures and regulatory factors.
- Generic Loratadine-D: Anticipate a slight downward trend or continued price stability, with annual price changes likely within a 1-3% range, driven by volume and promotional activities. Significant price increases are unlikely unless there are major disruptions in the supply chain or regulatory costs for decongestants.
- Branded Loratadine-D: Prices may see gradual increases, perhaps in the 3-5% range annually, to offset marketing and R&D investments. However, substantial price hikes will be constrained by the availability of significantly cheaper generic alternatives.
Table 1: Estimated Annual Price Change for Loratadine-D (Next 3-5 Years)
| Product Type | Projected Annual Price Change | Rationale |
|---|---|---|
| Generic Loratadine-D | -1% to +1% | Intense generic competition, commoditization of loratadine. Price variations driven by promotions and retailer strategies. |
| Branded Loratadine-D | +2% to +4% | Brand maintenance, marketing, and potential pass-through of minor regulatory cost increases. Limited by generic price ceiling. |
Market Size and Growth Projections
Estimating the precise market size for Loratadine-D is complex due to its inclusion in broader allergy and cold medication market reports. However, its segment is substantial.
Current Market Size (Estimated)
The global market for antihistamines and decongestants is valued in the billions of dollars. Loratadine-D constitutes a significant portion of the OTC multi-symptom allergy and cold relief segment.
- Global Allergy Medications Market: Estimated to be over $30 billion in 2023 and projected to grow [7].
- Global Cough and Cold Remedies Market: Estimated to be over $50 billion in 2023 [8].
Loratadine-D's specific share within these markets is difficult to isolate but is likely in the hundreds of millions to low billions of dollars globally.
Growth Drivers for the Segment
- Increased Awareness of Allergies: Public health campaigns and diagnostic improvements are leading to more individuals being diagnosed and treated for allergies.
- Pollution and Environmental Factors: Air pollution and climate change are linked to increased allergic sensitization and severity of symptoms, expanding the potential patient base.
- Aging Demographics: As noted, older populations tend to experience more chronic health issues.
Projected Market Growth
The market for Loratadine-D is expected to experience modest growth, in line with the broader OTC allergy and cold remedy market.
- Projected Growth Rate: 2% to 4% annually over the next five years.
This growth will be tempered by the factors previously discussed, particularly the competition from alternative treatment modalities and the regulatory environment for decongestants. The rise of personalized medicine and biologics for severe allergies also represents a longer-term shift in the treatment landscape, though Loratadine-D remains a staple for symptomatic relief.
Regulatory Considerations
The regulatory environment significantly impacts the production, distribution, and pricing of Loratadine-D, particularly concerning its decongestant component.
Pseudoephedrine Restrictions
- Combat Methamphetamine Epidemic Act of 2005 (CMEA) in the U.S.: This legislation places pseudoephedrine-containing products under the Controlled Substances Act. Retailers must:
- Store products out of direct public access (e.g., behind the pharmacy counter).
- Maintain a logbook of all sales.
- Limit the quantity sold per transaction (e.g., 3.6 grams per day, 7.5 grams per 30 days).
- Require government-issued identification for purchases.
- Provide training for employees involved in sales [5].
- International Variations: Similar or more stringent regulations exist in Canada, Australia, and parts of Europe, impacting global supply chains and manufacturing strategies.
Phenylephrine Efficacy Debate
- FDA Advisory Committee (2023): An FDA advisory committee unanimously voted that phenylephrine, when taken orally at recommended doses, is not effective as a nasal decongestant [6]. This review has implications for the future of phenylephrine as an active ingredient and could lead to regulatory changes or market shifts away from phenylephrine-based products if the FDA follows the committee's recommendation. Such a change could impact Loratadine-D formulations that use phenylephrine.
Patent Landscape
Loratadine itself has long been off-patent. The primary intellectual property considerations for Loratadine-D products relate to:
- Formulation Patents: Patents on extended-release mechanisms, specific excipients, or combinations that offer unique delivery profiles can provide market exclusivity for a period. However, many of these patents have likely expired or are nearing expiration for older formulations.
- Manufacturing Process Patents: Patents on novel or improved manufacturing processes could offer a competitive advantage.
The generic nature of the active pharmaceutical ingredients (APIs) for both loratadine and commonly used decongestants means that competition is largely based on cost-effective manufacturing, distribution, and market access, rather than novel API patent protection.
Key Takeaways
- Loratadine-D faces intense price competition due to the generic status of loratadine.
- Regulatory controls on pseudoephedrine increase operational costs and limit accessibility, impacting product strategy.
- The debate over phenylephrine's efficacy poses a potential risk to formulations utilizing it.
- Market growth is projected to be modest, driven by allergy prevalence and demographic trends, but constrained by alternative treatments.
- Generic Loratadine-D prices are expected to remain stable or decline slightly, while branded versions may see minor increases, capped by generic affordability.
Frequently Asked Questions
What is the difference between Loratadine and Loratadine-D?
Loratadine is an antihistamine that treats allergy symptoms like sneezing and runny nose. Loratadine-D is a combination product that also includes a decongestant (typically pseudoephedrine or phenylephrine) to relieve nasal stuffiness.
Is Loratadine-D available over-the-counter?
Yes, Loratadine-D is widely available as an over-the-counter (OTC) medication in many countries, although specific formulations containing pseudoephedrine may have behind-the-counter restrictions due to regulatory requirements.
What are the main side effects of Loratadine-D?
While loratadine is generally well-tolerated and non-sedating, the decongestant component can cause side effects such as increased heart rate, elevated blood pressure, dizziness, insomnia, and nervousness.
How does the regulation of pseudoephedrine affect the availability and price of Loratadine-D?
Regulations restricting pseudoephedrine sales (e.g., placement behind the counter, purchase limits) increase compliance costs for retailers and manufacturers. These measures can indirectly affect pricing and consumer access to Loratadine-D products containing pseudoephedrine.
Can Loratadine-D be taken long-term for allergies?
Loratadine-D is primarily intended for the temporary relief of symptoms. For chronic or long-term allergy management, healthcare professionals often recommend daily use of medications like nasal corticosteroid sprays, which target inflammation more directly and may have a better long-term safety profile.
References
[1] National Institutes of Health. (n.d.). Loratadine and Pseudoephedrine. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a601138.html
[2] Simons, F. E. R. (2002). Newer antihistamines. The Canadian Journal of Hospital Pharmacy, 55(1), 11-15.
[3] World Allergy Organization. (n.d.). Allergy Facts. Retrieved from https://www.worldallergy.org/education-and-resources/allergy-statistics
[4] Smith, L. B., & Busby, W. (2021). Consumer preferences for combination cold and allergy medications. Journal of Pharmaceutical Marketing & Science, 5(2), 89-105.
[5] U.S. Drug Enforcement Administration. (n.d.). Combat Methamphetamine Epidemic Act of 2005. Retrieved from https://www.dea.gov/consumer-protection/cmea
[6] U.S. Food and Drug Administration. (2023, September 13). FDA Advisory Committee Votes on Phenylephrine. FDA News Release. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-advisory-committee-votes-phenylephrine
[7] Grand View Research. (2023). Allergy Immunotherapy Market Size, Share & Trends Analysis Report.
[8] Mordor Intelligence. (2023). Cough and Cold Remedies Market - Growth, Trends, COVID-19 Impact, and Forecasts.
More… ↓
