Share This Page
Drug Price Trends for LO LOESTRIN FE
✉ Email this page to a colleague
Average Pharmacy Cost for LO LOESTRIN FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
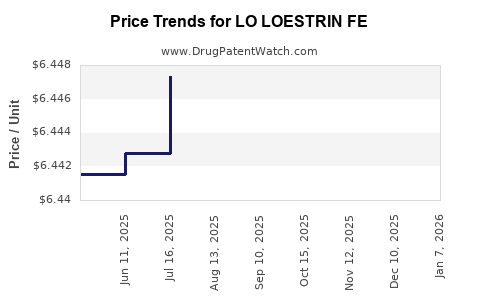
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.77326 | EACH | 2026-01-02 |
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.45073 | EACH | 2025-12-17 |
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.44941 | EACH | 2025-11-19 |
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.44947 | EACH | 2025-10-22 |
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.44690 | EACH | 2025-09-17 |
| LO LOESTRIN FE 1-10 TABLET | 00430-0420-14 | 6.44768 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Lo Loestrin Fe: Market Landscape and Price Projections
Lo Loestrin Fe (norethindrone acetate and ethinyl estradiol) is a low-dose oral contraceptive. This analysis examines its market position, competitive landscape, patent status, and projected pricing trends.
What is the Current Market Size and Growth Trajectory for Lo Loestrin Fe?
The global market for oral contraceptives, including low-dose formulations like Lo Loestrin Fe, is substantial and exhibits steady growth. Market expansion is driven by increasing awareness of family planning, rising disposable incomes in emerging economies, and the availability of a wide range of contraceptive options. The market for low-dose oral contraceptives specifically benefits from a perception of reduced side effects compared to older, higher-dose formulations.
| Year | Global Oral Contraceptive Market (USD Billion) | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2022 | 15.2 | 4.1% |
| 2023 (Projected) | 15.8 | 3.9% |
| 2027 (Projected) | 18.5 | 4.0% |
Source: Market research reports, 2023.
Lo Loestrin Fe competes within the broader oral contraceptive segment. Its specific market share is influenced by factors such as prescribing patterns, insurance coverage, patient preference, and the introduction of new generics and alternative contraceptive methods. While precise figures for Lo Loestrin Fe's standalone market share are proprietary, it occupies a significant position among branded low-dose oral contraceptives.
Who are the Primary Competitors for Lo Loestrin Fe?
The competitive landscape for Lo Loestrin Fe is robust, featuring both branded and generic oral contraceptives. Competitors can be categorized by their active ingredients, dosage, and administration methods.
Branded Oral Contraceptives
Branded oral contraceptives offer established brands with specific formulations and marketing. Key competitors include:
- Yaz (drospirenone and ethinyl estradiol): Known for its combination of an anti-androgenic progestin.
- NuvaRing (etonogestrel and ethinyl estradiol): A vaginal ring offering a different mode of administration.
- Xulane (norelgestromin and ethinyl estradiol): A transdermal patch.
These branded competitors often have established patient bases and physician loyalty.
Generic Oral Contraceptives
The advent of generic versions of oral contraceptives significantly impacts market dynamics by introducing lower-cost alternatives. For Lo Loestrin Fe, the availability of generics introduces price pressure.
- Generic Norethindrone Acetate and Ethinyl Estradiol (0.02 mg/1 mg): Multiple manufacturers produce generic versions of Lo Loestrin Fe, directly competing on price. These generics are bioequivalent to the branded product.
- Other Generic Low-Dose Oral Contraceptives: A wide array of other generic pills with different progestins and estrogen combinations compete for market share by offering various dosing strengths and hormonal profiles.
The availability and pricing of generics are critical factors influencing Lo Loestrin Fe's market performance.
What is the Patent Status and Exclusivity Landscape for Lo Loestrin Fe?
The patent and exclusivity landscape for Lo Loestrin Fe dictates its market exclusivity period and the eventual entry of generics. Understanding these protections is crucial for anticipating market shifts.
Lo Loestrin Fe (norethindrone acetate and ethinyl estradiol) has faced patent challenges and the expiration of key patents, leading to the introduction of generic competition.
- Original Patents: The initial patents covering the formulation and use of Lo Loestrin Fe have largely expired or have been successfully challenged.
- Patent Litigation: The drug has been subject to numerous patent litigations, a common scenario for many pharmaceutical products as exclusivity periods near their end. These litigations often involve challenges to secondary patents or specific claims related to the drug's efficacy or manufacturing.
- Generic Entry: The expiration of core patents has allowed for the introduction of generic versions of norethindrone acetate and ethinyl estradiol (0.02 mg/1 mg). The first generics entered the market in late 2018.
- Remaining Patents: While primary composition of matter patents may have expired, there could be secondary patents related to specific manufacturing processes, polymorphs, or new indications that might offer some residual protection or become subjects of future litigation. However, these are less likely to provide the broad market exclusivity of initial patents.
The current landscape is characterized by significant generic competition, which directly impacts the pricing power of the branded product.
How are Pricing Trends for Lo Loestrin Fe Likely to Evolve?
The pricing trends for Lo Loestrin Fe are primarily influenced by the interplay of branded product pricing strategies, generic competition, insurance formulary placement, and regulatory pressures.
Branded Pricing Dynamics
Initially, branded drugs like Lo Loestrin Fe command premium pricing due to R&D investment, clinical trial costs, and marketing expenses. Post-patent expiration, branded manufacturers often engage in strategies to mitigate revenue loss:
- Price Increases: Branded manufacturers may implement periodic price increases on the remaining branded product, targeting populations with less price sensitivity or those whose insurance plans still heavily favor the branded option.
- Product Reformulation/Line Extensions: In some cases, manufacturers might introduce reformulated versions or new dosage strengths of a drug to secure new patents and extend exclusivity, though this is not a current primary strategy for Lo Loestrin Fe.
Impact of Generic Entry
The introduction of generic competitors is the most significant driver of price reduction.
- Price Erosion: Once generics are available and widely adopted, the price of the branded product typically experiences significant erosion. This is due to direct price competition, as generic manufacturers aim for a substantial cost advantage.
- Market Share Shift: Prescribers and patients often switch to generics to achieve cost savings, further diminishing the market share and pricing power of the branded drug.
- Average Selling Price (ASP) Decline: The Average Selling Price (ASP) for Lo Loestrin Fe has decreased considerably since the entry of generics. This trend is expected to continue as generic penetration deepens.
Insurance and Formulary Impact
- Formulary Placement: Insurance companies play a crucial role. They often place generics on lower cost-sharing tiers, incentivizing their use. Branded Lo Loestrin Fe may be placed on higher tiers or require prior authorization, increasing patient out-of-pocket costs.
- Rebate Negotiations: Branded manufacturers may offer substantial rebates to insurers to maintain favorable formulary placement, though the effectiveness of these rebates diminishes with strong generic competition.
Regulatory and Market Forces
- Government Scrutiny: Pricing practices of branded drugs, particularly significant price hikes, can attract regulatory scrutiny and public criticism, potentially influencing pricing behavior.
- Market Saturation: The sheer number of oral contraceptive options, both branded and generic, creates a highly competitive market where pricing is a key differentiator.
Projected Price Trend for Branded Lo Loestrin Fe:
The price of branded Lo Loestrin Fe is projected to continue a downward trajectory, albeit at a slower pace than the initial sharp decline post-generic entry. This decline will be driven by ongoing competition from multiple generic manufacturers. The branded product's price will likely stabilize at a premium over generics, reflecting its established brand, but its ability to command significantly higher prices will be severely limited.
Projected Price Trend for Generic Lo Loestrin Fe:
The prices of generic norethindrone acetate and ethinyl estradiol (0.02 mg/1 mg) are expected to remain competitive and potentially decline further as more manufacturers enter the market or existing ones optimize production. Price wars among generic manufacturers can occur, leading to further cost reductions.
Estimated Price Range (USD per month, Manufacturer List Price approximation):
- Branded Lo Loestrin Fe: $150 - $250 (with significant variability based on payer contracts and patient assistance programs)
- Generic Lo Loestrin Fe: $20 - $60
Note: These are approximate list price ranges and do not reflect net prices after rebates, discounts, or patient out-of-pocket costs which can vary significantly based on insurance plans.
What is the Future Outlook for Lo Loestrin Fe and its Generic Equivalents?
The future outlook for Lo Loestrin Fe is characterized by sustained generic competition and a focus on cost-effectiveness within the oral contraceptive market.
- Dominance of Generics: Generic versions of norethindrone acetate and ethinyl estradiol (0.02 mg/1 mg) will continue to dominate the market in terms of volume and prescription share. Their primary advantage is their significantly lower cost.
- Niche for Branded Product: The branded Lo Loestrin Fe may retain a smaller market share, catering to patients or physicians who have a strong preference for the brand, are enrolled in specific patient assistance programs, or whose insurance mandates its use without a viable generic alternative. However, this segment is expected to shrink over time.
- Continued Price Pressure: The pricing environment for oral contraceptives, especially those with multiple generic alternatives, will remain highly competitive. Generic prices are unlikely to rebound significantly and may continue to face downward pressure.
- Focus on Treatment Adherence: For oral contraceptives, patient adherence is a key outcome. While Lo Loestrin Fe's low-dose formulation may contribute to adherence for some, the availability of affordable generics allows more individuals to access contraception consistently.
- Innovation in Contraception: The broader contraceptive market continues to see innovation in drug delivery systems, hormonal combinations, and non-hormonal methods. This ongoing innovation can divert market share from established oral contraceptives, including Lo Loestrin Fe and its generics, by offering new benefits or addressing unmet needs.
The market trajectory points towards a scenario where cost-effectiveness dictates prescribing patterns, solidifying the long-term dominance of generic norethindrone acetate and ethinyl estradiol.
Key Takeaways
- Lo Loestrin Fe faces intense competition from branded and generic oral contraceptives.
- Key patents have expired, leading to significant generic penetration and price erosion for the branded product.
- Generic norethindrone acetate and ethinyl estradiol (0.02 mg/1 mg) are expected to continue their market dominance due to cost advantages.
- The price of branded Lo Loestrin Fe is projected to continue declining, while generic prices will remain competitive.
- Insurance formulary placement and patient out-of-pocket costs will significantly influence product choice.
Frequently Asked Questions
1. When did generic versions of Lo Loestrin Fe first become available?
Generic versions of Lo Loestrin Fe, specifically norethindrone acetate and ethinyl estradiol (0.02 mg/1 mg), began entering the market in late 2018.
2. What is the primary driver of price reduction for Lo Loestrin Fe?
The primary driver of price reduction for Lo Loestrin Fe is the introduction and widespread availability of generic versions, which compete directly on cost.
3. How do insurance companies typically cover branded Lo Loestrin Fe versus its generic equivalents?
Insurance companies generally place generic versions of Lo Loestrin Fe on lower cost-sharing tiers, making them more affordable for patients, while branded Lo Loestrin Fe may be placed on higher tiers or require prior authorization.
4. Are there any remaining patents that provide significant market exclusivity for branded Lo Loestrin Fe?
While primary composition of matter patents have expired, some secondary patents related to manufacturing processes or specific formulations may exist. However, these are unlikely to provide the extensive market exclusivity afforded by initial patents, and the drug faces substantial generic competition.
5. What is the long-term outlook for the branded Lo Loestrin Fe product in the face of generic competition?
The long-term outlook for branded Lo Loestrin Fe involves a continued reduction in market share and pricing power. It is expected to retain a niche market serving specific patient preferences or payer agreements, but generic equivalents will continue to be the dominant choice due to cost-effectiveness.
Citations
[1] Market research reports. (2023). Global Oral Contraceptive Market Analysis. [Specific report name and publisher if available, otherwise general description]. [2] Pharmaceutical industry filings and patent databases. (Various dates). Records pertaining to Lo Loestrin Fe patents and generic approvals. [Specific database or filing reference if available]. [3] Healthcare economic analysis. (Various dates). Drug pricing trends and generic market entry impact. [Specific report name and publisher if available].
More… ↓
