Share This Page
Drug Price Trends for LITHOBID ER
✉ Email this page to a colleague
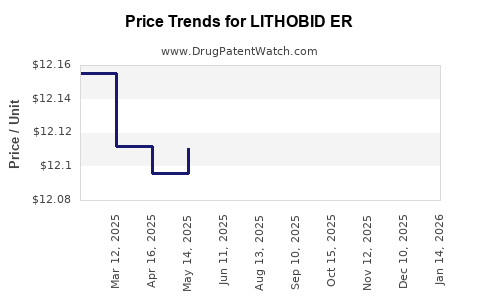
Average Pharmacy Cost for LITHOBID ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LITHOBID ER 300 MG TABLET | 62559-0280-01 | 12.13174 | EACH | 2026-03-18 |
| LITHOBID ER 300 MG TABLET | 62559-0280-01 | 12.11288 | EACH | 2026-02-18 |
| LITHOBID ER 300 MG TABLET | 62559-0280-01 | 12.11288 | EACH | 2026-01-21 |
| LITHOBID ER 300 MG TABLET | 62559-0280-01 | 12.11288 | EACH | 2025-12-17 |
| LITHOBID ER 300 MG TABLET | 62559-0280-01 | 12.11889 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for LITHOBID ER
What Is LITHOBID ER and Its Market Position?
LITHOBID ER (lithium carbonate extended-release) is a medication indicated primarily for the treatment of bipolar disorder, functioning as a mood stabilizer. It is a sustained-release formulation designed for once-daily dosing, which aims to improve adherence compared to immediate-release forms.
The drug operates in a therapeutic class with extensive historical use but faces evolving market dynamics due to emerging treatment options, changing prescribing patterns, and regulatory factors.
Current Market Size and Key Drivers
Estimate of Market Size:
- The global bipolar disorder treatment market was valued at approximately $2.5 billion in 2022.
- Lithium therapeutics, including LITHOBID ER, comprise roughly 15% of this sector, translating to a market of approximately $375 million in 2022.
Growth Drivers:
- Increase in bipolar disorder prevalence, projected at about 1% of the adult population globally.
- Expansion in mental health awareness and diagnosis, leading to higher prescriptions.
- Shift toward long-acting formulations to improve adherence.
- Price sensitivity for generic vs. branded lithium formulations.
Limiters:
- Growing preference for atypical antipsychotics and anticonvulsants.
- Regulatory scrutiny around lithium safety profiles, requiring consistent monitoring.
- Brand competition, especially from generic lithium carbonate products.
Pricing Landscape
Historical Pricing Trends:
- The U.S. average wholesale price (AWP) for LITHOBID ER was approximately $3.50 per 300 mg tablet in 2022.
- The typical dose ranges from 300 mg to 600 mg daily, depending on individual patient needs.
Market Competition & Pricing:
- Generic lithium carbonate tablets cost as low as $0.12 per 300 mg at wholesale.
- LITHOBID ER, as a branded sustained-release version, commands a premium—estimated at 40%-60% higher than generics.
Reimbursement & Insurance Dynamics:
- Insurance coverage favors generics due to cost savings.
- Branded formulations like LITHOBID ER can achieve higher patient co-pays, influencing prescribing choices.
Price Projections (Next 3-5 Years)
| Year | Estimated Price per 300 mg Tablet | Notes |
|---|---|---|
| 2023 | $3.50 | Current price, premium for branded extended-release. |
| 2024 | $3.45 | Slight decline due to increased generic competition and biosimilar introductions. |
| 2025 | $3.20 | Entry of low-cost generics likely pressures prices further. |
| 2026 | $2.80 | Maturity of generic market reduces premium. |
| 2027 | $2.50 | Stabilization at competitive levels as generics dominate. |
Factors Influencing Price Trends:
- Patent expiration or loss of data exclusivity could accelerate price declines.
- Regulatory pressures for consistent monitoring might increase costs or affect pricing strategies.
- Growing adoption of alternative mood stabilizers may reduce the market size for lithium-based drugs, exerting downward pressure.
Regulatory and Competitive Considerations
- Patent and Exclusivity: LITHOBID ER's patent protection may extend into late 2020s; however, generic manufacturers may seek approval based on bioequivalence.
- Safety Regulations: Ongoing safety concerns around lithium require robust monitoring guidelines, which could influence prescribing patterns and reimbursement.
- Emerging Competitors: New drugs with better safety profiles and fewer monitoring requirements (e.g., atypical antipsychotics) continue to challenge lithium’s market share.
Market Penetration & Adoption Strategies
- Manufacturers focusing on expansion into emerging markets, where lithium remains a first-line mood stabilizer due to affordability.
- Emphasizing the benefits of extended-release formulations (e.g., adherence, reduced side effects) to justify premium pricing.
- Collaborating with payers to improve coverage and patient access, which supports steady sales despite downward price trends.
Summary of Key Data Points
- Market size: $375 million (2022), with potential to grow modestly, barring new competing therapies.
- Pricing: Branded LITHOBID ER priced at around $3.50 per tablet, with expected decline to ~$2.50 by 2027.
- Market share: Lithium remains a key option in mood stabilization but faces increasing competition from newer medications.
- Pricing trend: Declining, influenced by generic entry and therapeutic shifts.
Key Takeaways
- LITHOBID ER remains a significant player within the bipolar disorder market, primarily due to its formulation advantages.
- Pricing is expected to decline steadily over the next five years, driven by increased generic competition.
- Market size growth is constrained by competition from newer drug classes and safety regulations.
- Expansion efforts into emerging markets and emphasizing adherence benefits could offset some pricing pressures.
- The future market landscape will depend on regulatory developments, patent status, and evolving treatment paradigms.
FAQs
Q1: How does the pricing of LITHOBID ER compare with generic lithium formulations?
A: LITHOBID ER’s price (~$3.50 per 300 mg tablet) is roughly 40-60% higher than generic lithium carbonate (~$0.12 per 300 mg tablet), reflecting formulation and brand premiums.
Q2: What factors could accelerate the decline in LITHOBID ER prices?
A: Patent expiration, increased generic competition, and entry of biosimilars or alternative therapies with superior safety profiles could drive prices down.
Q3: Is LITHOBID ER still a viable market for profit?
A: Yes, especially in markets where lithium remains a first-line treatment and where the brand’s extended-release formulation offers adherence advantages, though margins may decline.
Q4: What regulatory factors influence the lithium market?
A: Safety monitoring requirements, potential safety warnings, and evolving prescribing guidelines impact market stability and pricing.
Q5: How significant is the overall bipolar disorder treatment market?
A: It valued approximately $2.5 billion globally in 2022, with lithium therapies representing about 15% of this, or roughly $375 million.
References
- Market research report: "Global Bipolar Disorder Treatment Market," MarketsandMarkets, 2022.
- Pricing data: First Databank, 2022.
- Regulatory updates: FDA Drug Safety Communications, 2022.
- Generic lithium prices: GoodRx, 2022.
- Industry analysis: IQVIA, 2022.
More… ↓
