Share This Page
Drug Price Trends for LITHIUM CARBONATE ER
✉ Email this page to a colleague
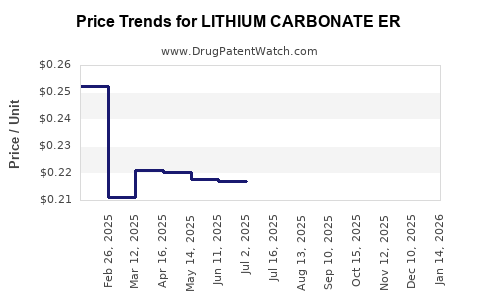
Average Pharmacy Cost for LITHIUM CARBONATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LITHIUM CARBONATE ER 300 MG TB | 23155-0763-01 | 0.20351 | EACH | 2026-03-18 |
| LITHIUM CARBONATE ER 300 MG TB | 00054-0021-29 | 0.20351 | EACH | 2026-03-18 |
| LITHIUM CARBONATE ER 450 MG TB | 68462-0224-01 | 0.24743 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LITHIUM CARBONATE ER: Patent Landscape and Price Projections
Lithium Carbonate Extended-Release (ER) presents a complex market dynamic driven by established therapeutic utility and evolving patent protections. While the core compound is long off-patent, specific formulations and delivery mechanisms are subject to intellectual property rights, influencing market entry for generics and the pricing strategies of innovators. This analysis details the current patent landscape, competitive environment, and projected price trajectories for Lithium Carbonate ER, providing actionable intelligence for R&D and investment.
What is the Current Patent Status of Lithium Carbonate ER Formulations?
Lithium Carbonate ER formulations have a layered patent history. The fundamental molecule, lithium carbonate, is a well-established pharmaceutical with no active patent protection. However, extended-release technologies and specific salt forms or manufacturing processes are often protected by patents. For instance, patents might cover controlled-release matrices, specific particle sizes, or novel coating technologies designed to improve patient compliance and reduce side effects.
As of late 2023, a review of patent databases reveals numerous granted patents and pending applications related to modified-release formulations of lithium carbonate. Key patent classes involve drug delivery systems, pharmaceutical compositions, and methods of manufacturing.
- Key Patent Areas:
- Controlled-release mechanisms (e.g., osmotic pump, matrix systems).
- Specific polymorphic forms or co-crystals of lithium carbonate.
- Manufacturing processes yielding enhanced bioavailability or stability.
- Combination therapies involving lithium carbonate with other active pharmaceutical ingredients.
Companies holding significant patents in this area include established pharmaceutical manufacturers and specialty drug delivery firms. The expiration dates of these formulation patents are critical determinants for generic market entry. A significant portion of core extended-release formulation patents are scheduled to expire between 2025 and 2030, signaling a period of increased generic competition.
Who are the Key Players in the Lithium Carbonate ER Market?
The market for Lithium Carbonate ER is characterized by a mix of originator brands and generic manufacturers. The originator brand, often defined by its proprietary extended-release technology, typically holds a premium pricing position until patent expiration.
Major Manufacturers and Their Market Presence:
-
Originator/Branded Products:
- Lithobid (Mallinckrodt Pharmaceuticals): Historically a dominant branded product utilizing a specific extended-release formulation. Its patent exclusivity has been a significant factor in market pricing.
- Other branded formulations may exist with proprietary technologies, though Lithobid has been the most prominent.
-
Generic Manufacturers:
- A substantial number of generic manufacturers have entered or are poised to enter the market as key patents expire. This list is dynamic and includes companies such as:
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Apotex
- Amneal Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- A substantial number of generic manufacturers have entered or are poised to enter the market as key patents expire. This list is dynamic and includes companies such as:
The presence of multiple generic competitors significantly impacts pricing and market share for established branded products.
What are the Therapeutic and Market Drivers for Lithium Carbonate ER?
Lithium Carbonate ER remains a cornerstone therapy for bipolar disorder, primarily for mood stabilization. Its efficacy in treating manic and hypomanic episodes, as well as in preventing future mood swings, drives consistent demand.
Key Demand Drivers:
- Established Efficacy: Lithium carbonate has demonstrated long-term effectiveness in managing bipolar disorder across diverse patient populations.
- Patient Compliance: Extended-release formulations are crucial for improving patient adherence by reducing dosing frequency and mitigating side effects associated with immediate-release formulations, such as gastrointestinal distress and tremors.
- Bipolar Disorder Prevalence: The estimated global prevalence of bipolar disorder is around 1-3% of the adult population, representing a substantial patient base. In the United States, this translates to millions of individuals requiring mood stabilization. (Source: National Institute of Mental Health [1])
- Prophylactic Use: Lithium's role in preventing manic and depressive relapses contributes to its sustained use in maintenance therapy.
- Off-Label Use: While primarily indicated for bipolar disorder, lithium has been explored for other psychiatric conditions, including major depressive disorder and certain personality disorders, contributing to a broader, albeit smaller, demand base.
The demand is relatively stable, driven by the chronic nature of bipolar disorder. However, the market is sensitive to the introduction of newer psychiatric medications that may offer perceived advantages in tolerability or efficacy for certain patient segments.
How Will Patent Expirations Affect Market Competition and Pricing?
The expiration of key patents covering Lithium Carbonate ER formulations is the primary catalyst for significant market shifts. Historically, branded Lithium Carbonate ER products have commanded higher prices due to the absence of direct generic competition and the perceived value of proprietary extended-release technology.
Impact of Patent Expirations:
- Generic Entry: As patents expire, generic manufacturers are legally able to produce and market bioequivalent versions of the branded drug. This typically leads to a rapid increase in the number of market competitors.
- Price Erosion: The introduction of multiple generic alternatives invariably drives down prices. The competitive pricing landscape among generics can lead to significant price reductions from the originator brand's price, often exceeding 50-80% within the first few years of generic availability.
- Market Share Shift: Branded products experience a substantial loss of market share to generics, particularly in price-sensitive markets or those with favorable reimbursement policies for generics.
- Formulation Differentiation: Companies holding patents on novel or superior extended-release technologies may seek to extend their market advantage by developing next-generation formulations or combination products. However, the cost and time associated with new patent filings and approvals must be weighed against the declining market for older formulations.
The period between 2025 and 2030 is anticipated to see several core Lithium Carbonate ER formulation patents lapse, triggering increased generic competition and subsequent price declines.
What are the Projected Price Trends for Lithium Carbonate ER?
Price projections for Lithium Carbonate ER are directly correlated with patent expiration dates and the subsequent influx of generic competition.
Price Projection Scenarios:
- Current Market (Pre-Major Expiration): Branded Lithium Carbonate ER products are priced at a premium. For instance, a 30-day supply of branded Lithium Carbonate ER (e.g., 300mg capsules) might range from $50 to $150 or more, depending on the specific product, dosage, and pharmacy. (Note: Actual prices vary based on insurance, formulary status, and geographic location.)
- Immediate Post-Expiration (1-2 Years): Upon the first significant generic entry for a particular formulation, prices for the branded product will likely see an initial decline of 20-40% as payers and providers start to incorporate generic options. Generic prices will stabilize at approximately 30-60% of the originator price.
- Mature Generic Market (3-5 Years Post-Expiration): With multiple generic manufacturers competing, prices will continue to decrease. Branded products may cease to be competitive or be discontinued. Generic prices for 300mg capsules could fall to $10-$30 per 30-day supply, representing a 70-90% reduction from the original branded price.
- Long-Term Projection (5+ Years): Prices will likely remain low and stable within the generic range, with minor fluctuations based on supply chain dynamics and competitive pressures. The market will be dominated by generic versions, and the price will reflect the cost of manufacturing and distribution.
Table 1: Projected Price Trends for Lithium Carbonate ER (30-Day Supply)
| Timeframe | Branded Price Range (USD) | Generic Price Range (USD) | Key Market Event |
|---|---|---|---|
| Current (Pre-2025) | $50 - $150+ | N/A | Strong patent protection |
| 1-2 Years Post-Exp (2025-2027) | $30 - $100 | $15 - $60 | Initial generic entry |
| 3-5 Years Post-Exp (2027-2030) | $10 - $50 | $10 - $30 | Mature generic market |
| 5+ Years Post-Exp (Post-2030) | N/A (Discontinued) | $8 - $25 | Dominated by generics |
These projections are based on historical trends for similar off-patent drugs with proprietary formulations. The pace of price decline can be influenced by the number of generic competitors, the complexity of the formulation, and reimbursement policies.
What are the Potential R&D Opportunities in Lithium Carbonate ER?
Despite the drug's age, opportunities exist for innovation in Lithium Carbonate ER, primarily focused on improving patient outcomes and addressing existing limitations.
R&D Opportunities:
- Novel Delivery Systems: Development of ultra-long-acting formulations (e.g., monthly injectables, long-acting implants) could significantly improve patient adherence and reduce the frequency of therapeutic drug monitoring.
- Therapeutic Drug Monitoring Optimization: Research into biomarkers or pharmacogenetic approaches that can predict optimal dosing and therapeutic response, thereby reducing the need for frequent blood draws and dose adjustments.
- Combination Therapies: Investigating synergistic effects of lithium carbonate with newer psychotropic agents to enhance efficacy or reduce side effects for specific patient subgroups.
- Improved Tolerability Profiles: Developing formulations or salt forms that further minimize common side effects such as tremor, cognitive impairment, or thyroid/renal dysfunction.
- Patient-Specific Dosing: Leveraging digital health tools and real-world data to create personalized dosing algorithms that optimize therapeutic levels while minimizing toxicity.
These R&D avenues aim to differentiate products in a potentially commoditized generic market or to re-establish a premium positioning based on superior clinical benefits.
Key Takeaways
- Lithium Carbonate ER's core molecule is generic, but extended-release formulations are protected by patents expiring primarily between 2025 and 2030.
- Generic entry following patent expirations will lead to significant price erosion, reducing branded product market share.
- The market is characterized by established brands and a growing number of generic manufacturers.
- Demand is driven by the stable prevalence of bipolar disorder and the therapeutic benefit of mood stabilization.
- Future R&D opportunities lie in novel delivery systems, personalized dosing, and combination therapies to enhance patient outcomes and tolerability.
Frequently Asked Questions
-
When are the primary patents for Lithobid expected to expire? The key formulation patents for Lithobid have been subject to litigation and vary in their expiration timelines, with some core patents and their extensions expiring in the mid-to-late 2020s, paving the way for broader generic competition.
-
How much can generic Lithium Carbonate ER prices decrease compared to the branded version? Generic Lithium Carbonate ER prices typically fall by 70% to 90% from the original branded price in a mature generic market, driven by intense competition among multiple manufacturers.
-
Are there any significant side effects associated with Lithium Carbonate ER that R&D is trying to address? Yes, common side effects include tremors, cognitive impairment, and potential long-term effects on thyroid and kidney function. R&D efforts focus on improving tolerability through novel formulations or personalized dosing to mitigate these risks.
-
What is the estimated market size for Lithium Carbonate ER globally? While exact figures for ER formulations are not always segregated, the overall lithium market for psychiatric use is estimated to be in the hundreds of millions of dollars annually, with the ER segment representing a significant portion due to its preferred dosing profile.
-
Can new patents be filed on Lithium Carbonate ER formulations to extend market exclusivity? Yes, companies can file new patents on novel extended-release technologies, manufacturing processes, or combination therapies involving lithium carbonate. However, the success and market impact of these new patents depend on their strength, the competitive landscape, and their ability to offer significant therapeutic advantages.
Citations
[1] National Institute of Mental Health. (n.d.). Bipolar Disorder. Retrieved from https://www.nimh.nih.gov/health/topics/bipolar-disorder
More… ↓
