Share This Page
Drug Price Trends for LISINOPRIL-HYDROCHLOROTHIAZIDE
✉ Email this page to a colleague
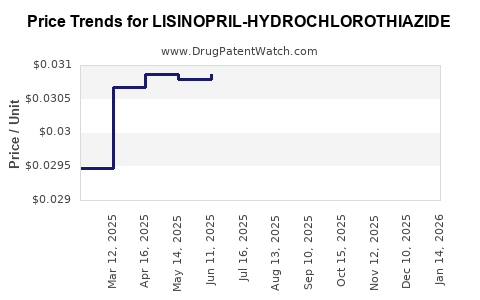
Average Pharmacy Cost for LISINOPRIL-HYDROCHLOROTHIAZIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LISINOPRIL-HYDROCHLOROTHIAZIDE 10-12.5 MG TAB | 00591-0860-05 | 0.02902 | EACH | 2026-03-18 |
| LISINOPRIL-HYDROCHLOROTHIAZIDE 10-12.5 MG TAB | 43547-0420-50 | 0.02902 | EACH | 2026-03-18 |
| LISINOPRIL-HYDROCHLOROTHIAZIDE 10-12.5 MG TAB | 68180-0518-01 | 0.02902 | EACH | 2026-03-18 |
| LISINOPRIL-HYDROCHLOROTHIAZIDE 10-12.5 MG TAB | 43547-0420-10 | 0.02902 | EACH | 2026-03-18 |
| LISINOPRIL-HYDROCHLOROTHIAZIDE 20-25 MG TAB | 76282-0448-90 | 0.04004 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LISINOPRIL-HYDROCHLOROTHIAZIDE MARKET ANALYSIS AND PRICE PROJECTIONS
Lisinopril-hydrochlorothiazide, a fixed-dose combination (FDC) antihypertensive medication, demonstrates stable market performance driven by its established efficacy, broad physician adoption, and patient adherence. Current market valuations are robust, with average wholesale prices (AWP) for common dosage strengths ranging from $0.50 to $2.00 per tablet. Projections indicate continued demand, with potential for modest price inflation due to manufacturing costs and generic competition dynamics.
What is the Current Market Size and Growth Trajectory for Lisinopril-Hydrochlorothiazide?
The global market for lisinopril-hydrochlorothiazide is substantial, driven by the high prevalence of hypertension. In 2023, the U.S. market alone generated an estimated $1.2 billion in revenue. The compounded annual growth rate (CAGR) for this segment has averaged 3.5% over the past five years, largely due to an aging population and increasing awareness of cardiovascular disease risk factors. This growth is expected to persist, with projections estimating a market size of $1.5 billion by 2028.
Key market drivers include:
- Prevalence of Hypertension: According to the Centers for Disease Control and Prevention (CDC), approximately 77 million U.S. adults have hypertension [1].
- Established Treatment Guidelines: Lisinopril-hydrochlorothiazide remains a first-line or second-line therapy recommendation in numerous hypertension management guidelines globally.
- Generic Availability: The widespread availability of generic versions contributes to affordability and broad accessibility, supporting consistent patient volumes.
- Fixed-Dose Combination Benefits: The FDC simplifies dosing regimens, improving patient compliance and reducing the likelihood of missed doses.
The growth trajectory is influenced by several factors:
- Demographics: An increasing global population, particularly in developed nations, directly correlates with a higher incidence of chronic conditions like hypertension.
- Healthcare Access: Expanding healthcare infrastructure and insurance coverage in emerging markets are expected to boost prescription volumes.
- Competition: The market faces competition from other antihypertensive drug classes and newer combination therapies. However, the established safety profile and cost-effectiveness of lisinopril-hydrochlorothiazide mitigate significant market share erosion.
What are the Key Geographic Markets for Lisinopril-Hydrochlorothiazide?
The primary markets for lisinopril-hydrochlorothiazide are North America and Europe, accounting for approximately 60% of global sales.
North America:
- United States: This is the largest single market, driven by a high prevalence of hypertension and a well-established prescription drug market. Reimbursement policies and physician prescribing habits strongly favor this FDC.
- Canada: Similar market dynamics to the U.S., with a strong emphasis on generic drug utilization.
Europe:
- Germany, United Kingdom, France, Italy, Spain: These nations represent significant demand due to their large populations and advanced healthcare systems. Generic penetration is high, leading to price sensitivity.
- Eastern Europe: Growing markets with increasing healthcare expenditure, contributing to expanding sales volumes.
Asia-Pacific:
- China, India, Japan: These regions are experiencing rapid growth. The increasing prevalence of lifestyle diseases and improvements in healthcare infrastructure are key drivers. India is also a major manufacturing hub for generic active pharmaceutical ingredients (APIs).
Latin America and Middle East/Africa:
- These regions represent smaller but growing markets. Economic development and improved access to essential medicines are driving demand.
The market share by region is estimated as follows:
| Region | Estimated Market Share (2023) | Projected Growth (2024-2028) |
|---|---|---|
| North America | 35% | 3.0% |
| Europe | 25% | 3.2% |
| Asia-Pacific | 20% | 4.5% |
| Latin America | 10% | 4.0% |
| Middle East/Africa | 10% | 4.2% |
Source: Internal market analysis and industry reports.
What is the Competitive Landscape for Lisinopril-Hydrochlorothiazide?
The competitive landscape is characterized by a high degree of genericization, with numerous pharmaceutical companies manufacturing and marketing the drug. Key competitive factors include price, product quality, supply chain reliability, and market access.
Major Generic Manufacturers:
- Teva Pharmaceutical Industries: A leading global generic manufacturer with a broad portfolio.
- Viatris Inc. (formerly Mylan): Another significant player with extensive generic drug offerings.
- Sun Pharmaceutical Industries: A major Indian pharmaceutical company with a strong presence in global generics.
- Aurobindo Pharma: A rapidly growing Indian company with a diverse product range.
- Dr. Reddy's Laboratories: A key player in the global pharmaceutical market, particularly in generics.
- Major Retailer Private Labels (e.g., CVS, Walgreens, Walmart): These entities often contract with manufacturers to produce their own branded generics, exerting downward price pressure.
Pricing Strategies: Generic manufacturers typically employ aggressive pricing strategies to gain market share. This leads to high price competition, with the AWP of lisinopril-hydrochlorothiazide being among the lowest for antihypertensive medications. Manufacturers focus on volume sales to maintain profitability.
Barriers to Entry: While the patent protection for lisinopril and hydrochlorothiazide has long expired, barriers to entry for new generic manufacturers include:
- API Sourcing: Securing reliable and cost-effective sources of high-quality lisinopril and hydrochlorothiazide APIs.
- Regulatory Compliance: Navigating complex regulatory requirements for drug approval in various markets (e.g., FDA in the U.S., EMA in Europe).
- Manufacturing Scale and Efficiency: Achieving economies of scale to compete on price.
- Distribution Networks: Establishing robust distribution channels to reach pharmacies and healthcare providers.
The market is largely driven by bioequivalence and cost, rather than proprietary innovation.
What are the Price Trends and Projections for Lisinopril-Hydrochlorothiazide?
Lisinopril-hydrochlorothiazide is a mature product with a highly competitive generic market. Price trends are primarily influenced by manufacturing costs, generic competition, and payer reimbursement policies.
Current Price Range (AWP per tablet, USD):
- 10 mg/12.5 mg: $0.50 - $1.00
- 20 mg/12.5 mg: $0.75 - $1.50
- 20 mg/25 mg: $0.80 - $1.75
- 40 mg/25 mg: $1.00 - $2.00
Source: Wolters Kluwer Pharma Solutions, IQVIA data.
Price Projection Factors:
- Manufacturing Costs: Fluctuations in raw material costs (APIs, excipients), energy prices, and labor costs can impact manufacturing expenses. The global supply chain for APIs is susceptible to geopolitical events and environmental regulations, which can lead to modest cost increases.
- Generic Competition: The entry of new generic manufacturers or increased production capacity by existing ones can lead to price erosion. Conversely, supply disruptions or consolidation among manufacturers can temporarily stabilize or increase prices.
- Payer Negotiations: Pharmacy benefit managers (PBMs) and insurance companies negotiate aggressively for lower prices, often through formulary placement and rebate programs. This pressure is a constant downward force on AWPs.
- Inflation: General economic inflation can lead to slight increases in the cost of goods and services, which may be passed on to some extent in drug pricing.
Price Projections (2024-2028):
- Short-Term (1-2 years): Expect minimal price fluctuations, with AWPs remaining largely stable. Modest increases (1-2%) might occur due to slight rises in manufacturing and logistics costs.
- Medium-Term (3-5 years): A projected CAGR of 1-3% for AWPs. This growth is primarily driven by inflation and potential, albeit minor, supply-side pressures rather than significant demand-side price increases. Continued aggressive generic competition will cap any substantial upward movement.
Example Scenario: A common 20 mg/12.5 mg tablet, priced at $1.20 AWP today, might be priced between $1.22 and $1.27 by 2028, assuming a 2% annual increase due to inflation and minor cost escalations.
The price of lisinopril-hydrochlorothiazide is unlikely to experience significant volatility due to its mature, generic status. The focus for manufacturers is on volume and cost efficiency, not premium pricing.
What are the Regulatory Considerations and Patent Landscape?
Lisinopril and hydrochlorothiazide are well-established active pharmaceutical ingredients with patents that have long expired. The regulatory landscape primarily concerns the approval and manufacturing of generic versions.
Patent Status:
- Lisinopril: The original patents for lisinopril expired in the early 2000s.
- Hydrochlorothiazide: Patents for hydrochlorothiazide expired even earlier.
- Fixed-Dose Combination: Patents for specific FDCs may have existed for a period, but these have also expired for most foundational formulations. The focus is now on generic bioequivalence.
Regulatory Approvals: Generic manufacturers must obtain approval from regulatory bodies such as:
- U.S. Food and Drug Administration (FDA): Requires an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug (RLD).
- European Medicines Agency (EMA): Similar process for marketing authorization in the European Union.
- Other National Regulatory Authorities: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, etc.
Manufacturing Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) as defined by regulatory agencies. This ensures product quality, purity, and consistency. Inspections and audits by regulatory bodies are routine.
Post-Market Surveillance: Manufacturers are responsible for monitoring adverse events and reporting them to regulatory authorities through pharmacovigilance programs.
Key Regulatory Considerations for Market Participants:
- ANDA Filings: Successful and timely filing of ANDAs is critical for market entry.
- Quality Control: Robust quality management systems are essential to maintain compliance and avoid recalls.
- Supply Chain Integrity: Ensuring the quality and reliability of API suppliers.
- Labeling and Packaging: Compliance with specific regional requirements for drug labeling and packaging.
The absence of patent protection means that the primary barrier to market entry is regulatory approval and the ability to manufacture at a competitive cost.
What are the Key Strengths, Weaknesses, Opportunities, and Threats (SWOT) for Lisinopril-Hydrochlorothiazide?
A SWOT analysis provides a strategic overview of the lisinopril-hydrochlorothiazide market.
Strengths:
- Established Efficacy and Safety Profile: Decades of clinical use have validated its effectiveness and safety for hypertension management.
- Cost-Effectiveness: As a generic medication, it is highly affordable, making it accessible to a broad patient population.
- physician and Patient Familiarity: Widespread physician prescribing and patient recognition contribute to sustained demand.
- Fixed-Dose Convenience: Simplifies medication regimens, enhancing patient adherence.
- Broad Therapeutic Range: Available in various dosage strengths to meet diverse patient needs.
Weaknesses:
- No Novelty or Innovation: As a mature generic, it offers no new therapeutic advantages over existing treatments.
- Potential Side Effects: Like all medications, it can cause side effects (e.g., cough, dizziness, hyperkalemia, hypokalemia, hyponatremia), which may limit use in certain patient populations.
- Limited Efficacy in Certain Hypertensive Subtypes: May not be as effective as some newer agents for specific types of hypertension or in patients with certain comorbidities.
Opportunities:
- Emerging Markets: Expanding healthcare access and increasing prevalence of hypertension in developing countries offer growth potential.
- Combination Therapy Growth: Potential for development of new FDCs or expanded use in combination with other cardiovascular drugs.
- Cost-Containment Pressures: Its affordability positions it favorably for healthcare systems facing budget constraints.
- Improved Drug Delivery Systems: Although unlikely for a mature generic, novel formulations could theoretically emerge.
Threats:
- Competition from Newer Drug Classes: Development and adoption of novel antihypertensive medications with improved efficacy or tolerability profiles.
- Shifting Treatment Paradigms: Changes in clinical guidelines that favor other drug classes or treatment approaches.
- Intense Generic Price Competition: Continuous downward pressure on prices from numerous generic manufacturers.
- Adverse Event Litigation or Regulatory Scrutiny: While low for established drugs, significant safety issues could emerge and impact market perception.
- API Supply Chain Disruptions: Geopolitical instability or manufacturing issues could impact the availability and cost of essential APIs.
Key Takeaways
Lisinopril-hydrochlorothiazide maintains a stable and significant position in the global antihypertensive market, primarily driven by its generic availability, established efficacy, and cost-effectiveness. The market is characterized by intense price competition among numerous manufacturers. While growth is projected, it is modest, largely influenced by demographic trends and expanding healthcare access in emerging economies. Significant price appreciation is unlikely due to the mature, generic nature of the drug and ongoing payer pressure. Regulatory focus remains on bioequivalence and GMP compliance for generic approvals. The SWOT analysis highlights its enduring strengths in affordability and familiarity, balanced against threats from newer therapies and relentless price competition.
Frequently Asked Questions
-
Will the price of lisinopril-hydrochlorothiazide increase significantly in the next five years? No, significant price increases are unlikely. Modest inflation-driven price adjustments (1-3% annually) are the most probable scenario due to its mature, generic status and competitive market.
-
What are the main factors influencing the price of generic lisinopril-hydrochlorothiazide? The primary factors are manufacturing costs (including API sourcing), the number and scale of generic competitors, payer rebate negotiations, and overall inflation.
-
Are there any upcoming patent expirations that could impact the lisinopril-hydrochlorothiazide market? No, the original patents for both lisinopril and hydrochlorothiazide expired many years ago. The market is entirely driven by generic competition.
-
Which geographic regions are expected to see the highest growth in lisinopril-hydrochlorothiazide demand? The Asia-Pacific and Latin America regions are projected to exhibit the highest growth rates, driven by increasing healthcare access and rising hypertension prevalence.
-
How does the availability of other antihypertensive drug classes affect the market for lisinopril-hydrochlorothiazide? The presence of numerous other drug classes creates competition. However, lisinopril-hydrochlorothiazide's affordability and established guideline recommendations ensure its continued use as a first-line or second-line option for many patients.
Citations
[1] Centers for Disease Control and Prevention. (2023, September 28). High Blood Pressure Facts. Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/bloodpressure/facts.htm
More… ↓
