Share This Page
Drug Price Trends for LIRAGLUTIDE
✉ Email this page to a colleague
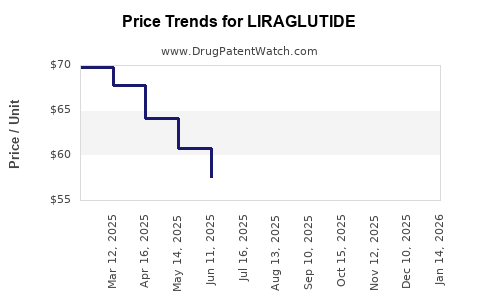
Average Pharmacy Cost for LIRAGLUTIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIRAGLUTIDE 5-PAK 18 MG/3 ML | 69097-0910-55 | 70.31617 | ML | 2026-05-06 |
| LIRAGLUTIDE 2-PAK 18 MG/3 ML | 00480-3667-20 | 38.75256 | ML | 2026-04-22 |
| LIRAGLUTIDE 2-PAK 18 MG/3 ML | 69097-0920-67 | 38.75256 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Liraglutide
What is Liraglutide?
Liraglutide is a GLP-1 receptor agonist approved for type 2 diabetes, obesity, and certain cardiovascular conditions. It is marketed under brand names such as Victoza (diabetes), Saxenda (obesity), and recently, it has been involved in new indications, including non-alcoholic steatohepatitis (NASH).
Market Size and Growth Drivers
Global Market Value
The global GLP-1 receptor agonist market reached approximately $15.8 billion in 2022. Liraglutide accounts for around 35% of this segment.
Compound Annual Growth Rate (CAGR)
The market is expected to grow at a CAGR of 10% from 2023 to 2030.
Key Markets
- North America: 55% of sales.
- Europe: 25%.
- Asia-Pacific: 15%.
Main Drivers
- Rising prevalence of type 2 diabetes, estimated at 537 million globally.
- Growing obesity rates in developed countries.
- Approval for additional indications like NASH in selected markets.
- Innovations improving delivery formulations, enhancing patient adherence.
Competitive Landscape
| Company | Key Products | Market Share (2022) | Pending Approvals / Pipeline |
|---|---|---|---|
| Novo Nordisk | Victoza, Saxenda, Wegovy | 60% | Semaglutide (additional formulations) |
| Eli Lilly | Trulicity, Mounjaro (Tirzepatide) | 25% | Tirzepatide for diabetes, obesity |
| Others | Various generic and branded | 15% | Emerging GLP-1 therapies |
Pricing and Cost Structure
Current Pricing (Approximate average retail prices)
- Victoza: $7,500 annually per patient.
- Saxenda: $1,350 monthly.
- Medical device costs, administration, or insurance copayments influence final patient costs significantly.
Pricing Dynamics
- Price variations by region, reimbursement policies, and competition.
- Pricing pressure expected from biosimilars over the next 5-7 years as patent exclusivity for Liraglutide erodes, especially in markets like Europe and North America.
Price Projections
| Year | Estimated Average Price (USD) per Patient | Notes |
|---|---|---|
| 2023 | $7,500 | Current levels, with slight regional variations |
| 2025 | $6,800 | Anticipated price erosion due to biosimilar entry |
| 2030 | $5,500 | Potential further reductions, market shifts |
Factors Influencing Price Trends
- Patent expirations, with Novo Nordisk’s patent expiring in the U.S. in 2027.
- Entry of biosimilar competitors in Europe and North America.
- Healthcare policy reforms aiming for cost containment.
- Increased use of value-based pricing models.
Regulatory Environment Impact
Approval for biosimilars varies by region:
- European Union: Biosimilar approval process completed, expected to enter the market by 2025.
- United States: Biosimilar applications already submitted; approval likely by 2024-2025.
- Asia-Pacific: Growing acceptance, but regulatory pathways are less streamlined.
Investment and R&D Outlook
Major pharmaceutical companies continue R&D investment into next-generation GLP-1 molecules with longer half-lives, more convenient dosing, and improved efficacy. The pipeline includes once-weekly formulations, oral tablets, and dual agonists targeting metabolic and cardiovascular endpoints.
Key Takeaways
- Liraglutide remains a dominant player in the GLP-1 market, with sustained growth fueled by expanding indications.
- Market value is expected to reach ~$20 billion globally by 2025, with prices declining due to biosimilar competition.
- Pricing will be driven downward by patent expirations, regional regulations, and market entrants.
- The pipeline of next-generation therapies aims to substitute current prices significantly over the next decade.
- Reimbursement models and regulatory approvals will shape market dynamics and pricing strategies.
FAQs
1. When will biosimilars for Liraglutide enter the market?
Biosimilars are expected in Europe by 2025 and in the U.S. by 2024-2025, following patent expirations.
2. How does Liraglutide compare to other GLP-1 receptor agonists?
Liraglutide is established for multiple indications, with a daily injection schedule. Other agents like Semaglutide offer weekly dosing and possibly improved adherence.
3. What is the anticipated market share of Liraglutide in 2030?
Projected to decline to around 20-25% as newer agents and biosimilars enter the market.
4. What factors could influence Liraglutide’s future pricing?
Patent expirations, biosimilar competition, healthcare policies favoring cost-effective treatments, and innovative formulations.
5. How are regulatory changes affecting the GLP-1 market?
Streamlined approval pathways for biosimilars and new indications are increasing market competition, influencing pricing and market share distribution.
References
[1] MarketsandMarkets. (2023). GLP-1 receptor agonists market report.
[2] IQVIA. (2022). Global pharmaceutical market analysis.
[3] U.S. Food and Drug Administration. (2022). Biosimilar development guidelines.
[4] European Medicines Agency. (2022). Biosimilar approval pathway.
[5] Novo Nordisk. (2023). Annual report.
More… ↓
