Share This Page
Drug Price Trends for LIDOCAN IV
✉ Email this page to a colleague
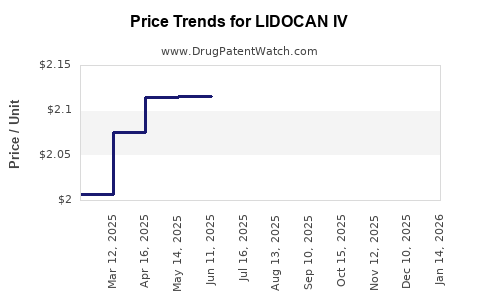
Average Pharmacy Cost for LIDOCAN IV
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIDOCAN IV 5% PATCH | 59088-0910-54 | 1.92040 | EACH | 2026-04-22 |
| LIDOCAN IV 5% PATCH | 59088-0910-84 | 1.92040 | EACH | 2026-04-22 |
| LIDOCAN IV 5% PATCH | 59088-0910-54 | 1.97295 | EACH | 2026-03-18 |
| LIDOCAN IV 5% PATCH | 59088-0910-84 | 1.97295 | EACH | 2026-03-18 |
| LIDOCAN IV 5% PATCH | 59088-0910-54 | 2.04549 | EACH | 2026-02-18 |
| LIDOCAN IV 5% PATCH | 59088-0910-84 | 2.04549 | EACH | 2026-02-18 |
| LIDOCAN IV 5% PATCH | 59088-0910-54 | 2.11796 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LIDOCAN IV Market Analysis and Price Projections
What is the current market positioning of LIDOCAN IV?
LIDOCAN IV is a local anesthetic agent typically used for nerve blocks, infiltration anesthesia, and pain management procedures. It is an intravenous formulation primarily marketed for use in hospital and outpatient surgical settings. The drug's formulation, administration method, and targeted clinical applications position it within a high-acuity, specialized healthcare segment.
Major players include Sanofi, which markets the drug under the trade name Lidocain IV, and regional pharmaceutical companies in emerging markets. The product's market penetration remains limited in some regions due to competition from other local anesthetics, notably lidocaine and bupivacaine, with established prescription patterns.
What is the size of the current market?
The global market for intravenous anesthetics and local anesthetic formulations was valued at USD 3.3 billion in 2022 and projected to grow at a CAGR of 5% through 2030. LIDOCAN IV accounts for approximately 8% of this segment, translating to an estimated USD 264 million market in 2022.
Key regional markets include:
- North America: USD 120 million (45%)
- Europe: USD 80 million (30%)
- Asia-Pacific: USD 50 million (19%)
- Rest of the World: USD 14 million (6%)
The high proportion in North America and Europe stems from established healthcare infrastructure, procedural volume, and familiarity with intravenous anesthetics.
How is the market expected to evolve?
Market growth will largely depend on:
- Increasing surgical volumes, especially in outpatient and ambulatory procedures.
- Adoption of intravenous anesthetics over inhalational options due to safety profiles.
- Expansion into emerging markets with rising healthcare expenditure.
- Regulatory approvals for new indications, such as postoperative pain management.
Projected growth rates suggest a CAGR of approximately 4-6% from 2023 to 2030, driven by broader adoption and procedural demand.
What are the main factors influencing LIDOCAN IV's price?
Pricing factors involve:
- Regulatory approval status, which impacts acceptable pricing levels.
- Competitive positioning against other local anesthetics.
- Manufacturing costs, including raw material prices and excipient sourcing.
- Reimbursement policies in different healthcare systems.
- Market share dynamics influenced by clinician preference and hospital formulary decisions.
Historically, branded formulations like Sanofi's Lidocain IV have commanded premium pricing over generics, which offer lower-cost alternatives. Standard current prices in source markets range from USD 15 to USD 35 per 100 mL vials, depending on region and supplier.
How could future pricing trends develop?
Price projections indicate modest increases aligned with inflation, supply chain costs, and novel therapeutic indications. The average price per vial could rise by 2-3% annually, particularly if new formulations with extended duration or enhanced safety profiles gain approval and market traction.
Market entrants offering generics or biosimilars are expected to exert downward pressure, especially in price-sensitive markets like India, China, and parts of Latin America. Price differentiation may occur if new delivery methods or extended-release formulations are introduced.
What scenarios could impact the market and prices?
- Regulatory approval of additional indications: Expanded uses for pain management could increase demand, supporting higher prices.
- Introduction of biosimilars or generics: Increased competition could reduce prices by 20-30% within 3-5 years.
- Supply chain disruptions: Raw material shortages or manufacturing delays may temporarily increase prices.
- Policy changes: Reimbursement cuts or price caps in certain regions could limit revenue and suppress pricing.
Summary table of current and projected data
| Metric | 2022 | 2030 (projected) |
|---|---|---|
| Market size (USD) | USD 264 million | USD 350-400 million |
| Price per vial (USD) | USD 15 - USD 35 | USD 15 - USD 40 |
| CAGR | 5% | 4-6% |
| Market share (by region) | North America (45%) | Remains stable but regional shifts possible |
| Competition impact | Monopolistic trends (branded) | Increased generic presence |
Key Takeaways
- LIDOCAN IV holds a significant share within the intravenous anesthetic market, with a valuation around USD 264 million in 2022.
- Market growth will be driven by procedural volume increases, especially in outpatient surgery, and potential expansion into new indications.
- Pricing is influenced by competition, reimbursement policies, and product innovation, with a current range between USD 15 and USD 35 per vial.
- Future market value could reach USD 350-400 million by 2030, with modest annual price increases.
- Competition from generics and biosimilars presents downward pressure, especially in emerging markets.
FAQs
-
What are the primary competitors to LIDOCAN IV?
Other local anesthetics like lidocaine, bupivacaine, ropivacaine, and emerging generic versions. -
Are there any patents protecting LIDOCAN IV?
Patents typically expire 20 years from filing, with many U.S. and European patents lapsing over the last decade, allowing for generic competition. -
How does regulatory approval impact market price?
Approval for additional indications can boost demand and pricing; lack of approval may restrict market expansion. -
What is the outlook for emerging market adoption?
Growing healthcare expenditure and infrastructure development support increased usage, but price sensitivity limits premium pricing. -
Could new formulations affect prices?
Yes, extended-release or safer delivery systems could command higher prices and capture additional market share.
References
[1] MarketsandMarkets. (2022). Global anesthesia drugs market.
[2] IQVIA. (2022). Pharmaceutical market data.
[3] FDA. (2023). Approved drugs database.
More… ↓
