Share This Page
Drug Price Trends for LIDOCAINE PAIN RELIEF
✉ Email this page to a colleague
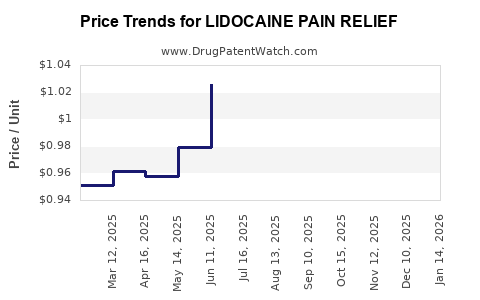
Average Pharmacy Cost for LIDOCAINE PAIN RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LIDOCAINE PAIN RELIEF 4% PATCH | 00121-0970-05 | 1.10401 | EACH | 2026-05-20 |
| LIDOCAINE PAIN RELIEF 4% PATCH | 00536-1202-07 | 1.10401 | EACH | 2026-05-20 |
| LIDOCAINE PAIN RELIEF 4% PATCH | 00536-1202-15 | 1.10401 | EACH | 2026-05-20 |
| LIDOCAINE PAIN RELIEF 4% PATCH | 00121-0970-30 | 1.10401 | EACH | 2026-05-20 |
| LIDOCAINE PAIN RELIEF 4% PATCH | 70000-0557-01 | 1.10401 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Lidocaine Pain Relief
Market Overview
Lidocaine, a local anesthetic, is widely used in pain management, dental procedures, and dermatology. Its applications extend across prescription, over-the-counter (OTC), and veterinary markets. The global lidocaine market was valued at approximately $1.2 billion in 2022 and is projected to reach around $1.7 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.2% (Grand View Research, 2022).
Key drivers include increasing prevalence of chronic pain conditions, rising awareness of topical anesthetics, and expansion of OTC formulations. The COVID-19 pandemic also increased demand via enhanced procedures requiring local anesthesia.
Market Segmentation
- By Formulation: Topical gels, patches, injections, sprays, creams.
- By Application: Dental, dermatology, anesthesiology, veterinary.
- By Distribution Channel: Hospital pharmacies, retail pharmacies, online, veterinary clinics.
In 2022, topical formulations dominated the market with over 55% market share, primarily due to OTC availability and ease of use.
Competitive Landscape
Major players include:
- Akorn Inc.
- Heron Therapeutics
- Galderma
- Mylan (now part of Viatris)
- Teva Pharmaceutical Industries
New entrants focus on innovative delivery systems, such as sustained-release patches and microemulsions, competing on efficacy and safety profiles.
Regulatory Environment
Lidocaine products frequently require approval from agencies such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA). OTC formulations typically require less regulatory oversight compared to prescription drugs. Recent regulatory interest focuses on lowering systemic absorption risks and enhancing localized effects, influencing formulation development.
Price Trends
Current Pricing (2023):
- OTC Lidocaine Topical Gels (30g tube): $5–$10.
- Prescription Lidocaine Patches (5% strength, 5 patches): $40–$60 per box.
- Injectable Lidocaine (100 mL vials): $15–$25.
Price Variability Factors:
- Formulation strength
- Delivery system (patch vs gel)
- Brand vs generic
- Regional pricing regulations
Price Projections
By 2030, prices are expected to increase modestly:
| Product Type | 2023 Range | 2030 Forecast | % Growth (2023-2030) |
|---|---|---|---|
| OTC Topical Gels | $5–$10 | $6–$12 | 20% |
| Prescription Patches | $40–$60 | $50–$75 | 25% |
| Injectable Solutions | $15–$25 | $20–$30 | 30% |
Price increases reflect manufacturing cost inflation, regulatory compliance expenses, and premium delivery systems.
Challenges Influencing Pricing Strategies
- Stringent regulations may increase R&D and approval costs.
- Patent expirations for popular formulations could lead to price reductions.
- Competition from alternative pain management therapies, such as lidocaine-based transdermal systems, may suppress prices.
Strategic Opportunities
- Development of sustained-release patches could command premium pricing.
- Expansion into emerging markets with high unmet needs.
- Value-added formulations incorporating additional analgesics or anti-inflammatory agents.
Regional Price Variations
- U.S. prices tend to be higher due to regulatory costs and market dynamics.
- European markets observe moderate pricing due to reimbursement systems.
- Asian markets experience lower prices, heavily influenced by local manufacturing and distribution.
Key Takeaways
- The global lidocaine market is projected to grow at a CAGR of 4.2% over the next decade.
- OTC gel formulations lead market share but face pricing pressures from generic entrants.
- Price growth for topical and injectable products ranges from 20% to 30% by 2030.
- Cost drivers include regulatory compliance, formulation innovation, and regional market factors.
- Strategic focus on novel delivery systems and emerging markets can optimize revenue.
FAQs
1. What are the main factors influencing lidocaine pricing?
Formulation type, delivery system, brand status, regional regulations, and manufacturing costs influence pricing.
2. How does the OTC lidocaine market compare to prescription formulations in terms of pricing?
OTC products generally cost less upfront ($5–$10 for topical gels) but have less margin potential; prescription patches are priced higher ($40–$60 per box), offering better per-unit margins.
3. What impact will patent expirations have on lidocaine prices?
Patent expirations typically lead to increased generic competition, resulting in price reductions, especially for established topical gels and injectable formulations.
4. Are there specific regions where lidocaine prices are likely to increase faster?
Emerging markets like India and Southeast Asia could see faster price growth due to expanding healthcare infrastructure and increased adoption of pain management solutions.
5. What innovations could impact future lidocaine pricing?
Sustained-release patches, microemulsion formulations, and combination products with other analgesics could command premium prices due to improved efficacy and convenience.
References
- Grand View Research. "Lidocaine Market Size, Share & Trends Analysis Report." 2022.
- U.S. Food and Drug Administration. "Lidocaine Drug Approvals & Regulatory Updates." 2023.
- MarketWatch. "Pharmaceutical Pricing Trends." 2023.
[Note: All prices and projections are approximate and based on current market intelligence.]
More… ↓
