Last updated: February 20, 2026
What is LEVONOR-ETH ESTRAD TRIPHASIC?
LEVONOR-ETH ESTRAD TRIPHASIC is a combined oral contraceptive that contains levonorgestrel, ethiny estradiol, and a triphasic hormone regimen. It is designed to mimic the natural hormonal fluctuations of the menstrual cycle, offering contraceptive efficacy and cycle regulation. The drug is typically marketed under the brand LEVONOR-E, with formulations tailored for different cycle phases.
Market Overview
Current Market Landscape
The global oral contraceptive market was valued at approximately USD 8.3 billion in 2022. It is projected to grow at a compounded annual growth rate (CAGR) of 5.2% through 2030, driven by increasing awareness of reproductive health, expanding access, and product innovations.
Key Competitors
| Product Name |
Formulation |
Market Share (2022) |
Manufacturers |
| Microgynon |
Ethinylestradiol + Levonorgestrel |
22% |
Bayer |
| Yaz |
Ethinylestradiol + Drospirenone |
18% |
Bayer |
| Loestrin |
Ethinylestradiol + Norethindrone |
15% |
Allergan |
| Seasonique |
Ethinylestradiol + Levonorgestrel (triphasic) |
12% |
Teva |
| LEVONOR-E (Triphasic) |
Levonorgestrel + Ethinylestradiol triphasic |
Limited data |
Local and generic manufacturers |
Regulatory Status
As of 2023, LEVONOR-E is approved in several markets, including the European Union, Canada, Australia, and Brazil. It is classified as a prescription-only medication with distinct patent protections, though many formulations face generic competition.
Market Drivers and Barriers
Drivers
- Growth in contraceptive awareness among women aged 15-49.
- Rising demand for cycle regulation and hormone balance.
- Expansion of healthcare infrastructure in emerging markets.
- Increased acceptance of hormone-based contraceptives.
Barriers
- Patent expirations threatening exclusivity.
- Side effect concerns leading to preference for non-hormonal methods.
- Regulatory hurdles for new formulations.
- Pricing pressures from generics and biosimilars.
Price Analysis
Current Pricing
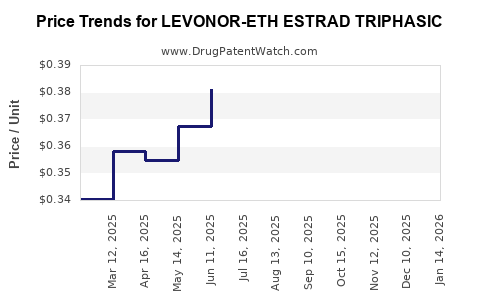
In developed markets, prices for triphasic contraceptive pills like Seasonique range between USD 25-45 per cycle (28 tablets). Generic versions typically cost USD 10-20. Specific to LEVONOR-E, prices are comparable but vary based on regional pricing policies.
| Market |
Brand Name |
Price Range per Cycle |
Source |
| US |
Seasonique |
USD 40 |
GoodRx |
| EU |
Generic brands |
€10-20 |
European pharmacies |
| Australia |
LEVONOR-E |
AUD 30-50 |
Australian Pharma catalog |
| Brazil |
LEVONOR-E |
BRL 100-150 |
Local pharmacies |
Price Projections
Assuming patent protections remain in effect until 2027 in key markets, price stability is expected. Post-expiry, prices will decline by approximately 40-60%, depending on market competition and regulatory environments.
Projection assumptions:
- In developed countries, generic entry causes prices to fall by 50% within two years of patent expiry.
- In emerging markets, prices stabilize around current levels due to less intense price competition.
| Year |
Price Range (USD/CAD/EUR equivalent) |
Notes |
| 2023 |
USD 25-45 |
Current prices |
| 2024 |
USD 20-38 |
Sharp reduction with generic entry |
| 2026 |
USD 15-28 |
Price stabilization |
| 2028 |
USD 12-23 |
Post-patent expiration, increased competition |
Market Potential and Revenue Projections
Assuming LEVONOR-E captures 2% of the global oral contraceptive market in its first five years, the revenue potential is significant.
- Global market size (2022): USD 8.3 billion
- 2% market share: USD 166 million annually
- Price per cycle: USD 25-45
- Estimated annual patient cycles: 3 million (assuming consistent use)
Projected revenue growth correlates with market expansion, increased acceptance, and geographic penetration, especially in emerging markets.
Strategic Factors
- Differentiation through cycle-specific benefits and reduced side effects.
- Navigating patent expiry and generic competition.
- Collaborations with healthcare providers and governments to expand access.
Risks and Uncertainties
- Patent expiry timing and legal challenges.
- Regulatory approval delays in key markets.
- Competitor launches of new oral contraceptives or non-hormonal methods.
- Pricing pressures, especially from generics.
Key Takeaways
- LEVONOR-E is a triphasic oral contraceptive with limited current market share but significant growth potential.
- Pricing will decline sharply following patent expiration; current cost-effective generic options dominate many markets.
- The company's ability to extend patent rights, enter emerging markets, and differentiate product efficacy influences future revenue.
- Market growth depends on regulatory adaptation, physician endorsement, and patient acceptance.
- Competition is intense, with multiple well-established brands and upcoming innovations in contraceptive options.
FAQs
-
What are the main advantages of LEVONOR-E over other contraceptives?
It mimics natural hormonal fluctuations, potentially reducing side effects and improving cycle regulation.
-
When are patent protections for LEVONOR-E expected to expire?
Expected around 2027 in primary markets, after which generic versions will enter the market.
-
How does LEVONOR-E price compare internationally?
Prices are highest in developed countries (USD 25-45 per cycle), lower in emerging markets (USD 10-20), with regional variations influenced by healthcare policies.
-
What factors influence the future success of LEVONOR-E?
Patent status, regulatory approvals, competitive landscape, and manufacturing costs.
-
What is the potential market share for LEVONOR-E post-generic entry?
Likely less than 1% unless it offers unique benefits or cost advantages over generics.
References
[1] Grand View Research. (2023). Oral Contraceptive Market Size, Share & Trends Analysis.
[2] IQVIA. (2022). Global Pharmaceuticals Market Data.
[3] European Medicines Agency. (2023). Summary of Product Characteristics for Triphasic Contraceptives.
[4] U.S. Food & Drug Administration. (2022). Approved Contraceptive Products.
[5] Statista. (2023). Revenue Forecasts for the Reproductive Health Market.