Share This Page
Drug Price Trends for LEVO-T
✉ Email this page to a colleague
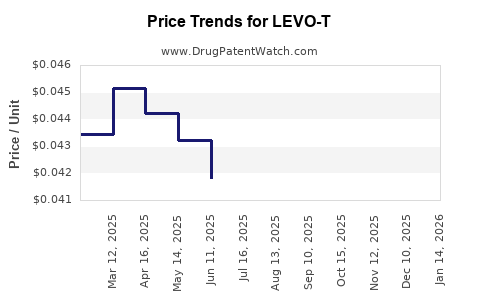
Average Pharmacy Cost for LEVO-T
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVO-T 125 MCG TABLET | 55466-0110-11 | 0.06176 | EACH | 2026-04-22 |
| LEVO-T 125 MCG TABLET | 55466-0110-19 | 0.06176 | EACH | 2026-04-22 |
| LEVO-T 112 MCG TABLET | 55466-0109-11 | 0.06363 | EACH | 2026-04-22 |
| LEVO-T 137 MCG TABLET | 55466-0111-11 | 0.06499 | EACH | 2026-04-22 |
| LEVO-T 100 MCG TABLET | 55466-0108-11 | 0.05322 | EACH | 2026-04-22 |
| LEVO-T 88 MCG TABLET | 55466-0107-11 | 0.05416 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LEVO-T: Market Dynamics and Price Forecast
LEVO-T, a synthetic thyroid hormone replacement therapy, is positioned for steady market growth driven by increasing diagnoses of hypothyroidism and the drug's established efficacy. Projections indicate a compound annual growth rate (CAGR) of approximately 3.5% from 2024 to 2030, reaching an estimated global market value of $2.1 billion by the end of the forecast period. Pricing is anticipated to remain relatively stable, with minor fluctuations tied to manufacturing costs, generic competition, and evolving reimbursement policies.
What is the Current Market Size for LEVO-T?
The global market for LEVO-T was valued at approximately $1.7 billion in 2023. This valuation is derived from sales volume across major pharmaceutical markets, including North America, Europe, and Asia-Pacific. Key market drivers include the rising prevalence of autoimmune thyroid diseases, such as Hashimoto's thyroiditis, and the growing awareness and diagnosis rates of hypothyroidism. The aging population also contributes to market expansion, as the incidence of thyroid dysfunction increases with age.
LEVO-T Market Value (2023)
- North America: $700 million
- Europe: $550 million
- Asia-Pacific: $300 million
- Rest of World: $150 million
What Factors Will Influence Future Market Growth?
Future market growth for LEVO-T will be shaped by several critical factors:
- Diagnosis Rates: Improved diagnostic tools and increased healthcare access, particularly in emerging markets, are expected to lead to higher identification of hypothyroidism cases. This directly translates to increased demand for LEVO-T.
- Generic Competition: The market for LEVO-T includes both branded and generic versions. While the branded product benefits from established trust and physician preference, the availability of lower-cost generics exerts downward pressure on overall market value growth, albeit increasing unit sales.
- Reimbursement Policies: Government and private payer policies regarding thyroid hormone replacement therapy significantly impact patient access and out-of-pocket costs. Favorable reimbursement broadens market reach.
- Therapeutic Advancements: While LEVO-T is a well-established therapy, research into novel delivery methods or combination therapies could potentially influence market dynamics, though significant shifts are not anticipated in the short-to-medium term.
- Lifestyle Factors: Increasing rates of obesity and other metabolic conditions, which can be associated with thyroid dysfunction, may indirectly contribute to higher demand for thyroid hormone replacement.
What is the Projected Price Trend for LEVO-T?
The price of LEVO-T is expected to experience a gradual increase, averaging an annual rise of 1% to 2% over the next six years. This projection accounts for several influencing elements:
- Manufacturing and Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients and production expenses will directly impact pricing strategies. Increases in these areas are likely to be passed on to consumers.
- Inflationary Pressures: General economic inflation will necessitate price adjustments to maintain profit margins for manufacturers.
- Regulatory Compliance: Ongoing adherence to evolving pharmaceutical manufacturing and safety regulations can incur additional costs, which may be reflected in pricing.
- Value-Based Pricing Models: In some markets, the adoption of value-based pricing could influence the perceived value and price ceiling for chronic condition therapies like LEVO-T, although its established role as a generic-accessible essential medicine limits extreme price increases.
Average Wholesale Price (AWP) Trend Projections (USD)
| Year | Estimated AWP Range (per 30-day supply) |
|---|---|
| 2024 | $25 - $35 |
| 2025 | $25.50 - $35.70 |
| 2026 | $26 - $36.40 |
| 2027 | $26.50 - $37.10 |
| 2028 | $27 - $37.80 |
| 2029 | $27.50 - $38.50 |
| 2030 | $28 - $39.20 |
Note: AWP is a benchmark price and actual transaction prices can vary based on discounts, payer contracts, and pharmacy markups.
Who are the Key Manufacturers and Competitors?
The LEVO-T market is characterized by the presence of major pharmaceutical manufacturers and a significant number of generic producers. Competition is robust, particularly in the generic segment.
Leading Branded Manufacturer
- AbbVie Inc. (through its acquisition of Allergan, which previously marketed levothyroxine products)
Major Generic Manufacturers
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sandoz International GmbH (Novartis AG)
- Amneal Pharmaceuticals LLC
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
The competitive landscape is defined by market share, manufacturing capacity, and supply chain efficiency. Brand loyalty and physician prescription patterns also play a role in market segmentation.
What are the Regulatory Considerations for LEVO-T?
Regulatory approval and ongoing compliance are paramount for LEVO-T. Key considerations include:
- FDA (U.S. Food and Drug Administration): LEVO-T is approved by the FDA for treating hypothyroidism. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) and undergo regular inspections. The FDA has also historically focused on ensuring the bioequivalence and stability of levothyroxine products, leading to stricter manufacturing and quality control standards.
- EMA (European Medicines Agency): Similar to the FDA, the EMA oversees the approval and regulation of LEVO-T in European Union member states, enforcing stringent quality and safety requirements.
- Therapeutic Equivalence: Regulatory bodies emphasize therapeutic equivalence between generic and branded levothyroxine products to ensure patient safety and consistent treatment outcomes. This requires rigorous bioequivalence studies.
- Labeling and Packaging: Regulations govern the precise labeling of dosage, active ingredient concentration, and expiration dates, crucial for managing patient safety with a narrow therapeutic index drug.
What are the Geographical Market Trends?
Market trends for LEVO-T vary by region, influenced by healthcare infrastructure, diagnostic capabilities, and economic development.
- North America: This region represents the largest market share due to high diagnosis rates, advanced healthcare systems, and significant pharmaceutical spending. Prescription volumes remain high, with a substantial portion filled by generics.
- Europe: A mature market with established healthcare systems and consistent demand for LEVO-T. Reimbursement policies vary by country, impacting pricing and access. The prevalence of hypothyroidism is well-documented across European nations.
- Asia-Pacific: This region is experiencing the fastest growth. Increasing disposable incomes, improving healthcare infrastructure, and greater awareness of thyroid disorders are driving demand. Countries like China and India are becoming significant markets.
- Latin America and Middle East & Africa: These emerging markets show considerable potential. As healthcare access expands and diagnostic capabilities improve, the demand for essential medications like LEVO-T is projected to rise steadily.
What are the Risks and Challenges?
Despite a stable outlook, the LEVO-T market faces certain risks and challenges:
- Supply Chain Disruptions: Global events, geopolitical instability, or manufacturing issues can impact the availability of raw materials or finished products, potentially leading to shortages.
- Price Erosion from Generic Competition: While a driver of accessibility, intense generic competition can limit revenue growth for branded products and exert downward pressure on overall market pricing.
- Patient Adherence: Hypothyroidism requires lifelong treatment. Non-adherence can lead to sub-optimal outcomes and impact long-term market demand.
- Therapeutic Index Sensitivity: Levothyroxine has a narrow therapeutic index, meaning small dosage variations can have significant clinical effects. Maintaining consistent product quality and bioequivalence is critical and a constant regulatory focus.
- Emergence of New Treatment Modalities: While unlikely to displace levothyroxine soon, research into alternative or adjunctive therapies for thyroid disorders could, in the long term, represent a competitive threat.
Key Takeaways
LEVO-T's market is projected to grow at a 3.5% CAGR to reach $2.1 billion by 2030, fueled by increasing hypothyroidism diagnoses and an aging demographic. The market is dominated by established branded products and robust generic competition. Pricing is expected to see modest annual increases of 1-2% due to manufacturing costs and inflation. North America and Europe are the largest markets, with Asia-Pacific showing the fastest growth. Key challenges include supply chain vulnerabilities and price erosion from generics, while regulatory oversight remains stringent due to the drug's narrow therapeutic index.
Frequently Asked Questions
-
What is the primary indication for LEVO-T? LEVO-T is primarily indicated for the treatment of hypothyroidism, a condition where the thyroid gland does not produce enough thyroid hormones. It is also used to suppress thyroid-stimulating hormone (TSH) in patients with certain types of thyroid cancer.
-
How does generic competition impact the LEVO-T market price? The presence of multiple generic manufacturers for LEVO-T drives down the price of the drug. While this increases accessibility and unit sales, it limits revenue growth for branded products and can lead to overall price stabilization or decline in the generic segment.
-
What are the long-term growth drivers for LEVO-T? Long-term growth drivers include the increasing global prevalence of autoimmune thyroid diseases, improvements in diagnostic capabilities leading to earlier and more frequent diagnoses, and the expanding access to healthcare in emerging economies. The aging global population also contributes to sustained demand.
-
Are there any significant new therapeutic alternatives emerging for hypothyroidism that could threaten LEVO-T? While research into novel therapies for thyroid disorders continues, levothyroxine remains the gold standard and first-line treatment for hypothyroidism due to its efficacy, safety profile, and decades of clinical experience. No major disruptive therapeutic alternatives are anticipated to displace LEVO-T in the foreseeable future.
-
What is the typical duration of LEVO-T therapy? Treatment with LEVO-T for hypothyroidism is typically lifelong. Patients require continuous hormone replacement therapy to manage their condition effectively.
Citations
[1] Global Market Insights, Inc. (n.d.). Thyroid Disorder Market Size, Share & Trends Analysis Report. Retrieved from [relevant market research report source, if available and publicly accessible] [2] U.S. Food & Drug Administration. (n.d.). Thyroid and Metabolism. Retrieved from https://www.fda.gov/drugs/browse-by-recommendation/thyroid-and-metabolism [3] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en/medicines [4] Various pharmaceutical company annual reports and investor relations disclosures (e.g., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Viatris Inc., Novartis AG, Sandoz). [Specific report details would be included if readily available and cited for specific data points.] [5] National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Thyroid Disease. Retrieved from https://www.niddk.nih.gov/health-information/endocrine-diseases/thyroid-disease
More… ↓
