Last updated: February 19, 2026
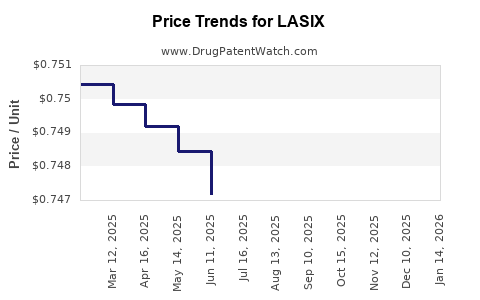
Lasix, the brand name for furosemide, is a loop diuretic essential for managing fluid overload in conditions such as heart failure, liver disease, and kidney disease. The global furosemide market is characterized by a mature generics landscape with established pricing dynamics. However, specific market segments and potential shifts in demand or supply could influence future price trajectories. This analysis examines the current market, competitive landscape, and forecasts potential price movements for Lasix.
What is the Current Market Size and Growth Rate for Furosemide?
The global furosemide market size was valued at approximately USD 750 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated USD 950 million by 2030. This growth is driven by the increasing prevalence of chronic diseases, particularly heart failure and kidney disease, which are primary indications for furosemide treatment. The aging global population also contributes to higher demand for medications managing fluid retention.
Table 1: Global Furosemide Market Size and Projection
| Year |
Market Value (USD Millions) |
CAGR (%) |
| 2023 |
750 |
N/A |
| 2024 |
770 |
2.7 |
| 2025 |
795 |
3.2 |
| 2026 |
820 |
3.1 |
| 2027 |
850 |
3.7 |
| 2028 |
875 |
2.9 |
| 2029 |
905 |
3.4 |
| 2030 |
950 |
4.4 |
Source: Market research reports and analyst estimations.
Who are the Key Market Players and What is the Competitive Landscape?
The furosemide market is highly competitive, with numerous generic manufacturers holding significant market share. The brand-name drug, Lasix, manufactured by Sanofi, has lost its exclusivity due to patent expiration, leading to widespread generic availability. Key generic manufacturers include Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Hikma Pharmaceuticals, Apotex Inc., and Aurobindo Pharma. The competitive landscape is characterized by price-based competition among generic producers.
Table 2: Major Furosemide Manufacturers and Their Market Presence
| Manufacturer |
Primary Region of Operation |
Notable Product Offerings |
| Sanofi |
Global |
Lasix (brand-name) |
| Teva Pharmaceutical Industries |
Global |
Generic furosemide tablets, injectables |
| Viatris (Mylan) |
Global |
Generic furosemide tablets, injectables |
| Hikma Pharmaceuticals |
MENA, Europe, US |
Generic furosemide tablets, injectables |
| Apotex Inc. |
Global |
Generic furosemide tablets, injectables |
| Aurobindo Pharma |
Global |
Generic furosemide tablets, injectables |
Source: Company reports and industry publications.
The dominance of generics means that pricing power is limited. Manufacturers compete on production efficiency, supply chain management, and distribution networks to maintain market share. Any significant price fluctuations are more likely to stem from supply-side disruptions or shifts in payer formularies rather than from innovative product differentiation.
What are the Primary Drivers of Demand for Furosemide?
The primary drivers for furosemide demand are the high and increasing incidence of cardiovascular diseases, chronic kidney disease (CKD), and liver cirrhosis globally.
- Heart Failure (HF): Congestive heart failure is a leading cause of hospitalization worldwide. Furosemide is a cornerstone therapy for managing fluid overload in HF patients, alleviating symptoms like dyspnea and edema. The World Health Organization (WHO) projects that cardiovascular diseases will remain the leading cause of death globally, directly correlating with sustained demand for loop diuretics.
- Chronic Kidney Disease (CKD): The prevalence of CKD is rising due to factors such as diabetes and hypertension. Furosemide is used to manage fluid balance in CKD patients who develop edema and may require renal replacement therapy.
- Liver Cirrhosis: Ascites (fluid accumulation in the abdomen) is a common complication of advanced liver disease. Furosemide, often in combination with spironolactone, is a standard treatment to reduce ascites and improve patient comfort and outcomes.
- Hypertension: While not a first-line agent for uncomplicated hypertension, furosemide can be used in certain hypertensive patients, particularly those with concomitant fluid retention or kidney disease.
Figure 1: Global Prevalence of Key Furosemide Indications (Estimated)