Share This Page
Drug Price Trends for LANSOPRAZOLE DR
✉ Email this page to a colleague
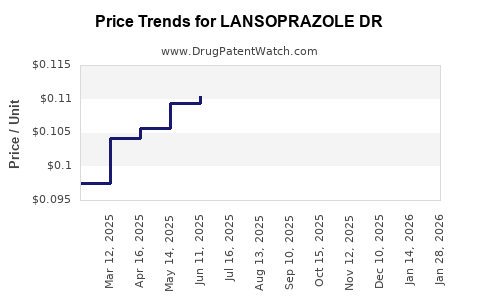
Average Pharmacy Cost for LANSOPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LANSOPRAZOLE DR 15 MG CAPSULE | 00536-1368-13 | 0.43232 | EACH | 2026-03-18 |
| LANSOPRAZOLE DR 15 MG CAPSULE | 00378-8015-93 | 0.16921 | EACH | 2026-03-18 |
| LANSOPRAZOLE DR 30 MG ODT | 72603-0314-10 | 1.94239 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
LANSOPRAZOLE DR Market Analysis and Financial Projection
What is Lansoprazole DR?
Lansoprazole delayed-release (DR) is a proton pump inhibitor (PPI) used primarily for treating gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. It reduces gastric acid secretion by blocking the H+/K+ ATPase enzyme system in gastric parietal cells. The drug's formulation as a delayed-release (DR) capsule enhances bioavailability and pharmacokinetic stability compared to immediate-release formulations.
What is the Current Market Size for Lansoprazole DR?
The global PPI market, valued at approximately $15.5 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of around 4.2% through 2030[1]. Lansoprazole retains a significant share within this segment, fueled by its long-standing approval status, familiarity, and lower cost in comparison with newer agents like vonoprazan.
Regionally, the emphasis varies:
- North America accounted for roughly 40% of the market in 2022, driven by high prevalence of GERD and a mature healthcare infrastructure[2].
- Europe represents approximately 25%, with a steady growth rate.
- Asia-Pacific, with considerable population size, witnesses rapid expansion (CAGR ~6%) owing to increased healthcare access and rising GERD incidence[3].
Estimates suggest global sales for Lansoprazole DR alone approached $2.2 billion in 2022, predominantly through generic manufacturers.
Who Are the Key Patent and Regulatory Players?
Lansoprazole was first approved by the FDA in 1995. Patent protections have largely expired, enabling multiple generic manufacturers. While the original patents have lapsed, certain formulation patents for specific delayed-release technologies or combination uses may still afford market exclusivity in limited regions until approximately 2025-2030.
Major producers include:
- Takeda Pharmaceuticals (original patent holder)
- Teva Pharmaceutical Industries
- Mylan (now part of Viatris)
- Sun Pharmaceutical
- Cipla
Regulatory pathways for generics often require demonstrating bioequivalence to the reference product, with some markets requiring additional clinical data for extended indications.
How Is the Pricing and Reimbursement Landscape Shaping?
Generic competition has driven prices downward. In the U.S., the average retail price for a 30-day supply of Lansoprazole DR ranges from $10 to $40, depending on dosage and pharmacy discounts. Private insurance and Medicaid coverage further influence net costs.
In Europe, reimbursement policies and tendering processes regulate prices, resulting typically in lower retail costs, especially in countries with centralized health systems.
Emerging biosimilar or branded formulations with improved pharmacokinetic profiles could alter pricing. However, since lansoprazole is off-patent, new entrants tend to engage in price competition rather than patent-based exclusivity.
What Are the Price Projections for Lansoprazole DR?
Given current trends:
-
Short-term (2023-2025): Prices are expected to stabilize with slight reductions due to intensified generic competition and price negotiations. The average retail price in the U.S. may decline to approximately $8–$12 for a 30-day supply.
-
Mid-term (2025-2030): Prices could decrease further by 10-15%, reaching around $5–$8 per 30-day supply, especially as procurement strategies favor low-cost generics.
-
Long-term: As patents in certain jurisdictions expire, manufacturers may seek to introduce lower-cost formulations or combination drugs, further pressing prices downward.
Pricing models suggest the global market value for Lansoprazole DR could decline by up to 40% over the next five years, contingent upon market penetration, regional reimbursement policies, and potential shifts toward newer PPIs like (vonoprazan) or combination therapies.
What Are the Key Drivers and Barriers?
Drivers:
- Rising prevalence of GERD and related conditions.
- Cost-containment pressures in healthcare systems.
- Preference for oral, low-cost, generic medications.
Barriers:
- Patent protections in certain markets until 2025-2030.
- Competition from other PPIs and novel acid suppressants.
- Patent litigation or formulation exclusivity claims.
Market Entry and Future Opportunities
Emerging markets and hospitals seeking cost reductions present growth opportunities for generic Lansoprazole DR producers. Partnerships with healthcare providers for formulary inclusion are likely to expand distribution.
Innovation in formulations—such as IV versions or combination drugs—may open higher-margin segments but are less immediate due to patent and regulatory hurdles.
Key Takeaways
- The global market for Lansoprazole DR was valued at around $2.2 billion in 2022.
- Price reductions of up to 40% are projected over the next five years due to intensified generic competition.
- Market growth is driven by increasing GERD prevalence and healthcare cost pressures.
- Patent expiration in key jurisdictions by 2025-2030 will influence pricing and market dynamics.
- Opportunities exist in emerging markets, hospitals, and through formulation innovations.
FAQs
Q1: When do the patents on Lansoprazole in major markets expire?
A: Patents for Lansoprazole vary by market; US patents expired in 2009, but some formulation or use patents could delay generic entry until around 2025-2030.
Q2: How does the cost of generic Lansoprazole compare with branded versions?
A: Generics are typically 60-80% cheaper than branded formulations, with prices declining further as competition intensifies.
Q3: What are the main factors influencing Lansoprazole market growth?
A: Increasing GERD prevalence, healthcare cost management, patent expiries, and regional market expansions.
Q4: Are there new formulations or alternatives that could impact Lansoprazole's market?
A: Yes; novel acid suppressants like vonoprazan offer alternative options but currently hold limited global market share compared to generic PPIs.
Q5: What regions offer the highest growth potential for Lansoprazole?
A: Asia-Pacific markets, due to demographic trends and expanding healthcare infrastructure, represent significant growth opportunities.
References
- MarketsandMarkets, "Proton Pump Inhibitors Market," 2022.
- IMS Health, "Global Gastrointestinal Drugs Market," 2022.
- WHO, "Global Burden of Gastroesophageal Reflux Disease," 2021.
More… ↓
