Share This Page
Drug Price Trends for LANSOPRAZOL-AMOXICIL-CLARITHRO
✉ Email this page to a colleague
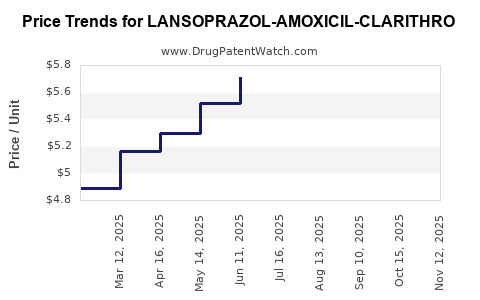
Average Pharmacy Cost for LANSOPRAZOL-AMOXICIL-CLARITHRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LANSOPRAZOL-AMOXICIL-CLARITHRO | 57237-0001-01 | 6.04223 | EACH | 2025-11-19 |
| LANSOPRAZOL-AMOXICIL-CLARITHRO | 57237-0001-14 | 6.04223 | EACH | 2025-11-19 |
| LANSOPRAZOL-AMOXICIL-CLARITHRO | 57237-0001-01 | 6.11684 | EACH | 2025-10-22 |
| LANSOPRAZOL-AMOXICIL-CLARITHRO | 57237-0001-14 | 6.11684 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for Lansoprazole–Amoxicillin–Clarithromycin combination
What is the current market landscape for this drug combination?
The combination of Lansoprazole, Amoxicillin, and Clarithromycin is predominantly used in the treatment of Helicobacter pylori (H. pylori) infections. This triple therapy is a standard first-line treatment in many regions but is not yet commonly marketed as a single fixed-dose combination (FDC). Instead, clinicians often prescribe the drugs separately or as two or three pills.
Therapeutic indications and regulatory status
The combination targets eradication of H. pylori, a major cause of peptic ulcers and gastritis. Regulatory approval varies; in some countries, single-pill formulations exist, while in others, the drugs are prescribed independently.
- The U.S. Food and Drug Administration (FDA) has approved various forms of Lansoprazole and individual drugs for H. pylori treatment.
- The European Medicines Agency (EMA) has approved some fixed-dose combinations but not universally.
Market size and demand drivers
The global H. pylori treatment market was valued at approximately $3–4 billion in 2022, with Proton Pump Inhibitors (PPIs) and antibiotics holding key segments. This market demonstrates steady growth due to rising H. pylori prevalence, particularly in Asia and developing nations, and increasing awareness of gastric disease management.
Key demand drivers:
- Growing global H. pylori prevalence, estimated at 44.3% according to the World Gastroenterology Organisation.
- Antibiotic resistance issues, leading to formulations with improved compliance.
- Regulatory approvals of fixed-dose combinations to improve adherence.
Competition and market shares
The market contains multiple approved therapies:
- Clarithromycin-based triple therapy with PPIs.
- Bismuth quadruple therapy.
- Sequential and concomitant therapies.
Few fixed-dose combinations of Lansoprazole–Amoxicillin–Clarithromycin exist, mainly in select markets like Japan and South Korea. The competition predominantly involves generic drugs, with branded combination products commanding premium prices.
Pricing trends and projections
Current prices for generic components are:
| Drug | Average Global Price per 14-day Course | Notes |
|---|---|---|
| Lansoprazole | Approx. $3–$7 | Widely available as generic |
| Amoxicillin | Approx. $2–$5 | Generic, low-cost |
| Clarithromycin | Approx. $4–$10 | Slightly higher due to market shortages in some areas |
Single-pill formulations fetch a premium, with prices around $20–$50 per course in developed markets.
Future price projections (2023–2028)
Price reductions for generic drugs are expected to continue at an annual rate of 5–10%, driven by increased competition and manufacturing efficiencies. Fixed-dose combination products are projected to maintain a price premium of 10–30% over the sum of generics, especially in markets emphasizing patient compliance.
Market entry of proprietary fixed-dose combinations may sustain higher prices temporarily, but patent expiration will likely pressure prices downward within five years.
Key factors influencing pricing
- Patent status: Patent expirations of components (2008 for Lansoprazole, 2014 for Clarithromycin) have increased generic competition.
- Manufacturing costs: Improvements and efficiencies reduce prices.
- Regulatory environment: Countries with streamlined approval may see faster market penetration and downward price pressure.
- Healthcare policies: Favor generic substitution and price control policies in most developed countries.
Conclusion
The Lansoprazole–Amoxicillin–Clarithromycin combination's market is constrained by patent expiry, with generics dominating current pricing. Fixed-dose combinations are limited but have potential to command higher prices, particularly if they improve patient compliance and treatment outcomes. Price reduction trends align with generic market dynamics, though branded combinations may sustain premium pricing temporarily.
Key Takeaways
- The global H. pylori treatment market exceeds $3 billion, with steady growth fueled by increased prevalence and resistance challenges.
- Fixed-dose combinations of Lansoprazole, Amoxicillin, and Clarithromycin are limited but supported by market demand for compliance.
- Generic prices for each component remain low, but fixed-dose products can price higher, especially in early market phases.
- Patent expirations and increased manufacturing capacity are driving prices downward; proprietary fixed-dose formulations may maintain higher margins temporarily.
- Market Entry Barriers include regulatory approvals and competition from existing therapies; opportunities exist where compliance advantages are significant.
FAQs
1. When are fixed-dose combinations of Lansoprazole–Amoxicillin–Clarithromycin expected to hit the market?
Currently, FDA and EMA approvals for fixed-dose combinations are limited. Some markets, like Japan, already have approved formulations, but global availability depends on regional regulatory processes, which typically take 2–4 years for new approvals.
2. How does antibiotic resistance influence pricing and market strategy?
Rising resistance to Clarithromycin has led to increased use of alternative regimens and combination therapies, impacting demand for traditional triple therapies. Fixed-dose combinations designed to improve adherence can maintain market share despite resistance issues.
3. What market segments are most likely to adopt fixed-dose formulations?
Markets with high H. pylori prevalence, such as Asia and Latin America, and healthcare systems emphasizing medication compliance are most receptive to fixed-dose products.
4. How do patent expirations impact future pricing?
Patent expirations increase generic competition, leading to price declines. Fixed-dose combinations may be patent-protected initially, allowing premium pricing, but eventual patent expiry will harmonize prices with generics.
5. What regulatory hurdles might delay market entry?
Approval delays can stem from need for clinical efficacy data, safety profiles, and regional regulatory requirements. Demonstrating bioequivalence and safety for fixed-dose formulations is critical for faster approval.
References
- World Gastroenterology Organisation. "H. pylori prevalence." 2022.
- MarketWatch. "H. pylori Treatment Market Size, Share & Trends." 2022.
- IQVIA. "Global Drug Price Reports," 2022.
- U.S. FDA. "Approved Drug Products," 2023.
- EMA. "European Medicines Agency approvals," 2023.
More… ↓
