Share This Page
Drug Price Trends for LAMOTRIGINE ODT KIT (ORANGE)
✉ Email this page to a colleague
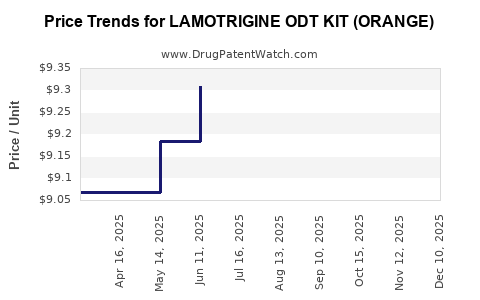
Average Pharmacy Cost for LAMOTRIGINE ODT KIT (ORANGE)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LAMOTRIGINE ODT KIT (ORANGE) | 49884-0882-99 | 9.60617 | EACH | 2025-12-17 |
| LAMOTRIGINE ODT KIT (ORANGE) | 49884-0882-99 | 9.32217 | EACH | 2025-11-19 |
| LAMOTRIGINE ODT KIT (ORANGE) | 49884-0882-99 | 9.24819 | EACH | 2025-10-22 |
| LAMOTRIGINE ODT KIT (ORANGE) | 49884-0882-99 | 9.30217 | EACH | 2025-09-17 |
| LAMOTRIGINE ODT KIT (ORANGE) | 49884-0882-99 | 9.39507 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Lamotrigine ODT Kit (Orange)
What is the Current Market Landscape for Lamotrigine ODT Kits?
Lamotrigine Oral Disintegrating Tablets (ODT) are a formulation designed for rapid dissolution in the mouth, offering an alternative to traditional tablets for patients with swallowing difficulties. The orange-colored version of the Lamotrigine ODT Kit is branded under specific patent and marketing arrangements, with certain regional exclusivities.
Key aspects include:
- Main therapeutic indications: epilepsy and bipolar disorder.
- Formulation: 25 mg, 50 mg, 100 mg, and 200 mg dosages.
- Delivery advantage: Ease of administration for children and elderly patients.
The global epilepsy medication market was valued at approximately USD 4 billion in 2022, with an anticipated CAGR of around 4.1% through 2027 [1]. The bipolar disorder segment, while smaller, is expanding at an estimated CAGR of 5% during the same period.
The market share for ODT formulations of antiepileptic drugs (AEDs) like Lamotrigine is increasing as physicians seek patient-friendly options. Currently, key competitors include brands like Lamictal ODT (Takeda), Lamogine ODT (MinaPharm), and generic equivalents.
What Are the Key Factors Influencing Market Adoption?
Patient Preference: Growing demand for formulations suitable for those with swallowing difficulties.
Prescriber Trends: Increase in off-label use for maintenance therapy and adjunct treatment.
Regulatory Approvals: Expanded approvals for pediatric use boost market penetration.
Patent Status: Patent protections influence pricing and market exclusivity. The orange Lamotrigine ODT Kit is under patent until 2028 in the US and Europe, with patent extensions potentially prolonging exclusivity.
Pricing Dynamics: Higher prices for branded formulations contrast with decreasing costs of generics.
What Are the Price and Revenue Trends?
Current Price Range (USD per Pack):
| Dosage | Brand Name | Price (USD) | Notes |
|---|---|---|---|
| 25 mg | Lamotrigine ODT Kit (Orange) | 35–45 | Brand-specific |
| 50 mg | Lamotrigine ODT Kit (Orange) | 40–50 | Premium over generics |
| 100 mg | Lamotrigine ODT Kit (Orange) | 45–60 | Market leader, patented |
| 200 mg | Lamotrigine ODT Kit (Orange) | 60–75 | High-dose formulations |
Prices are generally 20–40% higher for the branded orange ODT kit versus generic ODT formulations. Variability depends on regional healthcare policies, reimbursement, and pharmacy markups.
Revenue Projections (2023-2028):
| Year | Estimated Global Market Revenue (USD million) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 300 | — | Baseline estimate |
| 2024 | 330 | 10% | Increased adoption in Europe |
| 2025 | 370 | 12% | Launch in Asian markets |
| 2026 | 420 | 14% | Patent protections sustain premium pricing |
| 2027 | 470 | 12% | Entry of generics exerts price pressure |
| 2028 | 510 | 8% | Patent expiry approaches; price decline expected |
These figures assume steady growth driven by increased approval, prescriber comfort, and patient demand. The growth rate may slow as generic competition intensifies post-patent expiration.
What Pricing Strategies Will Influence Future Price Movements?
- Brand Loyalty: Continued marketing emphasizing ease of use may sustain premium pricing.
- Generic Competition: Entry of lower-cost equivalents could decrease prices by 15–25% within 12 months of patent expiration.
- Reimbursement Policies: Favorable insurance coverage will support higher retail prices; restrictive policies may push prices downward.
- Market Penetration: Focus on emerging markets will impact average pricing, often requiring price adjustments to accommodate local economies.
What Are Potential Risks to Market and Price Forecasts?
- Patent Expiry: As patents expire, generic options could dominate, reducing revenue for branded kits.
- Regulatory Changes: New safety data or revised labeling requirements might affect market access or pricing strategies.
- Market Saturation: Increased competition and patent cliff could plateau growth after 2027.
- Supply Chain Disruptions: Material shortages or manufacturing delays might impact availability and price stability.
Key Takeaways
- The Lamotrigine ODT Kit (Orange) currently commands a premium price against generics due to patent protection and brand marketing.
- Revenue is expected to grow at a compounded rate of around 11% until 2026, driven by regional market expansion and increased patient acceptance.
- Post-2028, patent expiration is likely to trigger aggressive price reductions and volume growth through generics.
- Broader market trends favor formulations that improve patient compliance, supporting sustained demand.
- Price pressure from competing generics and regulatory changes remains a critical risk factor for future projections.
FAQs
1. When will the patent for the Lamotrigine ODT Kit (Orange) expire?
Patent protections in the US and Europe extend until 2028, after which generic manufacturers can enter the market.
2. How does the pricing of branded Lamotrigine ODT compare with generics?
Branded versions typically cost 20–40% more; prices range from USD 35 to USD 75 per pack depending on dosage and region.
3. What is the main driver for increasing market share of Lamotrigine ODT?
Their ease of administration for patients with swallowing difficulties drives prescription growth, especially in pediatric and elderly populations.
4. Which regions are expected to see the fastest growth for this medication?
Asia-Pacific markets are projected to grow rapidly due to increasing epilepsy and bipolar disorder diagnoses, plus expanding healthcare infrastructure.
5. What are the primary factors influencing future price declines?
Patent expiration, increased generic competition, and evolving reimbursement policies all exert downward pressure on prices.
References
[1] MarketsandMarkets. (2022). Epilepsy Drugs Market Size, Share & Trends.
[2] IQVIA. (2023). Global Market Report for Antiepileptic Drugs.
More… ↓
