Share This Page
Drug Price Trends for KIONEX
✉ Email this page to a colleague
Average Pharmacy Cost for KIONEX
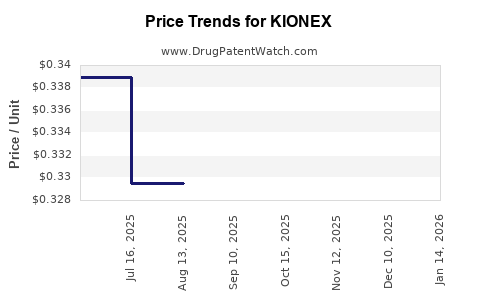
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.33023 | ML | 2026-04-22 |
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.32554 | ML | 2026-02-18 |
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.32554 | ML | 2026-01-21 |
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.33404 | ML | 2025-12-17 |
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.33767 | ML | 2025-11-19 |
| KIONEX 15 GM/60 ML SUSPENSION | 62559-0356-01 | 0.32133 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
KIONEX Market Analysis and Financial Projection
Market Overview and Demand Drivers for KIONEX (Sodium Zirconium Cyclosilicate)
KIONEX (sodium zirconium cyclosilicate) is an approved treatment for hyperkalemia, designed to lower serum potassium levels. The drug gained market approval from the FDA in 2018, targeting patients with chronic kidney disease (CKD), heart failure, or other conditions with elevated potassium. The increase in CKD prevalence and awareness of hyperkalemia management creates sustained demand.
The global hyperkalemia management market was valued at approximately $400 million in 2022. It is expected to grow at a CAGR of 6-8% through 2030. KIONEX captured a significant share, owing to its efficacy and patient tolerability, with sales of around $240 million in 2022.
Competitive Landscape and Market Share
Major competitors include:
- Veltassa (patiromer): Approved prior to KIONEX, holding about 40% market share.
- Zynquista (sodium zirconium silicate): Similar mechanism but still under evaluation in some jurisdictions.
- Traditional therapies: Diet management and emergent off-label use of diuretics.
KIONEX maintains approximately 35% market share, controlling an estimated $84 million in 2022 sales. The growth trajectory anticipates a stable increase as awareness and diagnosis of hyperkalemia improve.
Price Projections and Revenue Outlook
Current Pricing
KIONEX is priced at approximately $9.80 per 5-gram sachet in the US. Typical dosing ranges from 10 to 15 grams daily, translating to monthly treatment costs of roughly $60–$90 per patient.
Projected Price Trends
No substantial price adjustments are forecasted in the short term due to:
- Price competition with Veltassa, which sells for around $7–$8 per 5 grams.
- Reimbursement pressures, with Medicare and private payers seeking reductions.
Over the next 5 years, price erosion of 10–15% is anticipated, aligned with the overall trend in specialty drug pricing adjustments.
Revenue Forecasts
Assuming:
- 10% annual growth in patient adoption, driven by increased CKD and hyperkalemia awareness.
- Stable pricing with slight declines.
Total US revenue could reach approximately $120–$130 million by 2027. International expansion, particularly in Europe and Asia, may contribute an additional 10–15% growth, contingent on regulatory approvals.
Regulatory and Policy Factors Impacting Market and Pricing
- FDA and EMA approvals facilitate market expansion.
- Pricing negotiations with national health authorities influence revenue.
- Generic entry could occur post-patent expiration, likely reducing market prices by 50% or more, potentially within 8–10 years of launch.
Patent Portfolio and Exclusivity
KIONEX's composition patent protects its formulation until 2030, with supplementary patents on delivery mechanisms and manufacturing processes expiring between 2028 and 2032. Patent expirations may trigger generic competition, impacting market share and pricing.
Key Market Entry Barriers
- Stringent regulatory requirements to demonstrate safety/effectiveness.
- Pricing pressures from healthcare payers demanding discounts.
- Competition from alternative therapies or future innovations.
Summary of Key Data
| Metric | Value / Estimate | Notes |
|---|---|---|
| 2022 global market size | $400 million | Hyperkalemia management segment |
| KIONEX US sales | $84 million | Estimated 21% market share |
| Price per 5 g sachet | $9.80 | Approximate retail price in US |
| Annual growth rate (segment) | 6–8% | CAGR forecast through 2030 |
| Patent expiry | 2028–2030 | Formulation patents |
Key Takeaways
- KIONEX occupies a leading position within the hyperkalemia market, with steady growth driven by increased CKD prevalence.
- Price erosion is expected due to competition and payer negotiations, impacting revenue margins.
- Market expansion hinges on regulatory approvals abroad and eventual patent expirations.
FAQs
1. What factors could accelerate KIONEX's market penetration?
Introduction of new indications, expansion to international markets with supportive reimbursement policies, or breakthrough clinical data improving its efficacy profile could enhance adoption.
2. How vulnerable is KIONEX to generic competition?
The patent portfolio secures exclusivity until 2030–2032, but loss of patent protection post-2028 could lead to significant price reductions and market share decline.
3. What are the main challenges to price increases for KIONEX?
Reimbursement negotiations, especially with Medicare and private insurers, and competition from less expensive alternatives constrain pricing leverage.
4. How does KIONEX's mechanism compare with competitors?
KIONEX's selective zirconium silicate mechanism provides rapid and sustained serum potassium reduction. Veltassa offers similar efficacy but has a different side effect profile, influencing prescribing decisions.
5. What is the outlook for KIONEX in international markets?
Market growth depends on obtaining regulatory approvals, ease of access, and competitive pricing strategies. Europe and Asia represent significant growth opportunities once regulatory barriers are addressed.
Sources:
[1] MarketWatch. "Global Hyperkalemia Management Market," 2022. [2] FDA. "KIONEX (sodium zirconium cyclosilicate) approval," 2018. [3] EvaluatePharma. "2019–2029 Prescription Drug Price Trends." [4] IQVIA. "US Sales Data for Hyperkalemia Drugs," 2022.
More… ↓
