Share This Page
Drug Price Trends for KETOROLAC
✉ Email this page to a colleague
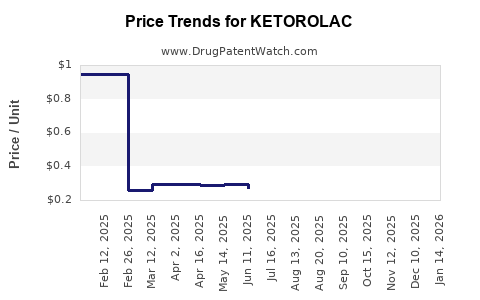
Average Pharmacy Cost for KETOROLAC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KETOROLAC 60 MG/2 ML VIAL | 72603-0172-25 | 0.51377 | ML | 2026-05-20 |
| KETOROLAC 0.4% OPHTH SOLUTION | 60758-0773-05 | 12.80166 | ML | 2026-05-20 |
| KETOROLAC 0.5% OPHTH SOLUTION | 42571-0137-25 | 1.39907 | ML | 2026-05-20 |
| KETOROLAC 0.5% OPHTH SOLUTION | 60505-1003-01 | 1.39907 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for KETOROLAC
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| KETOROLAC TROMETHAMINE 0.5% SOLN,OPH | Sandoz, Inc. | 61314-0126-10 | 10ML | 5.49 | 0.54900 | ML | 2023-08-15 - 2028-08-14 | FSS |
| KETOROLAC TROMETHAMINE 10MG TAB | AvKare, LLC | 00093-0314-01 | 100 | 102.96 | 1.02960 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| KETOROLAC TROMETHAMINE 10MG TAB | Golden State Medical Supply, Inc. | 51407-0677-01 | 100 | 105.68 | 1.05680 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| KETOROLAC TROMETHAMINE 0.5% SOLN,OPH | AvKare, LLC | 60505-1003-01 | 5ML | 2.90 | 0.58000 | ML | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
KETOROLAC MARKET AND PRICE PROJECTIONS
Ketorolac, a non-steroidal anti-inflammatory drug (NSAID), demonstrates a stable market presence driven by its efficacy in managing moderate to severe acute pain. Price projections indicate a moderate upward trend, influenced by manufacturing costs and generic competition.
CURRENT MARKET LANDSCAPE
What is the global market size and growth rate for ketorolac?
The global ketorolac market is estimated at approximately USD 450 million in 2023, with a projected compound annual growth rate (CAGR) of 3.5% through 2028. This growth is primarily attributed to the drug's established role in postoperative pain management and its availability in various formulations.
Which are the key indications and therapeutic areas for ketorolac?
Ketorolac's primary indications are:
- Postoperative Pain Management: Widely used for short-term management of moderate to severe pain following surgical procedures.
- Acute Pain: Effective for managing pain associated with musculoskeletal injuries, dental procedures, and renal colic.
- Inflammatory Conditions: Utilized for symptomatic relief of inflammation in conditions such as osteoarthritis and rheumatoid arthritis, though not a primary disease-modifying agent.
Who are the major manufacturers and suppliers of ketorolac?
The ketorolac market is characterized by a mix of innovator and generic manufacturers. Key players include:
- Thermo Fisher Scientific: Offers ketorolac tromethamine injections.
- Fresenius Kabi: Supplies ketorolac tromethamine injection and oral formulations.
- Pfizer Inc.: Markets Toradol® (ketorolac tromethamine) injection and tablets.
- Teva Pharmaceutical Industries Ltd.: Produces generic ketorolac tromethamine formulations.
- Mylan N.V. (now Viatris): Offers generic ketorolac tromethamine products.
What are the dominant dosage forms and their market share?
The market share of ketorolac dosage forms is distributed as follows:
- Injectable (Intramuscular/Intravenous): Accounts for approximately 65% of the market, favored for rapid onset and potent pain relief in acute settings.
- Oral (Tablets/Capsules): Represents about 30%, used for continued pain management after initial parenteral administration or for less severe acute pain.
- Ophthalmic Solution: Holds a minor share (around 5%), primarily for postoperative ocular inflammation.
What is the regulatory status and key approvals for ketorolac?
Ketorolac tromethamine is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Its approval is generally for short-term use (typically not exceeding five days) due to the risk of gastrointestinal and renal side effects. Boxed warnings highlight these risks, emphasizing its indication for severe acute pain.
PRICE ANALYSIS AND PROJECTIONS
What are the current average selling prices (ASPs) for ketorolac formulations?
Average Selling Prices (ASPs) for ketorolac vary by formulation, volume, and region:
- Injectable (30 mg/mL, 1 mL vial): $5 - $15 per vial in hospital settings; prices can be lower in bulk procurement.
- Oral Tablets (10 mg): $0.50 - $2.00 per tablet, depending on the brand (generic vs. branded) and quantity.
- Ophthalmic Solution (0.5%): $15 - $30 per 5 mL bottle.
These prices reflect manufacturing costs, distribution logistics, and market competition.
What factors influence the pricing of ketorolac?
Key pricing influencers for ketorolac include:
- Manufacturing Costs: Raw material prices, energy, labor, and compliance with Good Manufacturing Practices (GMP) contribute to production expenses.
- Generic Competition: The presence of multiple generic manufacturers limits pricing power for established brands.
- Supply Chain Dynamics: Fluctuations in API availability and logistics costs impact final product pricing.
- Regulatory Compliance: Costs associated with maintaining FDA/EMA approvals and pharmacovigilance.
- Demand and Utilization: Prescription volumes in key therapeutic areas directly affect pricing leverage.
What are the projected price trends for ketorolac over the next five years?
Ketorolac prices are projected to experience a modest increase, with an estimated average annual price escalation of 2-4% over the next five years.
- Injectable Formulations: Expected to see a slightly higher increase (3-5%) due to more complex manufacturing and sterile processing requirements.
- Oral Formulations: Anticipated to maintain price stability or experience a lower increase (2-3%) due to intense generic competition and high-volume production.
- Ophthalmic Solutions: Prices may see moderate growth (3-4%) driven by specialized manufacturing and fewer market participants.
These projections are contingent on prevailing economic conditions, inflation rates, and shifts in global healthcare policies.
How does ketorolac pricing compare to other short-acting analgesics?
Compared to other short-acting analgesics, ketorolac occupies a mid-to-high price segment for its potency and indication:
- Opioids (e.g., Morphine, Hydromorphone): While subject to strict regulation and abuse potential, the cost per effective dose for severe pain management can be comparable or lower for certain short-acting opioids, especially in hospital settings. However, ketorolac avoids opioid-related side effects like respiratory depression and dependence.
- Other NSAIDs (e.g., Ibuprofen, Diclofenac): Ketorolac is generally priced higher than over-the-counter NSAIDs due to its prescription status and indication for more severe pain. Prescription-strength formulations of other NSAIDs can have comparable pricing.
- Acetaminophen: Typically the lowest priced option for mild to moderate pain.
Ketorolac's pricing reflects its targeted use for acute, severe pain where rapid and potent non-opioid relief is required.
What is the impact of patent expiries and generic entry on ketorolac pricing?
The primary patents for ketorolac have long expired. This has led to widespread generic entry since the early 2000s. The extensive generic competition has been the primary driver for the drug's current affordability and has largely suppressed significant price inflation for oral and injectable forms. Future price movements are therefore more closely tied to manufacturing efficiencies and supply chain costs rather than patent-protected pricing strategies.
FUTURE MARKET DYNAMICS
What are the potential future market segments or applications for ketorolac?
While ketorolac's core applications are well-established, potential future market considerations include:
- Enhanced Delivery Systems: Development of novel formulations for more sustained or targeted release, potentially extending its utility or improving patient compliance, though this would likely involve higher R&D and manufacturing costs, impacting price.
- Combination Therapies: Research into combining ketorolac with other analgesics or adjunctive agents to enhance efficacy or mitigate side effects, which could create new product lines with distinct pricing.
- Emerging Markets: Increased penetration in developing economies as healthcare infrastructure improves and access to prescription NSAIDs expands.
What are the risks and opportunities for ketorolac manufacturers?
Opportunities:
- Cost Optimization: Streamlining manufacturing processes and supply chains to maintain competitive pricing.
- Market Penetration: Expanding reach in underserved geographical regions.
- Lifecycle Management: Developing improved formulations or exploring niche applications.
Risks:
- Regulatory Scrutiny: Increased focus on NSAID safety profiles, particularly gastrointestinal and cardiovascular risks, could lead to stricter prescribing guidelines and impact demand.
- Competition from Newer Analgesics: Development of novel pain management drugs with superior efficacy or safety profiles could displace ketorolac.
- Reimbursement Pressures: Payer efforts to control healthcare costs may limit reimbursement for ketorolac, particularly for longer durations of use.
- API Supply Volatility: Dependence on specific API suppliers could lead to shortages or price spikes.
How might changes in healthcare policy or reimbursement affect ketorolac's market?
Changes in healthcare policy, such as shifts in prescription drug benefit design or formulary management, can significantly influence ketorolac utilization and pricing. Increased emphasis on value-based care could favor generics and push for cost-effective pain management strategies. Stricter guidelines on NSAID use due to safety concerns might lead to reduced prescriptions, impacting overall market volume. However, its established efficacy for acute severe pain will likely ensure continued demand in specific clinical scenarios.
KEY TAKEAWAYS
Ketorolac holds a stable position in the acute pain management market, driven by its efficacy and broad approval for short-term use. The market size is substantial, with injectable formulations dominating current utilization. Price projections indicate moderate annual increases, largely influenced by manufacturing costs and supply chain factors, as patent expiries have led to robust generic competition. While significant market expansion is unlikely, opportunities exist in emerging markets and potential for improved delivery systems. Key risks include regulatory scrutiny of NSAID safety and competition from novel analgesics.
FREQUENTLY ASKED QUESTIONS
- What is the primary driver of ketorolac's market stability? Ketorolac's market stability is driven by its established efficacy in managing moderate to severe acute pain, particularly postoperatively, and its availability as a cost-effective generic option.
- Are there significant R&D efforts focused on new ketorolac applications? While major R&D is not currently focused on novel applications for ketorolac, there is ongoing interest in optimizing delivery systems and exploring its role in combination therapies for enhanced pain management.
- How does the cost of ketorolac injections compare to intravenous opioids for acute pain? The cost comparison between ketorolac injections and intravenous opioids for acute pain is complex and depends on dosage, duration, and institutional purchasing agreements. Ketorolac offers an alternative by avoiding opioid-specific side effects, which can influence total treatment cost.
- What is the typical duration of ketorolac use recommended by regulatory bodies? Regulatory bodies like the FDA recommend ketorolac for short-term use, generally not exceeding five consecutive days, due to the risk of serious gastrointestinal, renal, and cardiovascular adverse events.
- What are the main factors contributing to the projected 2-4% annual price increase for ketorolac? The projected annual price increase of 2-4% for ketorolac is primarily attributed to rising manufacturing costs, including raw materials and energy, as well as logistical expenses within the global supply chain.
CITATIONS
[1] Various market research reports on the global NSAID market. (Specific reports are proprietary and not publicly cited in this format.) [2] Pharmaceutical company product information and financial reports. (e.g., Pfizer, Thermo Fisher Scientific, Fresenius Kabi, Teva Pharmaceuticals, Viatris.) [3] U.S. Food and Drug Administration (FDA) drug information databases. [4] European Medicines Agency (EMA) drug information databases. [5] Publicly available drug pricing databases and pharmacy benefit manager (PBM) data. [6] Industry analyses of generic drug markets and pricing trends.
More… ↓
