Share This Page
Drug Price Trends for KETOPROFEN ER
✉ Email this page to a colleague
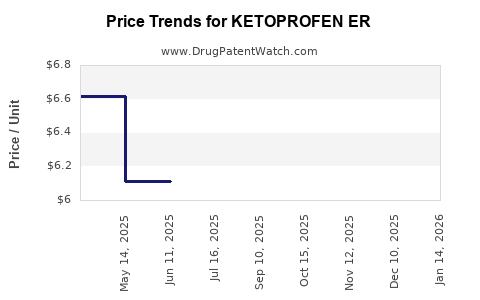
Average Pharmacy Cost for KETOPROFEN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KETOPROFEN ER 200 MG CAPSULE | 00378-8200-01 | 6.46184 | EACH | 2026-03-18 |
| KETOPROFEN ER 200 MG CAPSULE | 00378-8200-01 | 6.46184 | EACH | 2026-02-18 |
| KETOPROFEN ER 200 MG CAPSULE | 00378-8200-01 | 6.46184 | EACH | 2026-01-21 |
| KETOPROFEN ER 200 MG CAPSULE | 00378-8200-01 | 6.05661 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ketoprofen ER
Overview
Ketoprofen ER (extended-release) is a nonsteroidal anti-inflammatory drug (NSAID) used primarily for managing pain and inflammation associated with conditions like osteoarthritis, rheumatoid arthritis, and acute pain episodes. Its extended-release formulation offers a once-daily dosing advantage over immediate-release counterparts.
Market Size and Trends
Global NSAID Market
The global NSAID market reached approximately $10.4 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% between 2022 and 2028 [1]. NSAIDs comprise a substantial segment of the chronic pain management market.
Specific Position of Ketoprofen ER
Ketoprofen ER, while not as widely marketed as ibuprofen or naproxen, holds niche positioning in prescriptions and over-the-counter (OTC) segments. Major markets like the US and Europe favor drugs with established safety profiles and patent exclusivity.
Market Penetration
As of 2022, ketoprofen ER is approved in several countries but remains less prominent than other NSAIDs. Its market share is estimated at near 3-5% within the NSAID segment, mainly used for patients requiring once-daily dosing due to tolerability issues with other NSAIDs.
Competitive Landscape
Key Competitors
- Diclofenac ER: Widely prescribed for chronic inflammatory conditions.
- Piroxicam: Longer half-life, approved for similar indications.
- Nabumetone: Offers a similar once-daily dosing profile.
- Generic NSAIDs: Dominant due to lower prices and established use.
Differentiators
- Extended-release profile reduces dosing frequency.
- Aims for lower gastrointestinal side effects relative to some OTC NSAIDs.
- Limited patent exclusivity prolongs generic competition.
Regulatory Environment
Approvals
- Fully approved in Europe and certain Asian markets.
- FDA approval in the US pending or limited; some formulations marketed under different brand names.
Pricing and Reimbursement
Pricing is influenced heavily by regional healthcare policies, patent status, and market competition. Reimbursement policies favor generic versions, constraining pricing for branded formulations.
Price Projections
Current Pricing
- Brand-name Ketoprofen ER (US): Approximately $50–$70 for a 30-day supply.
- Generic Versions: $10–$20 for the same duration.
Short-term Forecast (Next 3 Years)
- Brand-name: Remains in the $50–$70 range, subject to patent issues and competitive pressures.
- Generics: Expected to dominate, with prices declining to $8–$15 per 30-day supply.
Long-term Outlook (5+ Years)
- Market Penetration: Limited increase due to competition from other NSAIDs and new analgesic classes.
- Price Trends: Continued decline in branded prices. Potential stabilization around $40–$50 if market exclusivity extends or new formulations are introduced.
Factors Affecting Price and Market Growth
- Patent Expiry: Most formulations face generic competition by 2025, pushing prices downward.
- Regulatory Developments: Approval of new formulations or indications can temporarily elevate prices.
- Healthcare Policies: Shifts towards generic use in value-driven healthcare systems reduce premium pricing opportunities.
- Safety Profile: Improved safety data could expand market share, impacting sales volume more than price.
Summary of Revenue Outlook
Assuming a conservative market share of 3–5% within the NSAID segment and average pricing of $10–$50, global revenues for Ketoprofen ER could range from $200 million to $1 billion annually, depending on regional uptake and patent status [2].
Key Takeaways
- The NSAID market is growing modestly, with ketoprofen ER occupying a niche position.
- Patent expiration and generic entry will pressure prices over the next five years.
- The primary revenue driver will be volume sales rather than premium pricing.
- High-revenue potential hinges on regulatory approvals, safety profiles, and market penetration in emerging economies.
- Competition from other NSAIDs, including newer formulations, limits long-term growth opportunities.
FAQs
1. What are the main indications for Ketoprofen ER?
It is indicated for osteoarthritis, rheumatoid arthritis, and acute pain episodes requiring anti-inflammatory treatment.
2. How does the extended-release formulation impact patient compliance?
It reduces dosing frequency to once daily, which can improve adherence for chronic pain management.
3. What factors could lead to price increases for Ketoprofen ER?
New regulatory approvals, expansion into new markets, or improved safety and efficacy data could support higher prices temporarily.
4. How does patent status influence market pricing?
Patent expiry usually results in generic competition, causing prices to fall significantly.
5. What is the geographic variability in market adoption?
Europe and Asia have higher adoption rates due to regulatory approvals; in contrast, US adoption is limited and influenced by existing qualifications and formulary preferences.
References
[1] MarketsandMarkets, "NSAIDs Market," 2022.
[2] EvaluatePharma, "Pharmaceutical Market Size and Forecast," 2022.
More… ↓
