Share This Page
Drug Price Trends for KETODAN
✉ Email this page to a colleague
Average Pharmacy Cost for KETODAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
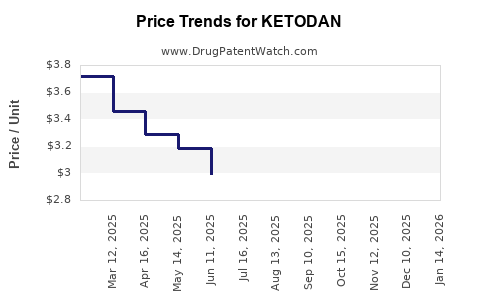
| KETODAN 2% FOAM | 43538-0530-10 | 2.78433 | GM | 2026-03-18 |
| KETODAN 2% FOAM | 43538-0530-10 | 2.53294 | GM | 2026-02-18 |
| KETODAN 2% FOAM | 43538-0530-10 | 2.33245 | GM | 2026-01-21 |
| KETODAN 2% FOAM | 43538-0530-10 | 2.35569 | GM | 2025-12-17 |
| KETODAN 2% FOAM | 43538-0530-10 | 2.37678 | GM | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ketodan
Overview
Ketodan (generic: ketoconazole) is an antifungal medication approved for treating specific dermatological conditions. Its brand version provides a topical formulation used in managing fungal skin infections. The market for topical antifungals, including Ketodan, is influenced by the prevalence of fungal skin conditions, competition from generics, regulatory changes, and evolving treatment guidelines. Pricing strategies and projections hinge on patent status, manufacturing costs, and market demand.
Market Size and Segmentation
Global Market Value
The topical antifungal market was valued at approximately USD 3.2 billion in 2022. It is projected to reach USD 4.1 billion by 2028, growing at a compound annual growth rate (CAGR) of 4.3% (2022-2028).
Key Drivers
- Increasing incidence of dermatophyte infections worldwide.
- Rising awareness and diagnosis of fungal skin conditions.
- Expanding aging population and immunocompromised patients.
Regional Breakdown
| Region | Market Share (2022) | Growth Rate (2022-2028) |
|---|---|---|
| North America | 40% | 4.0% |
| Europe | 25% | 4.5% |
| Asia-Pacific | 20% | 5.2% |
| Rest of World | 15% | 3.8% |
Competitive Landscape
- Generics Dominance: The patent exclusivity for Ketodan has lapsed in many jurisdictions, leading to increased generic competition.
- Major Players: Pfizer, Teva, Mylan, and Sandoz market their versions of ketoconazole topical formulations.
- Market Penetration: Branded versions capture premium pricing, but generics significantly erode margins and market share.
Pricing Dynamics
Current Pricing
- Brand (Ketodan): Estimated retail price per 30g tube ranges from USD 70 to USD 100 depending on region.
- Generic: Price drops to USD 20- USD 40 per 30g tube.
Pricing Factors
- Patent status: Patent expiration occurred globally between 2020-2022.
- Insurance coverage and reimbursement policies.
- Regulatory approval for multiple formulations influences price variations.
Price Projections (2023-2028)
| Year | Estimated Average Price (USD) | Notes |
|---|---|---|
| 2023 | USD 60 – USD 90 | Continued availability of branded with diminishing premium due to generics |
| 2024 | USD 55 – USD 85 | Entry of more generics; price erosion accelerates |
| 2025 | USD 50 – USD 80 | Margins shrink further; potential price stabilization in mature markets |
| 2026 | USD 45 – USD 75 | Market saturation and commoditization dominate |
| 2027 | USD 40 – USD 70 | Price pressures from increased biosimilar/generic proliferation |
| 2028 | USD 35 – USD 65 | Long-term steady decline in brand premiums; no new patent protections anticipated |
Regulatory and Policy Impact
- The expiration of patents in key markets, such as the United States in 2021, vaporizes significant exclusivity rights.
- Healthcare reimbursement policies favor cost-effective generics over brands.
- Patent litigation and data exclusivity periods in Asia and Europe influence market entry timing.
Additional Considerations
- Market acceptance of new formulations or combination products could elevate prices.
- New indications or delivery systems (e.g., foam, spray) could sustain premium pricing.
- Pricing pressure from regulatory authorities on drug prices could accelerate generic adoption.
Key Takeaways
- The global market for topical antifungals, including Ketodan, is expanding primarily due to rising fungal disease prevalence.
- The patent lapses have driven a rapid decline in branded product prices, with generics dominating the market.
- Price projections show a downward trend, with average retail prices expected to decrease by about 30% between 2023 and 2028.
- Market shares are primarily held by generic manufacturers, constraining the pricing power of brand-name Ketodan.
- Regulatory changes and reimbursement policies will significantly influence future pricing and market penetration.
FAQs
1. What factors most influence Ketodan's market price?
Patent status, competition from generics, regulatory policies, and insurance reimbursement are primary determinants.
2. How does patent expiration affect pricing?
Expiration opens the market to generics, leading to immediate price reductions for branded versions.
3. Are there upcoming formulations that could alter price projections?
Yes, formulations like foam or combination therapies could maintain higher prices if approved and adopted.
4. What regions present the highest growth opportunities?
Asia-Pacific and Latin America show higher growth rates due to rising infections and expanding healthcare access.
5. Will regulatory changes hinder future market growth?
Potentially, especially if governments enforce price controls or restrict reimbursement for high-cost treatments.
References
[1] MarketsandMarkets. "Topical Dermatological Drugs Market by Type, Route of Administration, Region - Global Forecast to 2028."
[2] IQVIA Reports. "Global Antifungal Market Analysis."
[3] FDA and EMA patent status disclosures for ketoconazole formulations.
More… ↓
