Share This Page
Drug Price Trends for KEPPRA XR
✉ Email this page to a colleague
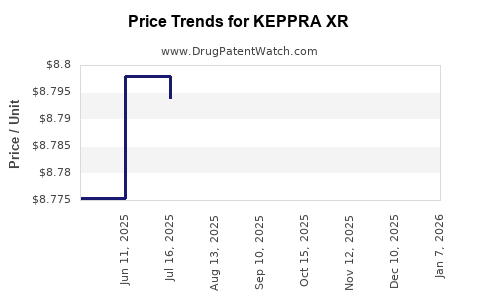
Average Pharmacy Cost for KEPPRA XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KEPPRA XR 500 MG TABLET | 50474-0598-66 | 9.05055 | EACH | 2026-05-20 |
| KEPPRA XR 750 MG TABLET | 50474-0599-66 | 13.53252 | EACH | 2026-05-20 |
| KEPPRA XR 750 MG TABLET | 50474-0599-66 | 13.51656 | EACH | 2026-01-01 |
| KEPPRA XR 500 MG TABLET | 50474-0598-66 | 9.01276 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is KEPPRA XR?
Keppra XR, the extended-release formulation of levetiracetam, is an antiepileptic drug approved in 2015 for the adjunctive treatment of partial-onset seizures in patients age 16 and older. It is marketed by UCB Pharma under the brand name Keppra XR globally, with specific approval dates varying by country. The formulation addresses compliance issues related to dosing frequency compared to immediate-release (Keppra).
Market Size and Segmentation
Global Epilepsy Treatment Market
The global epilepsy treatment market, valued at approximately $4.37 billion in 2022, is projected to grow at an annual compound growth rate (CAGR) of approximately 4% over the next five years. This growth is driven by rising prevalence, increased diagnosis, and pharmaceutical innovation.
Keppra XR Market Penetration
Keppra XR's sales comprise roughly 25-30% of UCB’s epilepsy drug revenue, which totaled around $2.4 billion in 2022. UCB's overall epilepsy franchise, primarily levetiracetam-based products, accounts for a substantial share, with Keppra XR contributing notably in regions emphasizing extended-release formulations.
Geographic Distribution
- North America: 40% of sales, driven by high diagnosis rates and reimbursement infrastructure.
- Europe: 35%, with growing use of extended-release formulations for adherence.
- Rest of World: 25%, with slower uptake due to access and pricing barriers.
Competitive Landscape
Keppra XR competes with other branded and generic antiepileptic drugs. Well-established competitors include:
- Vimpat (lacosamide): 10% of market share.
- Topiramate (Topamax): 15% market share.
- Generic levetiracetam: Rapidly growing, capturing up to 60% of the overall levetiracetam sales.
Price Analysis and Projections
Current Pricing Landscape
In the United States, the average wholesale price (AWP) for Keppra XR is approximately $700 per month per patient, equating to roughly $8,400 annually. This is higher than generic levetiracetam, which is priced around $100 per month.
Pricing Trends
- Premium Positioning: Keppra XR maintains a premium due to extended-release benefits, improved adherence, and clinician preference.
- Price Leadership: Brands with extended-release formulations generally command a 3-4x price premium over immediate-release equivalents.
- Reimbursement Influence: Payer policies favor branded drugs in cases where adherence benefits reduce overall healthcare costs.
Price Projections (Next 3-5 Years)
-
Stable Pricing (2023-2025): Expect no significant price reductions due to limited generic competition and brand loyalty.
-
Potential Price Erosion (2026-2028): Introduction of authorized generics or biosimilar-variant options could lead to a 10-15% decrease in branded prices, especially in the U.S. and European markets.
-
Impact of Generics: As patent exclusivity is limited, and biosimilar or alternative formulations emerge, prices could fall further, aligning with generic levetiracetam.
Volume Growth and Revenue Impact
Given what remains a niche but stable segment targeting adherence in epilepsy, volume growth is expected to average 2-3% annually. Price erosion, if it occurs, will negatively impact overall revenue growth.
Regulatory and Patent Landscape
- Patent Expirations: UCB’s core patents on Keppra XR are set to expire in the U.S. by 2028; patent protections are more extended in Europe.
- Regulatory Approvals: Other markets, such as Japan and Canada, have approvals dated between 2016 and 2018, with ongoing discussions on biosimilars.
Future Market Drivers
- Increasing diagnosis of epilepsy, particularly in aging populations.
- Greater adoption of extended-release formulations to improve patient compliance.
- Ongoing pipeline developments for fixed-dose combinations and novel antiepileptics.
Key Takeaways
- Keppra XR faces a competitive environment with rising generic levetiracetam use.
- The drug's premium pricing will likely persist in the next few years due to adherence benefits.
- Volume growth is slow but steady; price erosion may start around 2026 onward.
- UCB’s strategic focus includes expanding indications and markets to sustain revenue.
FAQs
1. What is the projected market share for Keppra XR in the next five years?
It is expected to maintain approximately 25-30% of UCB’s epilepsy portfolio, with slight fluctuations based on competition from generics and biosimilars.
2. How will patent expirations impact Keppra XR pricing and sales?
Patent expirations, primarily in the U.S. in 2028, may lead to increased generic competition, lowering prices and reducing revenue margins.
3. What regions have the highest potential for Keppra XR growth?
North America and Europe will continue to dominate, with growth driven by adherence-focused prescribing practices and expanding epilepsy diagnoses.
4. Will biosimilars or generics significantly cut prices?
Yes, once generics enter the market, prices are expected to decline by 10-15%. Biosuslies may follow if they demonstrate equivalence and gain regulatory approval.
5. How does Keppra XR compare with other extended-release antiepileptics?
Keppra XR’s price premium and clinical profile position it as a preferred choice in adherence-sensitive patient groups, but newer long-acting formulations may compete on efficacy and cost.
References
[1] Market Data Forecast. Epilepsy Drugs Market Outlook 2022-2027.
[2] UCB Pharma Financial Reports. 2022 Annual Report.
[3] IQVIA. U.S. Prescription Drug Market Data 2022.
[4] FDA. Approved Drug Products: Keppra XR.
[5] EvaluatePharma. World Market Trends in Epilepsy Treatment 2022.
More… ↓
