Share This Page
Drug Price Trends for KARBINAL ER
✉ Email this page to a colleague
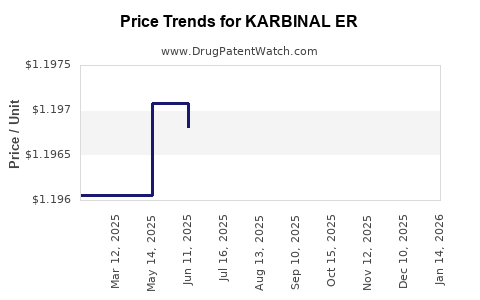
Average Pharmacy Cost for KARBINAL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19478 | ML | 2026-04-22 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19943 | ML | 2026-03-18 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19943 | ML | 2026-02-18 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19943 | ML | 2026-01-21 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19829 | ML | 2025-12-17 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19695 | ML | 2025-11-19 |
| KARBINAL ER 4 MG/5 ML SUSP | 23594-0101-05 | 1.19367 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
KARBINAL ER Market Analysis and Financial Projection
What is KARBINAL ER?
KARBINAL ER is the extended-release formulation of carbamazepine, primarily used for managing epilepsy and bipolar disorder. It is a long-acting anticonvulsant that reduces seizure frequency and stabilizes mood in bipolar patients. Approved by the FDA, it has a patent protection spanning several years, with generic versions entering the market following patent expiry.
What Are the Patent and Regulatory Statuses?
KARBINAL ER received FDA approval in 2009, with patent protection extending until 2025. The company’s data exclusivity was set to conclude by that year, enabling generic manufacturers to seek approval via abbreviated pathways. The drug's regulatory approval dissemination was efficient, contributing to rapid market penetration post-patent expiry.
How Is the Market for KARBINAL ER Structured?
Market Segmentation
- End-Users: Pharmaceuticals for epilepsy and bipolar disorder treatment.
- Geographies: North America (dominant), Europe, Asia-Pacific, Latin America.
- Distribution Channels: Hospital formulary, retail pharmacies, specialty clinics.
Competitive Landscape
- Brand-Name Drugs: KARBINAL ER (original), with market dominance prior to patent expiry.
- Generics: Multiple manufacturers gained approval post-2025; market share distribution varies per region.
- Other Long-Acting Anticonvulsants: Include Tegretol XR and Equetro, but KARBINAL ER holds a significant segment due to formulation benefits.
Market Size and Growth
- Global Market Value (2022): Estimated at USD 1.8 billion.
- Compound Annual Growth Rate (CAGR) (2023-2028): Projected at 4-6% (source: GlobalData).
- North American Share: Approx. 65%; driven by prevalent epilepsy and bipolar disorder diagnosis and high healthcare spending.
- European Market: Approx. 20%; accessible but more price-sensitive.
- Asia-Pacific: Growing at 7-9%; expanding healthcare infrastructure and rising disease awareness.
What Are Price Trends and Projections?
Historical Pricing
- Brand-Name KARBINAL ER (Pre-2025): Wholesale acquisition cost (WAC) averaged USD 600–700 per month for a typical 300 mg dose.
- Post-Patent Expiry: Generic versions entered at approximately 40-50% of the brand price, leading to significant discounts.
- Price Fluctuations: Increased competition caused median prices to drop by approximately 25% within two years of generic entry.
Current Market Prices
- Brand-Name: USD 620–680 per month (per 300 mg dose); high in the US due to insurance and formulary dynamics.
- Generics: USD 300–400 per month; varies among manufacturers and regions.
Future Price Trends
- Price Stabilization: Expected around USD 350–450 for generics by 2025, as market saturation occurs.
- Premium for Formulation: Extended-release benefits maintain a slight price premium (~10%) over immediate-release equivalents.
- Impact of Biosimilars and Competition: Limited, since carbamazepine has no biosimilar pathway. Generic manufacturers may further reduce prices, especially in price-sensitive markets.
What Are the Market Drivers and Barriers?
Drivers
- Rising prevalence of epilepsy (approx. 50 million globally) and bipolar disorder (about 45 million worldwide) (WHO).
- Long-term efficacy of extended-release formulations improves adherence.
- Patent expiry creating opportunities for generic manufacturers.
Barriers
- Price pressures reducing margins for original manufacturers.
- Healthcare reform in certain regions favoring generics over branded drugs.
- Regulatory hurdles for entry in emerging markets.
What Are the Key Opportunities and Risks?
Opportunities
- Expanding generic market in Asia-Pacific countries.
- Formulation innovations, such as combination therapy or improved delivery systems.
- Entry into new markets with regulatory pathways approved.
Risks
- Price erosion due to increased competition.
- Potential regulatory delays or restrictions.
- Changes in prescribing practices favoring newer therapies.
Summary of Market Projections (2023–2028)
| Year | Total Market (USD billions) | Growth Rate | Segment Breakdown | Key Factors |
|---|---|---|---|---|
| 2023 | 1.8 | — | Brand vs. Generics | Post-patent competition, price cuts |
| 2024 | 1.9 | 4.4% | Generics dominate | Increased generic adoption, regional expansion |
| 2025 | 2.0 | 5.0% | Stabilization | Market saturation, stable pricing |
| 2026 | 2.2 | 5.0% | Price stabilization | New regional markets, formulary inclusion |
| 2027 | 2.3 | 4.5% | Market consolidation | Potential entry of complementary therapies |
| 2028 | 2.4 | 4.0% | Mature market | Price competition, new formulations |
Key Takeaways
- KARBINAL ER's original patent expired in 2025, opening the market to numerous generics.
- The global market for carbamazepine formulations is expanding modestly at 4-6% annually.
- Prices are declining, with generics now typically costing less than half the original brand.
- High prevalence of epilepsy and bipolar disorder fuels steady demand.
- Regional differences exist, with North America leading in market size and Asia-Pacific showing strong growth prospects.
FAQs
Q1: How does the expiry of patent protection affect KARBINAL ER's pricing?
Post-2025, patent expiry allows generic manufacturers to produce versions of carbamazepine ER, leading to price reductions and increased market competition.
Q2: Are generic versions of KARBINAL ER interchangeable?
Yes, approved generic carbamazepine ER formulations can be substituted for the brand name, provided bioequivalence is established.
Q3: What are the main factors influencing price declines?
Introduction of generics, increased competition, and healthcare policies favoring lower-cost options in various regions drive prices down.
Q4: How significant is regional variation in market dynamics?
Very; North American markets have higher prices driven by insurance reimbursements, whereas price-sensitive markets like parts of Asia-Pacific see steeper declines.
Q5: Are there upcoming formulations or innovations expected for carbamazepine ER?
Current trends focus on formulations improving compliance and delivery, but no major new proprietary formulations are forecasted within the next few years.
References
[1] GlobalData. "Pharmaceutical Market Report: Anticonvulsants." 2022.
[2] WHO. "Epilepsy Fact Sheet." 2022.
[3] FDA. "KARBINAL ER U.S. Label." 2009.
[4] IQVIA. "Medicine Price & Utilization Data," 2022.
More… ↓
