Share This Page
Drug Price Trends for K-PHOS NEUTRAL TABLET
✉ Email this page to a colleague
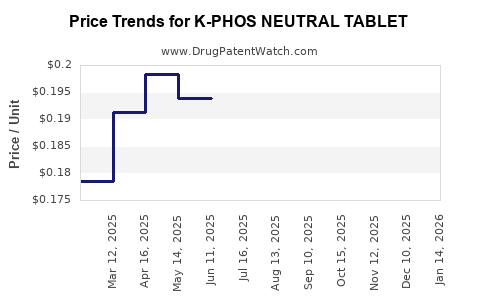
Average Pharmacy Cost for K-PHOS NEUTRAL TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| K-PHOS NEUTRAL TABLET | 00486-1125-01 | 0.26571 | EACH | 2026-03-18 |
| K-PHOS NEUTRAL TABLET | 00486-1125-05 | 0.26571 | EACH | 2026-03-18 |
| K-PHOS NEUTRAL TABLET | 00486-1125-01 | 0.25546 | EACH | 2026-02-18 |
| K-PHOS NEUTRAL TABLET | 00486-1125-05 | 0.25546 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
K-PHOS NEUTRAL TABLET Market Analysis and Financial Projection
Market Overview and Demand for K-Phos Neutral Tablet
K-Phos Neutral Tablet, a combined potassium supplement typically used to treat or prevent potassium deficiency in patients with chronic kidney disease, gastrointestinal losses, or on certain medications, operates within the broader electrolyte therapy market. This segment is driven by increasing prevalence of kidney disease, aging populations, and growing awareness of electrolyte imbalances.
The global electrolyte replacement therapy market was valued at approximately $3.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of about 6.2% through 2030 [1]. Potassium supplements occupy a significant share within this market, with their demand rising in hospital, outpatient, and home healthcare settings.
Competitive Landscape and Patent Status
Major competitors in the potassium supplement segment include brands like K-DUR (potassium chloride extended-release), Slow-K (potassium chloride), and potassium phosphate formulations like K-Phos. Market entry for new formulations such as K-Phos Neutral Tablet depends on patent status, manufacturing approvals, and physician acceptance.
The patent landscape indicates that many potassium phosphate products face expirations between 2025-2030. A new formulation like K-Phos Neutral Tablet, if patent-protected, could command premium pricing during exclusivity. Without patent protection, market penetration relies on price competitiveness and physician preference.
Regulatory and Manufacturing Considerations
K-Phos Neutral Tablet must receive regulatory approval from agencies such as the FDA in the US or EMA in Europe. Approval involves demonstrating bioequivalence, safety, and manufacturing compliance. Manufacturing costs include raw material procurement, tablet production, and quality testing.
Pricing Analysis and Projections
Current Market Prices
Potassium phosphate products generally retail at prices ranging from $0.10 to $0.25 per tablet in the US. Brand-name formulations like K-Phos are priced at the higher end, especially in hospital or specialty pharmacies.
Projected Pricing Trends
- Year 1 (Post-launch): Prices likely at $0.15 - $0.20 per tablet, assuming a premium positioning and patent protection.
- Year 3: Prices may decline to $0.10 - $0.15 per tablet as competitors introduce generics or alternative formulations.
- Year 5: If patent expiries occur, prices could fall below $0.10 per tablet, matching or undercutting generic equivalents.
Price erosion is projected to follow generic entry, typically within 3-5 years of patent expiry. Volume growth compensates for price reductions in the overall market.
Revenue Estimates
Assuming launch volume of 50 million tablets annually at an average price of $0.18:
| Year | Estimated Sales Volume | Average Price | Revenue |
|---|---|---|---|
| 1 | 50 million | $0.18 | $9 million |
| 3 | 70 million | $0.12 | $8.4 million |
| 5 | 90 million | $0.09 | $8.1 million |
Market expansion could increase volume, but price decline remains a factor.
Strategic Market Entry
Positioning K-Phos Neutral Tablet as a high-quality, cost-effective alternative could drive early adoption. Special considerations include:
- Securing patent protection if possible.
- Building relationships with hospitals and clinics.
- Developing patient education programs to increase adherence.
Key Challenges
- Patent expiries could lead to pricing pressure.
- Competition from established brands and generic products.
- Regulatory delays could impact timing and costs.
Key Takeaways
- The K-Phos Neutral Tablet occupies a growing segment within electrolyte therapy, with increasing demand driven by aging populations and chronic disease prevalence.
- Early pricing is aligned with premium formulations ($0.15-$0.20 per tablet), decreasing as patents expire and generics enter the market.
- Revenue growth depends on volume expansion amid price erosion, with projections around $8-$9 million annual revenue after three years.
- Market entry strategies should emphasize patent protection, clinical positioning, and cost competitiveness.
FAQs
1. What factors influence the price of potassium phosphate tablets?
Raw material costs, manufacturing expenses, patent status, competition, and regulatory approval timelines influence the tablet’s price.
2. How long does patent protection typically last for new drug formulations?
In the US, patents last up to 20 years from filing, but effective exclusivity for drug formulations often ranges between 7-12 years post-approval, depending on extensions and Patent Term Restoration.
3. What is the timeline for generic entry in this segment?
Generally, 3-5 years after patent expiration, depending on market conditions, patent challenges, and regulatory approvals.
4. How does the demand for electrolyte supplements compare globally?
Demand in developed regions like North America and Europe is driven by aging populations and healthcare infrastructure. Emerging markets see increasing demand due to rising chronic disease rates and improved healthcare access.
5. What strategies can extend a product’s market lifespan?
Developing combination therapies, obtaining additional indications, or patenting unique delivery mechanisms can prolong exclusivity and market relevance.
Citations:
[1] MarketsandMarkets, "Electrolyte Replacement and Oral Rehydration Therapy Market," 2022.
More… ↓
