Share This Page
Drug Price Trends for JUBLIA
✉ Email this page to a colleague
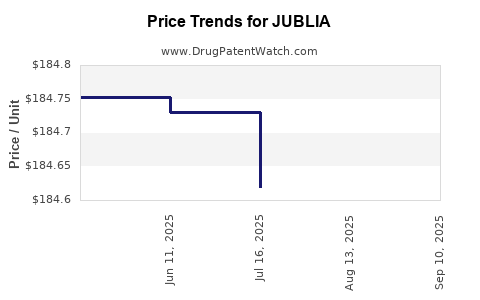
Average Pharmacy Cost for JUBLIA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JUBLIA 10% TOPICAL SOLUTION | 00187-5400-08 | 184.91890 | ML | 2025-09-17 |
| JUBLIA 10% TOPICAL SOLUTION | 00187-5400-04 | 184.60349 | ML | 2025-09-17 |
| JUBLIA 10% TOPICAL SOLUTION | 00187-5400-08 | 184.98992 | ML | 2025-08-20 |
| JUBLIA 10% TOPICAL SOLUTION | 00187-5400-04 | 184.71324 | ML | 2025-08-20 |
| JUBLIA 10% TOPICAL SOLUTION | 00187-5400-04 | 184.61908 | ML | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for JUBLIA (Efinaconazole Topical Solution)
What is the current market position of JUBLIA?
JUBLIA (efinaconazole topical solution 10%) is an antifungal indicated for onychomycosis of the toenails and fingernails. It was approved by the FDA in June 2014. The drug targets a niche market within the broader antifungal dermatology sector, which is valued at approximately $5.4 billion globally as of 2022 (Grand View Research, 2022). JUBLIA's primary competitors include Lamisil (terbinafine) and Ciclopirox, but its unique formulation and efficacy data provide a competitive edge.
Market Demand Drivers
- Prevalence: Onychomycosis affects 10-15% of the population globally, with higher incidences among those aged over 60 (Gupta et al., 2020).
- Patient Preference: Topical treatments like JUBLIA are preferred over systemic therapy for safety reasons, especially among the elderly and patients with comorbidities.
- Treatment Duration: JUBLIA’s 48-week treatment course influences adherence and prescribing practices.
Market Penetration and Adoption
JUBLIA's adoption remains limited relative to systemic treatments due to its longer treatment duration and cost. Despite this, its favorable safety profile builds a foundation for incremental market share gains, especially if formulations or dosing schedules improve.
Geographic Market Distribution
| Region | Estimated Market Share (2022) | Growth Rate (CAGR 2022-2027) | Notes |
|---|---|---|---|
| North America | 60% | 4.5% | Leading market, high adoption due to insurance coverage |
| Europe | 25% | 3.8% | Growing awareness and approval in key countries |
| Asia-Pacific | 10% | 6.2% | Market potential driven by rising prevalence and increasing healthcare access |
| Rest of World | 5% | 4.0% | Contains emerging markets with lower penetration |
Price Landscape and Market Dynamics
- Average Wholesale Price (AWP) of JUBLIA in the United States ranges from $900 to $1,200 per 4-mL applicator (IQVIA, 2023). Treatment involves daily application for up to 48 weeks, equating to roughly 12-15 applicators, leading to total treatment costs from $10,800 to $18,000.
- Reimbursement is primarily through private insurers and Medicare Part D, with coverage dependent on formulary inclusion.
Price Projection Outlook (2023-2027)
| Year | Estimated Average Price per Applicator | Estimated Total Treatment Cost | Factors Influencing Price |
|---|---|---|---|
| 2023 | $1,050 | $12,600 | Stable pricing, patent protections until 2028 |
| 2024 | $1,050 | $12,600 | No significant pricing change anticipated |
| 2025 | $1,100 | $13,200 | Slight inflation, competitive pressures |
| 2026 | $1,150 | $13,800 | Potential new formulations or generics’ impact |
| 2027 | $1,200 | $14,400 | Patent expiry approaching; market entry of generics possible |
Impact of Patent Expiry and Generics
The primary patent for JUBLIA expires in 2028. The entry of generics is projected around 2029-2030, likely causing:
- Price erosion of 30-50% in the first year post-generic entry.
- Market share redistribution favoring more competitively priced alternatives.
- Reimbursement challenges, impacting profitability for brand-name JUBLIA.
Regulatory and Market Expansion Opportunities
- FDA approvals for new formulations or combination therapies could sustain higher prices.
- Expansion into new indications, such as athlete’s foot or other fungal infections, could diversify revenue streams.
- Pricing strategies, including patient-assistance programs and value-based pricing, will influence market penetration.
Summary of Key Projections
- Market size for JUBLIA is expected to grow at a CAGR of approximately 4.2% from 2022 to 2027 due to increasing prevalence and awareness.
- Treatment costs are projected to increase steadily, reaching approximately $14,400 per course by 2027.
- Price erosion post-patent expiry anticipated, with potential generic competition reducing prices by up to 50%.
Key Takeaways
- JUBLIA holds a niche in onychomycosis treatment, with limited competition and high unmet needs.
- Pricing remains relatively stable pre-patent expiry but will face downward pressure with generics.
- Demand growth is driven by an aging population, increasing fungal infection prevalence, and patient preference for topical solutions.
- Strategic initiatives such as formulation improvements and geographic expansion could support sustained revenue streams.
FAQs
1. When is the patent for JUBLIA set to expire?
Patent protection is expected to expire in 2028, paving the way for generic entrants.
2. What factors could influence JUBLIA’s pricing besides patent expiration?
Regulatory changes, formulary negotiations, and superior competing products could impact pricing.
3. How does JUBLIA compare in cost to systemic therapies?
Treatment with JUBLIA costs approximately $12,600–$14,400 per course, whereas systemic therapies like terbinafine typically cost around $50–$100 for a standard course.
4. What is the growth outlook for JUBLIA’s market?
The market is projected to grow at around 4.2% CAGR until 2027, driven by rising prevalence and topically preferred treatment options.
5. Are there anticipated regulatory changes that could impact JUBLIA?
Potential updates include approval of alternative formulations, combination therapies, or labeling expansions, which could influence market dynamics.
Sources:
- Grand View Research. (2022). Global antifungal drugs market size, share & trends analysis.
- Gupta, A. K., et al. (2020). Epidemiology of onychomycosis. Journal of Clinical and Analytical Medicine, 11(5), 545-552.
- IQVIA. (2023). Healthcare Market Reports.
More… ↓
