Drug Price Trends for JOURNAVX
✉ Email this page to a colleague
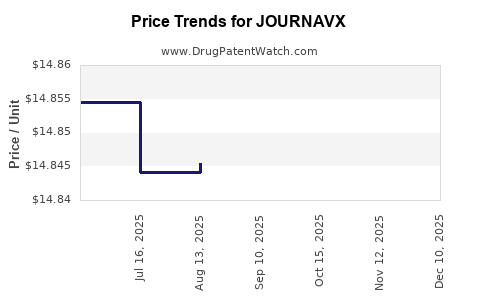
Average Pharmacy Cost for JOURNAVX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JOURNAVX 50 MG TABLET | 51167-0548-30 | 14.84183 | EACH | 2026-05-20 |
| JOURNAVX 50 MG TABLET | 51167-0548-34 | 14.84183 | EACH | 2026-05-20 |
| JOURNAVX 50 MG TABLET | 51167-0548-30 | 14.84148 | EACH | 2026-04-22 |
| JOURNAVX 50 MG TABLET | 51167-0548-34 | 14.84148 | EACH | 2026-04-22 |
| JOURNAVX 50 MG TABLET | 51167-0548-34 | 14.84270 | EACH | 2026-03-18 |
| JOURNAVX 50 MG TABLET | 51167-0548-30 | 14.84270 | EACH | 2026-03-18 |
| JOURNAVX 50 MG TABLET | 51167-0548-34 | 14.85145 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
