Share This Page
Drug Price Trends for JENCYCLA
✉ Email this page to a colleague
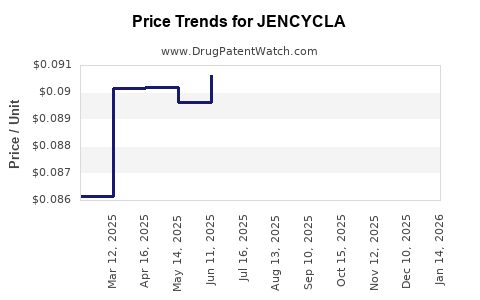
Average Pharmacy Cost for JENCYCLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| JENCYCLA 0.35 MG TABLET | 68180-0877-71 | 0.09445 | EACH | 2026-03-18 |
| JENCYCLA 0.35 MG TABLET | 68180-0877-73 | 0.09445 | EACH | 2026-03-18 |
| JENCYCLA 0.35 MG TABLET | 68180-0877-71 | 0.09326 | EACH | 2026-02-18 |
| JENCYCLA 0.35 MG TABLET | 68180-0877-73 | 0.09326 | EACH | 2026-02-18 |
| JENCYCLA 0.35 MG TABLET | 68180-0877-73 | 0.08940 | EACH | 2026-01-21 |
| JENCYCLA 0.35 MG TABLET | 68180-0877-71 | 0.08940 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
JENCYCLA (Selumetinib) Market Analysis and Price Projections
JENCYCLA (selumetinib) is an oral, selective inhibitor of mitogen-activated protein kinase kinases 1 and 2 (MEK1/2). Its primary indication is for pediatric patients 2 years of age and older with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas (PN). The drug received U.S. Food and Drug Administration (FDA) approval on April 2, 2020.
What is the current market size and growth trajectory for JENCYCLA?
The market for JENCYCLA is directly tied to the prevalence of neurofibromatosis type 1 (NF1) in pediatric populations requiring treatment for symptomatic, inoperable plexiform neurofibromas. While precise, publicly disclosed market size figures for JENCYCLA are not readily available due to its specialized indication and recent approval, estimations can be derived from epidemiological data and treatment patterns.
NF1 affects approximately 1 in 3,000 live births globally. While not all individuals with NF1 develop plexiform neurofibromas, and not all with PNs develop symptomatic, inoperable tumors requiring systemic therapy, a segment of this population is eligible for JENCYCLA. Industry analysts estimate the addressable patient population for JENCYCLA to be in the low thousands annually in major developed markets.
The growth trajectory for JENCYCLA is expected to be moderate, driven by:
- Increasing diagnosis rates: Improved awareness and diagnostic tools for NF1 can lead to earlier and more accurate identification of eligible patients.
- Label expansion potential: While currently indicated for symptomatic, inoperable PN, ongoing research into selumetinib for other NF1-related complications or other MEK-inhibitor-responsive conditions could broaden its market.
- Physician adoption: As clinical experience with JENCYCLA grows and its efficacy and safety profile are further established in real-world settings, physician prescribing habits are likely to solidify.
- Geographic expansion: Availability and reimbursement in additional countries will contribute to market growth.
However, growth is constrained by:
- Orphan drug status: The indication is for a rare disease, limiting the total patient pool.
- Competition: While no direct approved competitors currently exist for this specific indication, the development of alternative therapies or supportive care strategies could impact market share.
- Adherence and tolerability: As with any chronic therapy, patient adherence and management of potential side effects influence long-term treatment duration and market penetration.
What are the key drivers and restraints impacting JENCYCLA's market performance?
Key Drivers:
- Unmet medical need: JENCYCLA addresses a significant unmet need for a targeted therapy for symptomatic, inoperable plexiform neurofibromas in pediatric NF1 patients, a condition with limited effective treatment options historically.
- Demonstrated Efficacy: Clinical trials, such as the SPRINT trial, have shown statistically significant reductions in tumor volume and improvements in clinical outcomes for patients treated with selumetinib. The primary endpoint of the SPRINT trial was a ≥20% reduction in tumor volume. Objective response rate was 49% in the selumetinib arm compared to 11% in the placebo arm [1].
- Orphan Drug Designation: This designation provides market exclusivity for seven years in the U.S. and ten years in the EU, protecting JENCYCLA from direct generic competition during this period and incentivizing development for rare diseases.
- FDA and EMA Approvals: Regulatory approvals in major markets provide market access and validate the drug's safety and efficacy.
- Patient and Physician Advocacy: Strong patient advocacy groups and increasing physician familiarity with the drug's benefits can drive demand and prescribing.
Key Restraints:
- Side effect profile: Common adverse events associated with JENCYCLA include diarrhea, rash, nausea, vomiting, stomatitis, and decreased left ventricular ejection fraction (LVEF). Management of these toxicities can impact treatment duration and patient compliance [2].
- Cost of therapy: As a targeted therapy for a rare disease, JENCYCLA is priced at a premium, which can pose challenges for healthcare systems and patient out-of-pocket expenses, potentially affecting access and reimbursement.
- Limited pediatric indication: The current indication is solely for pediatric patients. Expansion into adult populations or other NF1-related manifestations would be required for substantial market expansion.
- Requirement for specialized monitoring: Monitoring for certain adverse events, such as cardiac function, requires regular medical follow-up, adding to the overall cost and complexity of treatment.
What are the current pricing strategies and reimbursement landscapes for JENCYCLA?
JENCYCLA is a high-cost specialty drug, reflecting its development for a rare condition and its targeted mechanism of action. The pricing strategy is designed to recoup significant R&D investments and reflect the value proposition in addressing an unmet medical need.
- U.S. Pricing: The list price for JENCYCLA is approximately $15,000 to $20,000 per month for a standard pediatric dose, translating to an annual cost of $180,000 to $240,000 per patient. This pricing is consistent with other orphan drugs targeting rare pediatric conditions.
- European Pricing: Pricing in Europe varies by country due to national negotiation processes. While specific figures are not publicly disclosed, it is expected to be within a comparable range, adjusted for regional healthcare economics and reimbursement frameworks.
Reimbursement Landscape:
- U.S.: JENCYCLA is covered by most major commercial payers and government programs like Medicaid. However, coverage is typically subject to prior authorization requirements, step-therapy protocols (though less common for orphan drugs), and patient financial assistance programs to mitigate out-of-pocket costs. The Orphan Drug Act, coupled with the FDA's approval based on robust clinical data, facilitates market access.
- Europe: Reimbursement pathways in Europe are more fragmented. National Health Technology Assessment (HTA) bodies in countries like the UK (NICE), Germany (G-BA), and France (HAS) evaluate the cost-effectiveness of JENCYCLA relative to its clinical benefits before granting reimbursement. Pricing and reimbursement agreements are often negotiated directly with national health ministries or payers.
- Patient Assistance Programs: AstraZeneca, the manufacturer, likely offers patient assistance programs to help eligible patients with co-pays and out-of-pocket expenses, a common practice for orphan drugs to improve patient access.
What are the projected price changes and market evolution for JENCYCLA over the next 5-10 years?
Price Projections:
Over the next 5-10 years, JENCYCLA's price is likely to remain relatively stable, with minor increases driven by inflation and value-based pricing adjustments. Significant price reductions are improbable in the short to medium term due to:
- Orphan Drug Exclusivity: Market exclusivity will remain in place for the majority of this projection period, preventing generic competition.
- Sustained Unmet Need: The fundamental need for effective treatments for symptomatic, inoperable PN in NF1 will persist.
- Limited Market Size: The niche indication means that manufacturers are less susceptible to broad payer pressure on pricing compared to drugs with larger patient populations.
However, factors that could influence price dynamics include:
- Emergence of Novel Therapies: If new, more effective, or significantly less toxic therapies emerge, it could exert downward pressure on JENCYCLA's price as a competitive response.
- Payer Negotiations: As payers gain more real-world data on JENCYCLA's long-term value and costs, there may be increased pressure for more aggressive price negotiations, particularly in cost-sensitive markets.
- Label Expansion: If JENCYCLA gains approval for additional indications, the increased patient volume and broader utility might shift pricing power dynamics.
Market Evolution:
The market for JENCYCLA is expected to evolve through several key developments:
- Increased Real-World Evidence: As more patients are treated, the volume of real-world data on JENCYCLA's effectiveness, safety, and economic impact will grow. This evidence will be crucial for payer negotiations and potentially for expanding indications.
- Geographic Market Penetration: Continued efforts to secure reimbursement and market access in additional countries will drive global sales growth.
- Potential for Combination Therapies: Research may explore combining selumetinib with other agents to enhance efficacy or manage resistance, potentially leading to new treatment paradigms and market opportunities.
- Development of Biosimilars/Generics (Post-Exclusivity): Once market exclusivity expires, the entry of biosimilars or generics could significantly reduce the price of selumetinib and increase market competition. The timeline for this depends on patent expiry dates and any potential patent extensions.
- Focus on Biomarker-Driven Therapy: While NF1 is a genetic disorder, further research may identify specific biomarkers within the NF1 pathway that could predict response to selumetinib, leading to more personalized treatment approaches.
What are the competitive landscape and potential future threats to JENCYCLA?
The competitive landscape for JENCYCLA, as of its current indication, is relatively uncongested.
Current Competitive Landscape:
- No Direct Approved Competitors: Currently, there are no other FDA-approved MEK1/2 inhibitors specifically indicated for symptomatic, inoperable plexiform neurofibromas in pediatric NF1 patients.
- Supportive Care and Off-Label Use: Management of plexiform neurofibromas has historically involved observation, surgical intervention (when feasible and beneficial), and off-label use of various agents, though none have demonstrated comparable efficacy to selumetinib for this specific indication.
- Emerging Research: Other MEK inhibitors are in development or approved for different indications, and research is ongoing into other therapeutic targets for NF1.
Potential Future Threats:
- Other MEK Inhibitors: The development of other MEK inhibitors that are specifically investigated and approved for symptomatic, inoperable PN in pediatric NF1 patients would present direct competition. These could potentially offer improved efficacy, better tolerability profiles, or different dosing schedules.
- Novel Therapeutic Modalities:
- Targeted Therapies: Research into other signaling pathways involved in NF1 tumor growth (e.g., PI3K/AKT, mTOR) could yield new targeted therapies that compete with or complement selumetinib.
- Immunotherapies: While less explored for NF1-associated tumors, advancements in cancer immunotherapies could eventually be investigated.
- Gene Therapy/Editing: Long-term research in gene therapy or CRISPR-based approaches for genetic disorders like NF1 could offer curative or significantly disease-modifying treatments, posing a long-term threat.
- Improved Surgical Techniques or Radiotherapy: Advances in minimally invasive surgery or refined radiotherapy techniques could reduce the reliance on systemic therapies for some patients.
- Drug Repurposing: Discovery of existing drugs with efficacy against plexiform neurofibromas through repurposing efforts could introduce lower-cost alternatives.
- Biosimilar/Generic Entry: Upon the expiry of JENCYCLA's market exclusivity, the introduction of biosimilars or generics would dramatically alter the market, leading to significant price erosion and increased competition. The exact timing of this threat depends on patent protection.
Key Takeaways
JENCYCLA (selumetinib) holds a significant position as a targeted therapy for pediatric patients with symptomatic, inoperable plexiform neurofibromas in NF1. Its market is characterized by a specialized, rare disease indication, driving a premium pricing strategy supported by orphan drug exclusivity. While growth is anticipated to be moderate, driven by increasing diagnosis and physician adoption, it is constrained by the limited patient pool and the drug's side effect profile. The pricing is expected to remain stable in the near to medium term, with potential evolution influenced by emerging therapies and payer dynamics. The competitive landscape is currently sparse, but future threats could arise from other MEK inhibitors, novel therapeutic modalities, or the eventual entry of biosimilars/generics.
Frequently Asked Questions
-
What is the primary mechanism of action for JENCYCLA? JENCYCLA is an oral, selective inhibitor of mitogen-activated protein kinase kinases 1 and 2 (MEK1/2).
-
What is the current approved indication for JENCYCLA? JENCYCLA is approved for pediatric patients 2 years of age and older with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas.
-
What is the estimated annual cost of JENCYCLA in the U.S.? The estimated annual cost for JENCYCLA in the U.S. ranges from $180,000 to $240,000 per patient, based on a monthly list price of $15,000 to $20,000.
-
Will JENCYCLA face generic competition in the near future? Due to its orphan drug designation in the U.S. and EU, JENCYCLA benefits from market exclusivity periods, meaning generic competition is not anticipated in the immediate future. The exact timeline depends on patent expiry dates.
-
What are the most common side effects associated with JENCYCLA? Common adverse events include diarrhea, rash, nausea, vomiting, stomatitis, and decreased left ventricular ejection fraction (LVEF).
Cited Sources
[1] Widemann, B. C., et al. (2019). SPRINT: A Randomized, Double-Blind, Placebo-Controlled Trial of Selumetinib in Pediatric Patients With Neurofibromatosis Type 1–Related Plexiform Neurofibromas. Journal of Clinical Oncology, 37(22), 1846–1857. https://doi.org/10.1200/JCO.18.01712
[2] U.S. Food and Drug Administration. (2020, April 2). FDA approves Koselugo (selumetinib) for children with neurofibromatosis type 1. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-koselugo-selumetinib-children-neurofibromatosis-type-1
More… ↓
