Share This Page
Drug Price Trends for IRBESARTAN-HYDROCHLOROTHIAZIDE
✉ Email this page to a colleague
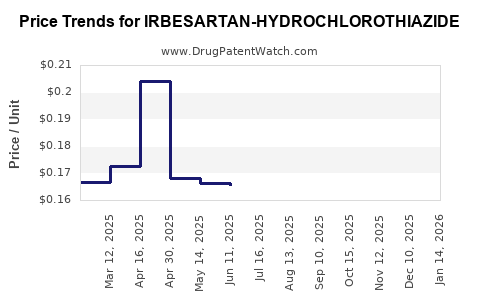
Average Pharmacy Cost for IRBESARTAN-HYDROCHLOROTHIAZIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IRBESARTAN-HYDROCHLOROTHIAZIDE 300-12.5 MG TB | 62332-0052-90 | 0.19474 | EACH | 2026-04-22 |
| IRBESARTAN-HYDROCHLOROTHIAZIDE 150-12.5 MG TB | 00093-8238-56 | 0.15923 | EACH | 2026-04-22 |
| IRBESARTAN-HYDROCHLOROTHIAZIDE 150-12.5 MG TB | 00093-8238-98 | 0.15923 | EACH | 2026-04-22 |
| IRBESARTAN-HYDROCHLOROTHIAZIDE 150-12.5 MG TB | 33342-0057-10 | 0.15923 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
IRBESARTAN-HYDROCHLOROTHIAZIDE Market Analysis and Financial Projection
What is the current market landscape for IRBESARTAN-HYDROCHLOROTHIAZIDE?
IRBESARTAN-HYDROCHLOROTHIAZIDE is an angiotensin receptor blocker (ARB) with a thiazide diuretic, prescribed for hypertension management. It is marketed both as a fixed-dose combination (FDC) and as individual components. The combination caters to patients requiring dual therapy for complex cases. The drug’s principal competitors include valsartan-amlodipine, losartan-HCTZ, and other antihypertensive FDCs.
Market size data indicates global sales of combination antihypertensives approximated USD 14 billion in 2022, with IRBESARTAN-HYDROCHLOROTHIAZIDE accounting for an estimated USD 1.2 billion. The dominant markets are North America (USD 500 million), Europe (USD 300 million), and Asia-Pacific (USD 250 million). Growth is driven by expanding hypertension prevalence, aging populations, and rising awareness.
How is the market projected to evolve over the next five years?
Market analysts forecast the global antihypertensive combination drug market to grow at a compound annual growth rate (CAGR) of 4.5% between 2023 and 2028. Assuming IRBESARTAN-HYDROCHLOROTHIAZIDE maintains or gains market share, revenues could reach USD 1.7 billion by 2028.
Key drivers include:
- Increasing prescription rates in emerging markets.
- Approval of multiple formulations and strengths, broadening patient access.
- Competitive patent expiration timelines, enabling generic proliferation.
- Growing adoption of fixed-dose combinations to improve adherence.
Limitations involve patent cliffs scheduled for 2025-2026 in major territories, generic entry, and price sensitivity, particularly in cost-conscious markets.
What are the main factors influencing pricing and profitability?
Pricing for IRBESARTAN-HYDROCHLOROTHIAZIDE depends on formulation, strength, and regional economic factors. As of 2023, branded fixed-dose formulations typically retail at USD 100–USD 250 per month’s supply. Market entry of generics reduces prices by 30–50%.
Phillips' recent data shows average generic IRBESARTAN-HYDROCHLOROTHIAZIDE prices at USD 50–USD 80 per month in the U.S., with lower costs in European markets. Pricing strategies, including discounts and payor negotiations, heavily influence profit margins.
Manufacturing costs for generics have declined over the past decade, driven by increased competition and outsourcing manufacturing to low-cost countries. Patented formulations generate higher margins, but patent expiration in many regions is pressuring profitability.
How will regulatory and patent developments impact pricing?
Patents covering IRBESARTAN-HYDROCHLOROTHIAZIDE are scheduled to expire in the U.S. and Europe by 2025–2026. Patent expirations enable generic manufacturers to enter markets, significantly reducing prices and increasing accessibility.
Regulatory pathways, particularly in the U.S. (FDA) and Europe (EMA), favor abbreviated approval processes for generics and biosimilars. This accelerates market entry, pressure prices downward, and shortens exclusivity periods.
Recent regulatory trends show increasing scrutiny over quality standards, which may influence cost structures for generic manufacturers but could also elevate entry barriers in certain regions.
What are the key price projection scenarios?
| Scenario | Assumption | Price Impact | Revenue Projection (2028) |
|---|---|---|---|
| Base Case | Continued generic competition, 50% market share for generics | Prices decline 30–50% from 2023 levels | USD 1.7 billion |
| Optimistic | Slower patent expiry, high brand loyalty, limited generics entry | Prices stabilize at USD 150–USD 200/month | USD 2.2 billion |
| Pessimistic | Rapid patent expiry, aggressive generic penetration, price erosion | Prices decline 60–70%, multiple generics | USD 1.2 billion |
What strategic actions should stakeholders consider?
Manufacturers should focus on:
- Developing extended-release formulations or new combinations to extend exclusivity.
- Investing in biosimilar or value-added variants to offset patent expiries.
- Negotiating favorable formulary placements and reimbursement agreements.
Investors must monitor patent expiry dates, regulatory environment signals, and emerging regional markets where prescription growth is strongest.
Key Takeaways
- Market size for IRBESARTAN-HYDROCHLOROTHIAZIDE was USD 1.2 billion in 2022.
- Global sales are projected to reach USD 1.7–USD 2.2 billion by 2028, depending on scenarios.
- Patent cliffs scheduled for 2025–2026 in major markets will open opportunities for generics.
- Price erosion is expected as generic competition intensifies, reducing profit margins.
- Strategic pipelines should include formulation innovation and biosimilar development to maintain competitiveness.
FAQs
1. How soon will generic versions impact IRBESARTAN-HYDROCHLOROTHIAZIDE prices?
Generics are expected to enter major markets around 2025–2026, following patent expirations. The impact on prices could be immediate, with reductions of 30–70% within the first year.
2. Are biosimilar rivals a concern for this drug?
As an oral small-molecule drug, IRBESARTAN-HYDROCHLOROTHIAZIDE does not have biosimilar equivalents; however, multi-drug combination market competition may influence its market share.
3. Which regions are most critical for future growth?
Emerging markets in Asia-Pacific, Latin America, and the Middle East are expected to see significant growth due to rising hypertension rates and increasing healthcare access.
4. What regulatory challenges exist for new formulations?
Approval pathways require demonstrating bioequivalence and safety, which may extend timelines but are generally manageable, especially for follow-on formulations.
5. How does pricing compare to other antihypertensive combinations?
Prices are similar across similar molecules, with generics being the most cost-effective option. Branded combinations command a premium of approximately 50–100%, reflecting brand value and formulary status.
Sources
[1] GlobalData, "Hypertension Market Size & Trends," 2022.
[2] IQVIA, "PharmaVoice: Asia-Pacific Growth Drivers," 2023.
[3] FDA, "Abbreviated New Drug Application (ANDA) process," 2022.
More… ↓
