Share This Page
Drug Price Trends for INVOKANA
✉ Email this page to a colleague
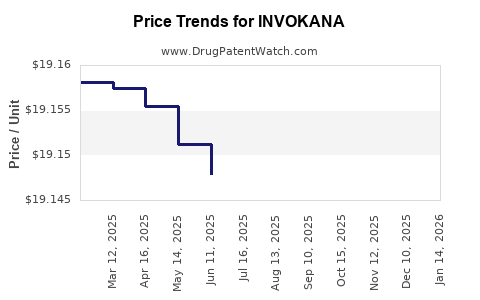
Average Pharmacy Cost for INVOKANA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INVOKANA 100 MG TABLET | 50458-0140-30 | 19.10376 | EACH | 2026-04-22 |
| INVOKANA 100 MG TABLET | 50458-0140-90 | 19.10376 | EACH | 2026-04-22 |
| INVOKANA 300 MG TABLET | 50458-0141-90 | 19.13024 | EACH | 2026-04-22 |
| INVOKANA 300 MG TABLET | 50458-0141-30 | 19.13024 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for INVOKANA (Canagliflozin)
Market Position
INVOKANA (canagliflozin) is a sodium-glucose co-transporter 2 (SGLT2) inhibitor manufactured by Johnson & Johnson. Approved by the FDA in 2013 for type 2 diabetes mellitus (T2DM), it functions by reducing renal glucose reabsorption, increasing urinary glucose excretion, and lowering blood glucose levels. As an early entrant in the SGLT2 inhibitor space, INVOKANA has maintained a strong market presence.
Market Size and Growth
The global diabetes drugs market is valued at approximately $82 billion in 2022, with the SGLT2 inhibitor segment accounting for roughly 25% ($20.5 billion). INVOKANA's market share in the SGLT2 space is estimated at 30-35%, translating to revenue of about $6.15-$7.2 billion annually in peak periods.
The market is expanding due to increased diabetes prevalence, lifestyle-related risk factors, and expanded label indications. The global diabetic population is projected to reach 700 million by 2045, driving long-term growth for SGLT2 inhibitors.
Competitive Landscape
Key competitors include:
- Jardiance (empagliflozin): Market leader with 45% share.
- Farxiga (dapagliflozin): Holds 20-25% market share.
- Invokana (canagliflozin): About 30-35%.
Other drugs, such as Eli Lilly's Jardiance and Astela's dapagliflozin, have launched additional indications including heart failure and chronic kidney disease, expanding market potential.
Pricing Trends
In the U.S., the wholesale acquisition cost (WAC) for INVOKANA was approximately $550 per month per patient in 2022. Patient out-of-pocket costs vary based on insurance plans; branded drug prices tend to decrease through rebates and discounts.
Average annual treatment cost: $6,600 (~$550/month).
Historical and Projected Price Dynamics
-
Historical trends: Price reductions of 10-15% annually have been observed driven by generic competition (expected to emerge within 5 years), insurance negotiations, and formulary shifts.
-
Future projections: With patent expiration anticipated around 2027-2028, generic versions could enter the market, pressuring prices downward by 40-60%. Prior instances after patent expiry of similar drugs suggest prices could fall to $200–$300 per month, roughly $2,400–$3,600 annually.
Market Drivers Affecting Future Price and Volume
- Expanded FDA-approved indications, including heart failure (2020) and chronic kidney disease (2021), broaden INVOKANA's patient base.
- Shift toward cardiovascular and renal protection, increasing off-label use.
- Growing reimbursement emphasis on cost-effectiveness metrics.
Potential Revenue Impact
Assuming patent expiry and generic entry in 2028, revenues could decline by 50%, from peak sales of ~$7 billion to ~$3.5 billion, unless the drug secures new indications or formulations.
Summary Table
| Aspect | Details |
|---|---|
| Current market size | ~$20.5 billion (SGLT2 segment) |
| INVOKANA market share | 30-35% |
| Current annual revenue | $6.1–$7.2 billion |
| Current price (monthly) | ~$550 |
| Expected patent expiry | 2027–2028 |
| Price post-generic entry | $200–$300/month (projected) |
| Revenue after patent expiry | Potential decrease by 50% |
Key Takeaways
- INVOKANA remains a leading SGLT2 inhibitor with substantial market share.
- The market is expanding due to additional approved indications.
- Price reductions are inevitable upon patent expiration, with potential revenue declines.
- Competition (Jardiance and Farxiga) influences pricing and market dynamics.
- Future growth depends on expanding indications, formulary coverage, and clinical trial results.
FAQs
-
When does INVOKANA face generic competition?
Patent expiration is expected around 2027–2028, after which generic versions are likely to enter the market. -
How will new indications influence INVOKANA's market?
Approval for heart failure and kidney disease broadens patient populations, potentially sustaining revenue before patent expiry. -
What factors could slow down price declines post-generic entry?
Limited number of generic entrants and exclusive licensing agreements can mitigate price erosion. -
How does insurance coverage impact revenue projections?
Reimbursement policies and formulary placements significantly influence actual market penetration and drug pricing. -
Are there new formulations or delivery methods under development?
No major formulations are announced, but ongoing clinical trials aim to demonstrate additional benefits, possibly influencing future pricing strategies.
Citations
[1] IQVIA. "Global Diabetes Drugs Market Analysis," 2022.
[2] FDA. "Canagliflozin (Invokana) Approval and Indications," 2013.
[3] Johnson & Johnson. "Invokana Prescribing Information," 2022.
[4] EvaluatePharma. "Pharmaceutical Sales and Forecast Data," 2022.
[5] ClinicalTrials.gov. "Invokana Clinical Trials and Indications," 2022.
More… ↓
