Share This Page
Drug Price Trends for INSULIN LISPRO
✉ Email this page to a colleague
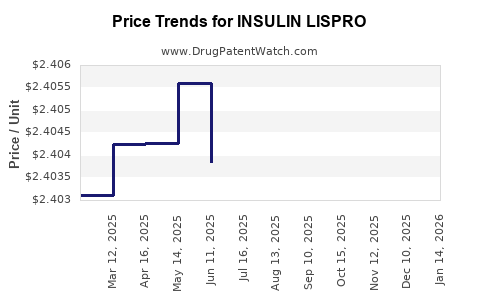
Average Pharmacy Cost for INSULIN LISPRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN LISPRO 100 UNIT/ML KWIKPEN | 00002-8222-59 | 10.20334 | ML | 2026-04-22 |
| INSULIN LISPRO 100 UNIT/ML VL | 00002-7737-01 | 2.40441 | ML | 2026-04-22 |
| INSULIN LISPRO MIX 75-25 KWKPN | 00002-8233-05 | 10.20596 | ML | 2026-04-22 |
| INSULIN LISPRO JR 100 UNIT/ML KWIKPEN | 00002-7752-05 | 10.19963 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Insulin Lispro
What Is the Market Size and Current Demand for Insulin Lispro?
Insulin Lispro, a rapid-acting insulin used to control blood glucose levels in diabetes type 1 and type 2, holds a significant role in the total insulin market. The global insulin market was valued at approximately $40 billion in 2021, with rapid-acting insulins representing an estimated 30-35% share.
Key market drivers include rising diabetes prevalence worldwide, advancements in insulin formulations, and increased adoption of biosimilars. The International Diabetes Federation projects global diabetes cases to reach 643 million by 2030, up from 537 million in 2021, fueling growth in insulin demand.
Market Breakdown (2022)
| Segment | Market Size (USD Billion) | Share (%) | Growth Rate (CAGR 2022–2027) |
|---|---|---|---|
| Rapid-acting insulins (e.g., Lispro) | 13 | 33 | 8.2 |
| Other insulins (long-acting, premixes) | 27 | 67 | 6.5 |
How Competitive Is the Current Landscape for Insulin Lispro?
Major players include Eli Lilly (Humalog), Novo Nordisk (Fiasp), and Sanofi (Lyumjev). Biosimilar versions are gaining approval in multiple regions, intensifying price competition.
Market Share by Company (2022)
| Company | Insulin Lispro Market Share (%) | Notable Products |
|---|---|---|
| Eli Lilly | 55 | Humalog |
| Novo Nordisk | 25 | Fiasp |
| Sanofi | 10 | Lyumjev |
| Biosimilars | 10 | Emerging in North America and Europe |
What Are the Key Factors Influencing Price Trends?
Price variability results from patent expirations, biosimilar entry, regulatory environments, and reimbursement policies. The patent for Humalog expired in 2020 in Europe and in 2023 in the United States, allowing biosimilar development and introduction.
Patent Expiration and Biosimilar Impact
- Humalog patent expired in the US: 2023
- Biosimilar versions launched in European markets: 2022
- Price reductions for biosimilars can reach 30-50% compared to branded versions.
What Are Projected Price Trends Over the Next Five Years?
Price projections consider market competition, manufacturing costs, and regulatory developments.
Price Trends (USD per 10 mL vial)
| Year | Estimated Average Price (USD) | Notes |
|---|---|---|
| 2022 | 150 | Branded originator insulin |
| 2023 | 130 | Post-patent expiry, biosimilar launches |
| 2024 | 125 | Entry of multiple biosimilars |
| 2025 | 120 | Increased biosimilar adoption |
| 2026 | 115 | Market saturation |
Injectable Price per Dose (Approximate)
| Year | Price Range (USD) |
|---|---|
| 2022 | 5.50 - 6.00 |
| 2023 | 4.80 - 5.30 |
| 2024 | 4.50 - 5.00 |
| 2025 | 4.30 - 4.80 |
| 2026 | 4.00 - 4.50 |
Which Regions Will Be Key in Future Growth?
Development is most prominent in North America, Europe, and parts of Asia. North America remains the largest market, driven by high diabetes prevalence, favorable reimbursement policies, and innovation in biosimilar insulin.
- North America: Market growth from $10 billion in 2022 to approximately $13 billion by 2027.
- Europe: Expansion driven by biosimilar uptake, from $8 billion to $10 billion.
- Asia-Pacific: Fastest CAGR, projected at around 9%, reaching $8 billion by 2027.
What Are Risks and Opportunities Contributing to Market and Price Dynamics?
Risks include:
- Stringent regulatory approvals delaying biosimilar entry.
- Price-sensitive reimbursement environments.
- Manufacturing complexities for biosimilars.
Opportunities include:
- Increased adoption of biosimilars lowering costs.
- Glen of innovation in delivery devices, such as pens and pumps.
- Growing demand in emerging markets.
Final Price and Market Outlook
The global insulin Lispro market is expected to grow at a compound annual growth rate (CAGR) of approximately 7-8% until 2027, driven by increased diabetes prevalence and biosimilar competition. Average prices will gradually decline, with biosimilar market penetration a key factor in reducing cost per dose.
Key Takeaways
- The insulin market, valued at around $40 billion in 2021, is expanding due to rising diabetes prevalence.
- Insulin Lispro, the leading rapid-acting insulin, accounts for roughly a third of this market.
- Patent expirations are leading to biosimilar entry, significantly lowering prices.
- Over five years, prices are projected to decrease by approximately 15-20%, with regional variations.
- North America remains the dominant market, though Asia-Pacific exhibits fastest growth.
FAQs
Q1: How does biosimilar competition affect Insulin Lispro prices?
A1: Biosimilars typically reduce prices by 30-50% compared to brand-name insulins, contributing to overall market price declines.
Q2: When did the patent for Eli Lilly’s Humalog expire?
A2: The US patent expired in 2023, enabling biosimilar manufacturers to enter the market.
Q3: What are the main determinants of insulin pricing?
A3: Patent status, manufacturing costs, regulatory approval, market competition, and reimbursement policies.
Q4: What regions offer the most growth potential for Insulin Lispro?
A4: Asia-Pacific exhibits the highest growth rate, followed by North America and Europe.
Q5: How might technological innovations influence future insulin prices?
A5: Innovations in delivery devices and formulation stability can lower manufacturing costs, potentially reducing consumer prices further.
References
[1] International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th edition.
[2] MarketWatch. (2022). Global insulin market analysis.
[3] Evaluate Pharma. (2022). Biosimilar insulin market trends.
[4] U.S. Food and Drug Administration. (2022). Biosimilar approval updates.
[5] European Medicines Agency. (2022). Biosimilar landscape overview.
More… ↓
