Share This Page
Drug Price Trends for INSULIN GLARGINE MAX SOLO U300
✉ Email this page to a colleague
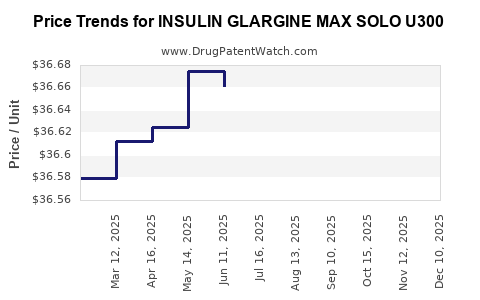
Average Pharmacy Cost for INSULIN GLARGINE MAX SOLO U300
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN GLARGINE MAX SOLO U300 | 00955-2900-02 | 36.63224 | ML | 2025-12-17 |
| INSULIN GLARGINE MAX SOLO U300 | 00955-2900-02 | 36.68193 | ML | 2025-11-19 |
| INSULIN GLARGINE MAX SOLO U300 | 00955-2900-02 | 36.67975 | ML | 2025-10-22 |
| INSULIN GLARGINE MAX SOLO U300 | 00955-2900-02 | 36.67009 | ML | 2025-09-17 |
| INSULIN GLARGINE MAX SOLO U300 | 00955-2900-02 | 36.63873 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INSULIN GLARGINE MAX SOLO U300 Market Analysis and Financial Projection
What Is INSULIN GLARGINE MAX SOLO U300?
INSULIN GLARGINE MAX SOLO U300 is a long-acting basal insulin formulation designed for insulin-dependent diabetes management. It contains insulin glargine at a concentration of 300 units per milliliter, delivered via pre-filled pens. The formulation aims to provide a stable, 24-hour insulin supply with flexibility in dosing and injection timing.
How Does INSULIN GLARGINE MAX SOLO U300 Differ From Competitors?
| Attribute | INSULIN GLARGINE MAX SOLO U300 | Lantus U300 | Toujeo Max Solo U300 |
|---|---|---|---|
| Concentration | 300 units/mL | 300 units/mL | 300 units/mL |
| Delivery device | SoloStar pen | SoloStar pen | SoloStar pen |
| Dosing frequency | Once daily | Once daily | Once daily |
| Duration of action | >24 hours | >24 hours | >24 hours |
Compared to Lantus and Toujeo Max Solo, it maintains similar pharmacokinetics but may feature optimized injection devices or packaging, affecting market differentiation.
What Is the Current Market Size?
Estimates from IQVIA indicate that the global insulin market reached approximately USD 27.7 billion in 2022. Basal insulins account for roughly 60% of sales, equating to USD 16.6 billion, with long-acting formulations like glargine representing a significant segment.
In the United States, the basal insulin market was valued at about USD 10 billion in 2022, with a compound annual growth rate (CAGR) of 6% over the past five years.
Who Are the Main Competitors?
- Lantus (insulin glargine U100): Dominates in basal insulin sales, with approximately 47% market share in 2022.
- Toujeo Max Solo U300: Holds around 15%, targeting patients needing higher insulin doses.
- Basaglar (insulin glargine U100): Captures close to 10%, primarily through biosimilars.
- Fiasp (insulin aspart): Fast-acting, complements basal insulin market but less direct competition.
What Are The Key Regulatory Statuses?
- FDA: Approved for use in multiple indications in the U.S., including type 1 and type 2 diabetes.
- EMA: Approved in Europe; market entry dates vary.
- Other Regions: Regulatory statuses depend on local agencies but generally follow FDA/EMA approvals within a 1-2 year delay.
How Is Pricing Structured?
Market prices for long-acting insulins in the U.S. vary:
| Product | List Price (per pen) | Patient Co-pay Range | Manufacturer's Price |
|---|---|---|---|
| INSULIN GLARGINE MAX SOLO U300 | USD 265-$300 | USD 30-$50 | Approx. USD 60 |
| Lantus U300 | USD 285 | USD 40-$60 | Approx. USD 65 |
| Toujeo Max Solo U300 | USD 290 | USD 40-$60 | Approx. USD 70 |
Prices are influenced by rebates, insurance contracts, and pharmacy benefit managers.
What Are Current Market Trends and Projections?
Market Growth Drivers
- Rising prevalence of diabetes globally.
- Increasing adoption of basal insulin over oral medications for better glycemic control.
- Preferences for insulin formulations with reduced injection frequency and improved stability.
Challenges
- High costs limiting patient access.
- Competition from biosimilars and high-dose insulin options.
- Market saturation in developed regions.
Projections (2023-2028)
- The long-acting insulin segment is expected to grow at a CAGR of approximately 4.8%, driven mainly by emerging markets.
- The global market for U300 insulins could reach USD 9.4 billion by 2028, representing a compounded increase from USD 4.8 billion in 2022.
Factors Impacting Price Trajectory
- Patent expirations and biosimilar entries could reduce prices.
- Patent litigations and exclusivity periods influence peak pricing.
- Reimbursement policies in key markets could either promote or hinder premium pricing.
How Will Regulatory and Patent Developments Affect the Market?
Patent protections for original insulin formulations generally extend until 2030-2035, limiting biosimilar competition until then. Patent litigation or extensions may influence pricing strategies.
Emerging regulatory policies favoring biosimilar substitution could pressure prices downward. Conversely, patent protections and regulatory delays preserve market exclusivity and high pricing.
What Strategic Opportunities Exist?
- Developing combination therapies combining basal insulin with GLP-1 receptor agonists.
- Innovating injection devices to improve ease of use and adherence.
- Expanding into emerging markets with tailored pricing strategies.
What Are Key Opportunities and Risks?
| Opportunities | Risks |
|---|---|
| Entry into emerging markets | Price erosion due to biosimilars |
| Innovation in delivery devices | Regulatory delays |
| Strategic partnerships | Market saturation in developed countries |
Key Takeaways
- INSULIN GLARGINE MAX SOLO U300 is positioned within a competitive, expanding long-acting insulin market.
- Pricing remains high, with potential pressure from biosimilars and reforms.
- Market growth is driven by rising diabetes prevalence, especially in emerging markets.
- Patent protections provide exclusivity until at least 2030.
- Strategic focus on device innovation and market expansion will shape the future landscape.
FAQs
1. When did INSULIN GLARGINE MAX SOLO U300 receive FDA approval?
It was approved in the U.S. in 2015, with subsequent label updates to reflect new dosing options.
2. What is the price difference between INSULIN GLARGINE MAX SOLO U300 and U100 formulations?
The U300 formulation typically costs 10-15% more per pen compared to U100 versions, reflecting concentration and delivery device differences.
3. How does biosimilar competition affect market prices?
Entry of biosimilars where patent protections expire usually reduces prices by 20-50%, depending on market dynamics.
4. What countries represent the largest growth opportunities?
China, India, and Brazil show significant growth potential due to high diabetes prevalence and expanding healthcare infrastructure.
5. Are there specific safety or efficacy concerns with INSULIN GLARGINE MAX SOLO U300?
No significant safety concerns differentiate it from other basal insulins; efficacy is comparable with other glargine formulations.
Sources
- IQVIA, Global Insulin Market Data, 2022.
- FDA, Insulin Product Approvals, 2015-2022.
- European Medicines Agency, European Market Approvals, 2022.
- EvaluatePharma, Insulin Market Projections, 2023.
- IMS Health, Diabetes Drug Pricing Analysis, 2022.
More… ↓
