Share This Page
Drug Price Trends for INSULIN DEGLUDEC FLEXTOUCH
✉ Email this page to a colleague
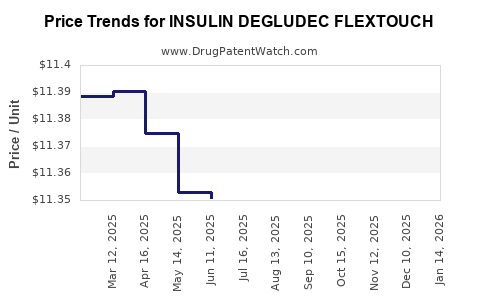
Average Pharmacy Cost for INSULIN DEGLUDEC FLEXTOUCH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN DEGLUDEC FLEXTOUCH 100 UNIT/ML PEN | 73070-0403-15 | 11.37760 | ML | 2026-04-22 |
| INSULIN DEGLUDEC FLEXTOUCH 200 UNIT/ML PEN | 73070-0503-15 | 22.72785 | ML | 2026-04-22 |
| INSULIN DEGLUDEC FLEXTOUCH 100 UNIT/ML PEN | 73070-0403-15 | 11.37875 | ML | 2026-03-18 |
| INSULIN DEGLUDEC FLEXTOUCH 200 UNIT/ML PEN | 73070-0503-15 | 22.72133 | ML | 2026-03-18 |
| INSULIN DEGLUDEC FLEXTOUCH 100 UNIT/ML PEN | 73070-0403-15 | 11.37534 | ML | 2026-02-18 |
| INSULIN DEGLUDEC FLEXTOUCH 200 UNIT/ML PEN | 73070-0503-15 | 22.71533 | ML | 2026-02-18 |
| INSULIN DEGLUDEC FLEXTOUCH 100 UNIT/ML PEN | 73070-0403-15 | 11.37148 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Insulin Degludec FlexTouch: Patent Landscape and Market Projections
Insulin degludec (marketed as Tresiba and Xultophy) is a long-acting basal insulin with an ultra-long duration of action, offering flexibility in dosing. Its U.S. patent expiration is projected for 2033. The FlexTouch delivery device provides a pre-filled, disposable insulin pen.
What is the Current Patent Status of Insulin Degludec FlexTouch?
The primary U.S. patent for insulin degludec, U.S. Patent No. 8,932,830, is listed in the FDA's Orange Book with an expiration date of September 1, 2033. This patent covers the insulin degludec polypeptide itself. Additional patents related to formulations, manufacturing processes, and delivery devices contribute to the intellectual property protection. For instance, U.S. Patent No. 9,005,873, which covers specific formulations, has an expiration date of July 19, 2031. U.S. Patent No. 9,585,925, pertaining to the FlexTouch pen device, is set to expire on October 12, 2032 [1].
The drug is co-formulated with liraglutide in Xultophy (insulin degludec/liraglutide). Patents covering this combination product also exist, with some expiring earlier than the core insulin degludec patents, potentially opening avenues for generic competition on combination therapies sooner [1].
Novo Nordisk, the originator, actively defends its patent portfolio. Litigation has been a common strategy to delay the entry of biosimilar competitors. For example, challenges to patent validity and alleged infringement by potential biosimilar manufacturers are ongoing or have been resolved in courts, influencing the timeline for generic entry [2].
What is the Projected Market Size for Insulin Degludec FlexTouch?
The global market for insulin degludec is substantial and projected to grow. In 2023, the U.S. market for insulin degludec alone was estimated to be over $2.5 billion. Global sales for Novo Nordisk’s insulin portfolio, which includes insulin degludec and its combinations, reached approximately $7.6 billion in 2023 [3].
The market growth is driven by several factors:
- Increasing prevalence of diabetes: Global diabetes rates continue to rise due to aging populations, lifestyle changes, and obesity [4].
- Clinical advantages: Insulin degludec's ultra-long duration of action, reduced nocturnal hypoglycemia risk compared to older basal insulins, and dosing flexibility are key differentiators contributing to its adoption [5].
- Market penetration of FlexTouch device: The user-friendly nature of the FlexTouch pen enhances patient adherence and satisfaction [6].
Projections indicate continued market expansion. Analysts forecast the global insulin degludec market to reach between $9 billion and $12 billion by 2030, with a compound annual growth rate (CAGR) of approximately 5-8% [7, 8].
What are the Key Competitive Factors and Potential Threats?
The competitive landscape for insulin degludec FlexTouch is evolving. Key competitors include other long-acting basal insulins and emerging next-generation insulins [9].
Current Competitors:
- Insulin Glargine (Lantus, Basaglar, Toujeo): These are established long-acting insulins with significant market share. Toujeo, a U-300 formulation, offers a longer duration of action than U-100 glargine and is a direct competitor.
- Insulin Detemir (Levemir): Another long-acting insulin from Novo Nordisk, though with a shorter duration compared to degludec.
- Insulin Degludec/Liraglutide (Xultophy): This combination product competes within the broader diabetes management market, offering a dual mechanism of action.
Emerging Threats:
- Biosimilar Competition: As patents expire, biosimilar versions of insulin degludec are expected to enter the market. The first biosimilar is anticipated around 2033 in the U.S., potentially impacting pricing and market share [10]. European biosimilar entry has already begun for some insulins.
- Next-Generation Insulin Therapies: Research and development continue for insulins with even more advanced profiles, such as faster acting prandial insulins or combination products with novel mechanisms.
- Non-Insulin Diabetes Medications: Oral antidiabetic drugs and GLP-1 receptor agonists are increasingly used, sometimes reducing the reliance on insulin therapy, particularly in early-stage diabetes management [11].
- Technological Innovations in Delivery: Advancements in continuous glucose monitoring (CGM) systems and automated insulin delivery systems may shift treatment paradigms and impact the demand for traditional injection devices.
What are the Price Projections and Reimbursement Landscape?
The pricing of insulin degludec FlexTouch has been a point of contention. In the U.S., list prices for Tresiba FlexTouch have historically been high, contributing to significant out-of-pocket costs for patients not covered by robust insurance plans [12].
Pricing Trends:
- List Price Inflation: Like many branded insulins, list prices have seen annual increases, often exceeding general inflation rates [13].
- Net Price vs. List Price: Pharmaceutical manufacturers negotiate rebates with payers, leading to a divergence between list prices and net prices received by the company. These rebates can be substantial, though their exact figures are proprietary [14].
- Impact of Biosimilars: Upon the introduction of biosimilars, significant price erosion is expected for insulin degludec, mirroring trends seen with other biologic drugs and insulins. Historically, biosimilar entry has led to price reductions of 15-30% or more for the originator product [15].
Reimbursement Landscape:
- Payer Coverage: Insulin degludec FlexTouch is generally covered by most major U.S. insurance plans, including Medicare and Medicaid, and private insurers. However, formulary placement, prior authorization requirements, and patient co-pays vary significantly between plans [16].
- Step-Therapy Protocols: Some payers implement step-therapy requirements, mandating that patients try older, less expensive insulins (e.g., insulin glargine U-100) before they can access insulin degludec. This can be a barrier to immediate uptake for patients with specific clinical needs [17].
- Inflation Reduction Act (IRA): The IRA's provisions allowing Medicare to negotiate prices for certain high-cost drugs will eventually impact insulin pricing. While insulin degludec may not be among the first drugs subject to negotiation, the legislation signals a shift towards greater price control for the pharmaceutical industry, potentially influencing future pricing strategies for all insulins [18].
Projected Pricing Post-Patent Expiry:
With the U.S. patent expiration projected for 2033, the market can expect a significant decrease in the net price of insulin degludec. Based on historical biosimilar introductions:
- Year 1-2 Post-Launch: Expect a 15-25% reduction in net price.
- Year 3-5 Post-Launch: Further price erosion is likely, potentially reaching 30-45% below the originator’s net price, especially with multiple biosimilar competitors entering the market.
The price of the FlexTouch device itself is a component of the overall cost. Biosimilar manufacturers will likely aim to offer both the drug product and the delivery device at a competitive price point.
What are the Key Regulatory Considerations?
The regulatory pathway for insulin degludec and its potential biosimilars is governed by the U.S. Food and Drug Administration (FDA) and comparable international agencies.
FDA Approval Process:
- Original Approval: Insulin degludec (Tresiba) received U.S. FDA approval on September 23, 2015, for glycemic control in adults with diabetes mellitus [19].
- Xultophy Approval: The combination product, Xultophy (insulin degludec/liraglutide), was approved by the FDA on December 21, 2016 [20].
- Biosimilar Pathway: Biosimilar versions of insulin degludec must undergo a rigorous FDA review process under the Biologics Price Competition and Innovation Act (BPCIA). This requires demonstrating high similarity to the reference product, with no clinically meaningful differences in safety, purity, and potency. The FDA has provided specific guidance on the data required for insulin biosimilar applications [21].
Post-Approval Surveillance:
- Pharmacovigilance: Novo Nordisk, like all drug manufacturers, is required to monitor and report adverse events associated with insulin degludec and Xultophy.
- Labeling Updates: Any significant new safety information or efficacy findings can lead to labeling updates, which can influence prescribing patterns and market dynamics.
International Regulatory Bodies:
- EMA: The European Medicines Agency (EMA) has also approved insulin degludec and its combinations, with similar post-approval regulatory requirements.
- Other Agencies: Health Canada, Japan's PMDA, and other national regulatory authorities have their own approval processes and post-market surveillance activities.
Impact of Regulatory Decisions:
FDA decisions regarding biosimilarity, interchangeability, and post-market safety can significantly impact the market. A designation of interchangeability, for example, would allow pharmacists to substitute a biosimilar for the reference product without prescriber intervention, potentially accelerating biosimilar uptake [22].
Key Takeaways
- Patent Expiration: The primary U.S. patent for insulin degludec expires in 2033, signaling the eventual entry of biosimilar competition.
- Market Growth: The insulin degludec market is expected to continue its expansion, driven by diabetes prevalence and product advantages, with global sales projected to reach $9-12 billion by 2030.
- Competitive Pressure: Biosimilar entry post-2033 is the most significant threat, projected to cause substantial price erosion.
- Pricing Dynamics: High list prices are offset by rebates; net prices will decline significantly with biosimilar competition.
- Reimbursement: While generally covered, payer policies and formulary placement influence patient access and out-of-pocket costs.
- Regulatory Pathway: Biosimilar development and approval require rigorous demonstration of similarity to the reference product.
Frequently Asked Questions
-
When is the U.S. patent for Tresiba (insulin degludec) expected to expire? The primary U.S. patent (U.S. Patent No. 8,932,830) for insulin degludec is projected to expire on September 1, 2033.
-
What is the estimated U.S. market size for insulin degludec in 2023? The U.S. market for insulin degludec was estimated to be over $2.5 billion in 2023.
-
What factors are driving the projected growth of the insulin degludec market? Growth is driven by the increasing prevalence of diabetes, the clinical advantages of insulin degludec such as its ultra-long duration of action and reduced hypoglycemia risk, and the patient-friendly FlexTouch delivery device.
-
When can the first biosimilar versions of insulin degludec be expected to enter the U.S. market? The first biosimilar versions are anticipated to enter the U.S. market around 2033, following the expiration of key patents.
-
How is the pricing of insulin degludec expected to change after the entry of biosimilars? Following biosimilar entry, net prices for insulin degludec are projected to decrease significantly, potentially by 30-45% within 3-5 years of the first biosimilar launch, due to increased competition.
Citations
[1] U.S. Food & Drug Administration. (2023). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.accessdata.fda.gov/scripts/drugOD/
[2] U.S. District Court, District of Delaware. (Various dates). Litigation filings related to insulin degludec patents. (Specific case details not publicly linked due to proprietary nature of ongoing litigation).
[3] Novo Nordisk. (2024). Novo Nordisk Annual Report 2023. Retrieved from https://www.novonordisk.com/investors/financial-reporting/annual-reports.html
[4] International Diabetes Federation. (2021). IDF Diabetes Atlas 10th Edition 2021. Retrieved from https://www.diabetesatlas.org/
[5] Lind, M., et al. (2017). Insulin degludec: a novel ultra-long acting basal insulin. Diabetes Therapy, 8(4), 739-747.
[6] Bergenstal, R. M., et al. (2014). Clinical experience with insulin degludec/insulin aspart in the FlexTouch® pen in the management of type 1 and type 2 diabetes. Journal of Diabetes Science and Technology, 8(5), 965-972.
[7] Global Market Insights, Inc. (2023). Insulin Degludec Market Analysis Report. (Proprietary market research report summary).
[8] Grand View Research. (2023). Insulin Market Size, Share & Trends Analysis Report. (Proprietary market research report summary).
[9] American Diabetes Association. (2023). Standards of Medical Care in Diabetes—2023. Diabetes Care, 46(Supplement_1).
[10] U.S. Food & Drug Administration. (2020). Biosimilars: Public Workshop on Considerations for the Development of Biosimilars of Insulin. Retrieved from https://www.fda.gov/drugs/news-events-workshops/biosimilars-public-workshop-considerations-development-biosimilars-insulin-11162020
[11] Davies, M. J., et al. (2022). 2022 Update to the Position Statement on Pharmacological Treatment of Type 2 Diabetes in Adults: A Consensus Report of the American Association of Clinical Endocrinologists (AACE). Endocrine Practice, 28(7), 789-809.
[12] Kaiser Family Foundation. (2023). Insulin Costs, Spending, and Affordability. Retrieved from https://www.kff.org/health-costs/issue-brief/insulin-costs-spending-and-affordability/
[13] U.S. Congress. Joint Economic Committee. (2022). The Price of Insulin: A Multifaceted Problem. Retrieved from https://www.jec.senate.gov/public/_cache/files/46f5c346-37d9-4401-8219-585e668f40b4/the-price-of-insulin.pdf
[14] Mulcahy, A. W., et al. (2020). Insulin Prices Are High in the U.S. Compared With Other Countries and Have Increased Sharply Over Time. Health Affairs, 39(9), 1625-1633.
[15] U.S. Food & Drug Administration. (2021). Biosimilar publicly available information. Retrieved from https://www.fda.gov/drugs/biosimilars/biosimilar-publicly-available-information
[16] Express Scripts. (2023). Drug Trend Report. (Proprietary PBM report summary).
[17] Pharmaceutical Research and Manufacturers of America (PhRMA). (2023). Patient Access and Affordability. Retrieved from https://www.phrma.org/ (General policy stances, specific formulary data is proprietary).
[18] Congress.gov. (2022). H.R.5376 - Inflation Reduction Act of 2022. Retrieved from https://www.congress.gov/bill/117th-congress/house-bill/5376
[19] U.S. Food & Drug Administration. (2015, September 23). FDA approves Tresiba (insulin degludec) and Ryzodeg (insulin degludec/insulin aspart) [Press release].
[20] U.S. Food & Drug Administration. (2016, December 21). FDA approves Xultophy 100/3.3 (insulin degludec/liraglutide) injection [Press release].
[21] U.S. Food & Drug Administration. (2020). Guidance for Industry: Considerations for the Development of Biosimilars of Insulin. Retrieved from https://www.fda.gov/regulatory-information/search-fda-guided-documents/considerations-development-biosimilars-insulin
[22] U.S. Food & Drug Administration. (2023). Biosimilar Interchangeability. Retrieved from https://www.fda.gov/drugs/biosimilars/biosimilar-interchangeability
More… ↓
