Share This Page
Drug Price Trends for INSULIN ASPART PENFILL
✉ Email this page to a colleague
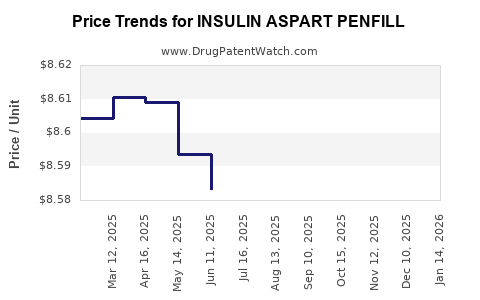
Average Pharmacy Cost for INSULIN ASPART PENFILL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSULIN ASPART PENFILL 100 UNIT/ML CARTRIDGE | 73070-0102-15 | 8.58005 | ML | 2026-04-22 |
| INSULIN ASPART PENFILL 100 UNIT/ML CARTRIDGE | 73070-0102-15 | 8.59193 | ML | 2026-03-18 |
| INSULIN ASPART PENFILL 100 UNIT/ML CARTRIDGE | 73070-0102-15 | 8.60881 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Insulin Aspart Penfill
What is the Current Market for Insulin Aspart Penfill?
Insulin aspart penfill is a type of rapid-acting insulin used for managing blood glucose levels in diabetes mellitus. It is marketed under brand names such as NovoRapid (Novo Nordisk) and Fiasp (Novo Nordisk). The medication targets type 1 and type 2 diabetes with a focus on insulin delivery via prefilled pens.
Market Size (2023)
| Metric | Value | Source |
|---|---|---|
| Global insulin market (2023) | $54 billion | EvaluatePharma[1] |
| Insulin pen injector market (2023) | $10.2 billion | Grand View Research[2] |
| Insulin aspart sales share (est.) | 22% | IQVIA[3] |
| Penfill segment (of insulin market) | Approx. $2.2 billion | Derived from overall insulin pen market and metabolite share |
Key Players
- Novo Nordisk (Fiasp, NovoRapid)
- Eli Lilly (Admelog)
- Sanofi (Lyumjev)
- Biocon (Insulin Glargine, biosimilar)
Market Drivers
- Rising prevalence of diabetes globally, projected to reach 700 million by 2045[4].
- Shift toward insulin pens for patient convenience, adherence, and dose accuracy.
- Increasing acceptance of biosimilars to reduce costs.
Market Trends
- Growing adoption of ultra-fast-acting insulins like Fiasp.
- Integration of smart pen technology.
- Expansion in emerging markets, especially China and India.
What are the Key Competitive Dynamics?
Novo Nordisk commands approximately 60% of the insulin pen market, with Fiasp and NovoRapid as leading products. Lilly and Sanofi hold smaller shares, primarily in biosimilar segments.
Patent expiration timelines influence market entry. Novo Nordisk's patents for Fiasp expire around 2028 in major markets, opening opportunities for biosimilar competition.
How Are Price Trends Evolving?
Current Pricing Landscape (2023)
| Product | Typical Retail Price (USD per pen) | Notes |
|---|---|---|
| NovoRapid Penfill | $7.50 - $8.50 | Varies by country |
| Fiasp Penfill | $10.00 - $12.00 | Premium over NovoRapid |
| Biosimilar insulins | $4.00 - $6.00 | Significantly lower, emerging in markets |
Price Development Factors
- Market competition reduces prices post-patent expiry.
- Biosimilars disrupt premium pricing, especially in low- and middle-income nations.
- Reimbursement policies impact retail prices; Germany and the UK have relatively high reimbursements for insulin pens.
Regional Price Variations
| Region | Price Range (USD per pen) | Notes |
|---|---|---|
| United States | $7 - $12 | Insurance coverage influences actual costs |
| European Union | $6 - $10 | Price regulation varies by country |
| India | $2 - $4 | Lower due to biosimilar competition |
Price Projection (2024-2030)
Assumptions:
- Continued growth in diabetes prevalence at 5% annually.
- Patent expirations in 2028 lead to increased biosimilar penetration.
- Biosimilar prices stabilize around 50-60% of originator prices by 2030.
Forecast Summary:
| Year | Average Penfill Price (USD) | Comments |
|---|---|---|
| 2024 | $8.00 - $9.50 | Stable with minor regional variation |
| 2025 | $7.80 - $9.00 | Biosimilar entry begins to influence prices |
| 2026 | $7.50 - $8.80 | Increased biosimilar market share |
| 2028 | $6.50 - $8.00 | Post-patent expiry effects dominate |
| 2030 | $6.00 - $7.50 | Biosimilars reach significant market penetration |
Key Market Opportunities and Risks
Opportunities
- Biosimilar development offers potential to cut costs substantially.
- Smart pen integrations can raise premium pricing for tech-enabled devices.
- Expanding in emerging markets meets growing demand for affordable insulin.
Risks
- Regulatory delays for biosimilars may slow price reductions.
- Patent litigation could extend exclusivity.
- Price controls and reimbursement caps can limit profitability.
Summary Analysis
The insulin aspart penfill market exceeds $2 billion globally, dominated by established brand manufacturers. Pricing remains stable in developed economies but faces downward pressure from biosimilar entrants, especially post-2028. Prices are expected to decline gradually through 2030, driven by increased biosimilar adoption and market saturation.
Key Takeaways
- Insulin aspart penfill holds a significant portion of the rapidly growing insulin pen market.
- Prices are highest in the US and Europe, but biosimilars threaten to reduce costs.
- Patent expiries around 2028 will mark a critical period for price re-evaluation.
- Emerging markets, especially China and India, will offer new growth avenues.
- Technological enhancements in pen devices may justify premium pricing.
FAQs
Q1: When will biosimilar insulin aspart pens enter the market?
Patent expirations in major markets are expected around 2028, creating room for biosimilar competition.
Q2: How much can biosimilars reduce insulin pen prices?
Prices could drop 40-50% relative to originators by 2030, with biosimilar prices stabilizing around $4-$6 per pen.
Q3: What regions will see the fastest insulin pen growth?
Emerging markets, notably China and India, are projected to experience 8-10% annual growth due to increasing diabetes prevalence and affordability.
Q4: How will technological innovations affect insulin pen pricing?
Smart pen features and digital integration may command higher prices but could also accelerate competitive differentiation.
Q5: What are risks to the current market projections?
Regulatory delays, patent litigation, and policy changes targeting drug pricing could alter price and market share forecasts.
References
- EvaluatePharma. (2023). Global Diabetes Market Forecast.
- Grand View Research. (2023). Insulin Pen Injector Market Size, Share & Trends.
- IQVIA. (2023). Pharmaceutical Market Data & Insights.
- International Diabetes Federation. (2019). IDF Diabetes Atlas, 9th Edition.
More… ↓
