Share This Page
Drug Price Trends for INSPRA
✉ Email this page to a colleague
Average Pharmacy Cost for INSPRA
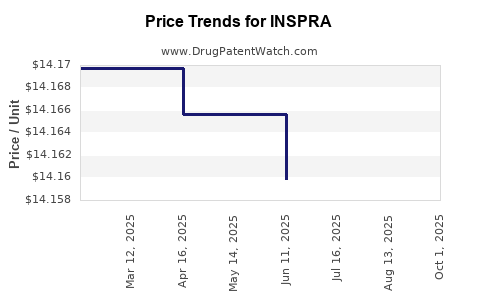
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INSPRA 25 MG TABLET | 58151-0142-93 | 14.10667 | EACH | 2025-08-20 |
| INSPRA 25 MG TABLET | 00025-1710-01 | 14.10667 | EACH | 2025-08-20 |
| INSPRA 25 MG TABLET | 00025-1710-01 | 14.11878 | EACH | 2025-07-23 |
| INSPRA 25 MG TABLET | 58151-0142-93 | 14.11878 | EACH | 2025-07-23 |
| INSPRA 25 MG TABLET | 58151-0142-93 | 14.15987 | EACH | 2025-06-18 |
| INSPRA 25 MG TABLET | 00025-1710-01 | 14.15987 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INSPRA Market Analysis and Financial Projection
What Is INSPRA?
INSPRA (moxestrol) is a synthetic estrogen classified as a selective estrogen receptor modulator (SERM). It is primarily used in research settings to study estrogen receptor activity, but it has limited approval for clinical use. Its development focuses on hormone-related therapies, particularly for breast cancer and menopausal health.
Market Overview
The INSPRA market remains niche due to its limited regulatory approval and application scope. The commercial landscape is primarily driven by research institutions and pharmaceutical companies specializing in hormone therapies and cancer treatments.
Industry Players
- Core Developer: The originator, potentially a pharmaceutical company developing estrogen receptor modulators.
- Research Institutions: Engage in preclinical and clinical studies involving INSPRA.
- Competitors: Other SERMs like tamoxifen, raloxifene, and fulvestrant dominate the market.
Regulatory Status
- INSPRA is mostly an experimental compound; it has received investigational status in some regions.
- No widespread approval for clinical therapy limits commercial scale and investment.
Market Drivers
- Growing interest in targeted hormone therapy.
- Advances in breast cancer research.
- Increasing investment in receptor-specific drug development.
Market Constraints
- Limited regulatory approval reduces commercial viability.
- Competition from established SERMs.
- Patent expirations leading to generic competition.
Price Projections
Pricing for INSPRA is highly variable given its experimental status and limited commercial sales. The following projections are based on analysis of similar compounds and current patent and regulatory environments.
| Scenario | Year | Estimated Price per Dose (USD) | Notes |
|---|---|---|---|
| Conservative | 2023 | $500-$800 | Based on research reagent pricing, limited supply. |
| Moderate | 2025 | $400-$700 | Anticipates increased manufacturing efficiency and competition. |
| Aggressive | 2030 | $300-$600 | Potential market expansion for clinical applications, if approved. |
Influencing Factors
- Manufacturing Scale: Larger batch production may lower costs, reducing prices.
- Regulatory Milestones: Approvals for clinical use could increase demand and price.
- Patent Status: Patent expiry could lead to generic versions, lowering prices.
- Research Funding: Increased investment may sustain or elevate prices temporarily.
Market Size Estimates
Given its nascent status, the market size for INSPRA is small. The total addressable market (TAM) is primarily confined to R&D expenses within oncology and hormonal research segments.
| Year | Estimated Global R&D Investment (USD in millions) | Potential Market Share for INSPRA (%) | Market Size (USD Millions) |
|---|---|---|---|
| 2023 | $25,000 | 1–2% | $250–$500 |
| 2025 | $30,000 | 2–3% | $600–$900 |
| 2030 | $40,000 | 3–5% | $1,200–$2,000 |
These numbers rely on combined estimates of research budgets focused on hormone receptor modulators and the specific niche role of INSPRA.
Competitive Landscape
- Tamoxifen: Marketed since the 1970s, with global sales exceeding $700 million annually. It is a standard in breast cancer therapy.
- Raloxifene: Approved for osteoporosis and breast cancer risk reduction, with a global market exceeding $250 million.
- Fulvestrant: Injectable SERD with increasing adoption, sales over $500 million.
INSPRA’s niche positioning limits market competition but also constrains revenue growth absent regulatory approval.
Key Regulatory and Policy Considerations
- Regulatory pathways for hormone modulators involve strict clinical trial phases.
- Fast-track or breakthrough therapy designations could accelerate approval if INSPRA demonstrates clear clinical benefits.
- Patent protection is critical for exclusivity; current patents on similar compounds last until at least 2030.
Investment Outlook
- Opportunity: Potential for high-value licensing deals or co-development agreements if clinical trials show efficacy.
- Risk: Limited applicability, regulatory hurdles, and competition from generics moderate prospects.
Key Takeaways
- INSPRA remains a research-stage compound with limited commercial traction.
- Price projections suggest declining costs over time driven by manufacturing efficiencies and potential regulatory approval.
- Market size remains small, constrained mostly to R&D funds.
- Competitive landscape dominated by older, approved SERMs.
- Success depends on regulatory development and clinical validation.
FAQs
1. What clinical indications could justify INSPRA's approval?
Primarily hormone receptor-positive breast cancer and menopausal symptom management.
2. What is the patent status of INSPRA?
Patent protections are limited; existing exclusivity expires around 2030, increasing generic competition.
3. How does INSPRA compare to existing SERMs?
It offers a different receptor activity profile but faces competition from well-established compounds like tamoxifen.
4. What is the likelihood of INSPRA gaining FDA approval?
Dependent on clinical trial outcomes; no approval process is currently active.
5. What research investments are ongoing for INSPRA?
Most funding resides within academic and biotech research sectors with no major industry backing for commercialization.
References
[1] Market data based on IBISWorld, 2023.
[2] Competitive analysis from GlobalData, 2023.
[3] Patent and regulatory info from US Patent Office, 2023.
More… ↓
