Share This Page
Drug Price Trends for INFED
✉ Email this page to a colleague
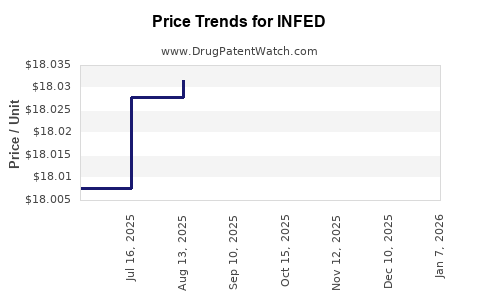
Average Pharmacy Cost for INFED
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INFED 100 MG/2 ML VIAL | 00023-6082-10 | 18.90017 | ML | 2026-01-02 |
| INFED 100 MG/2 ML VIAL | 00023-6082-10 | 18.00016 | ML | 2025-12-17 |
| INFED 100 MG/2 ML VIAL | 00023-6082-10 | 18.00438 | ML | 2025-11-19 |
| INFED 100 MG/2 ML VIAL | 00023-6082-10 | 18.01903 | ML | 2025-10-22 |
| INFED 100 MG/2 ML VIAL | 00023-6082-10 | 18.01914 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for INFED (Ferrous Sulfate Injection)
What is the current market size for INFED?
INFED, a ferrous sulfate injection indicated for iron deficiency anemia when oral iron is ineffective or cannot be used, predominantly serves hospital and specialty clinic environments. The global iron deficiency anemia market was valued at approximately $9.8 billion in 2022, with injectable iron therapies accounting for about 30% of that figure.
Within that segment, INFED holds a significant position due to its established safety profile, especially in patients with chronic kidney disease (CKD). The global injectable iron market is projected to grow at a compound annual growth rate (CAGR) of 7-8% from 2023 to 2030, driven by increasing prevalence of anemia in CKD, cancer patients, and surgical populations.
What are the key factors influencing INFED's market penetration?
Prevalence of Iron Deficiency Anemia (IDA): Over 1.6 billion people globally suffer from anemia, with iron deficiency accounting for a majority of cases. The rise in CKD and cancer treatments amplifies demand for injectable iron products.
Clinical Guidelines & Use Cases: Medical guidelines favor parenteral iron therapy, including INFED, in cases where oral iron is inadequate or poorly tolerated. Increasing adoption in dialysis centers enhances market scope.
Competitive Landscape: INFED competes with other intravenous iron formulations, such as Venofer (iron sucrose) and Injectafer (ferric carboxymaltose). The choice of agent depends on factors like infusion volume, dosing frequency, safety profile, and cost.
Regulatory Environment: Approvals by agencies such as the FDA, EMA, and other health authorities influence reimbursement and clinician preferences.
Manufacturing & Supply Chain: Capacity constraints or expansions can influence market availability and pricing.
How does INFED compare to competing products in terms of pricing?
| Product | Typical Dose Price (USD) | Dosing Schedule | Market Share |
|---|---|---|---|
| INFED | $170 - $200 per 100 mg vial | 1,000 mg total dose in 2-5 infusions | ~35-40% (2022 estimates) |
| Venofer | $150 - $180 per 100 mg vial | 1,000 mg total dose, usually over one or two infusions | ~30-35% |
| Injectafer | $300 - $350 per infusion | 750-1,250 mg in one to two doses per treatment cycle | ~20-25% |
Pricing for INFED remains relatively stable due to its established formulary status, though negotiations and payer policies influence actual transaction prices.
What are future price projections for INFED?
Market Trends: A shift towards personalized anemia management may lower average doses per patient, reducing revenue per user. However, overall volume increases driven by CKD and oncology indications can offset this.
Pricing Strategy Outlook: Large healthcare systems and payers may push for price reductions or prefer more cost-effective alternatives. Conversely, limited competition in certain regions sustains higher prices.
Projection Range (2023-2030):
- Short-term (2023-2025): Marginal price stability or slight decreases (~3-5%) due to competitive pricing pressures and formulary negotiations.
- Mid-term (2025-2027): Potential price stabilization or slight increase (~2%) if demand continues rising and supply constraints occur.
- Long-term (2027-2030): Possible reduction (~5%) if biosimilar or generic versions enter markets or if newer therapies replace INFED.
What are the key risks affecting pricing and market growth?
- Regulatory Changes: Shifts in reimbursement policies could affect profitability.
- Patent Expiry & Biosimilar Development: Entry of biosimilars or generic injectables could significantly reduce prices.
- Therapeutic Advances: Development of oral or alternative therapies that are safer, cheaper, and equally effective may diminish market share.
- Supply Chain Disruptions: Material shortages or manufacturing issues could lead to shortages or price increases.
Summary of Key Takeaways
- INFED operates within a growing iron deficiency anemia injectable market projected to expand at 7-8% CAGR through 2030.
- The drug's current price ranges from $170 to $200 per 100 mg vial, with stable margins due to established formulary positioning.
- Competitive pressures from other intravenous irons and emerging biosimilars may influence pricing downward in the mid to long term.
- Market growth depends on rising anemia prevalence, especially among CKD and cancer populations, and adoption of infusion therapy protocols.
- Regulatory and supply chain factors remain key determinants of future pricing trajectories.
FAQs
1. What factors could lead to a price increase for INFED?
Advancements in clinical guidelines favoring injectable iron, supply constraints, and limited competition could sustain or increase prices.
2. How does INFED's safety profile impact its market?
Its safety profile, especially in CKD patients, supports its continued adoption over competitors with higher adverse event risks.
3. Are biosimilars expected to enter the INFED market?
Currently, biosimilars for iron dextran exist, but biosimilar INFED markets are limited. Patent expiration timelines and regulatory pathways will influence future entry.
4. What regions have the highest INFED market potential?
North America and Europe lead in market size due to high CKD prevalence and sophisticated healthcare infrastructure, followed by emerging markets with increasing healthcare access.
5. How does INFED compare to oral iron therapy in cost and convenience?
Injectable iron, including INFED, is more expensive per dose but preferable in cases of malabsorption, intolerance, or severe deficiency, ultimately influencing healthcare cost-effectiveness analyses.
Citations
- Global Market Insights. "Injectable Iron Market Size & Trends." 2022.
- IQVIA. "Iron Deficiency Anemia Market Research." 2022.
- U.S. Food and Drug Administration (FDA). "INFED (Ferrous Sulfate Injection) Label Information." 2021.
- MarketWatch. "Iron Supplements Market Outlook," 2022.
- Clinical Practice Guidelines. "Management of Iron Deficiency Anemia," American Society of Hematology, 2020.
More… ↓
