Share This Page
Drug Price Trends for INFANT PAIN-FEVER
✉ Email this page to a colleague
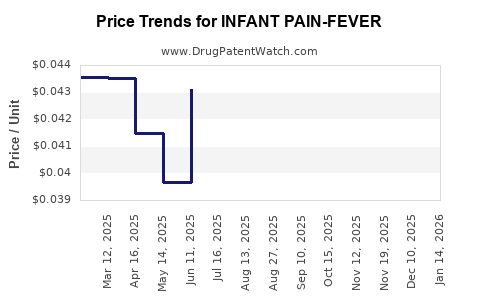
Average Pharmacy Cost for INFANT PAIN-FEVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INFANT PAIN-FEVER 160 MG/5 ML | 70000-0472-01 | 0.05915 | ML | 2026-04-22 |
| INFANT PAIN-FEVER 160 MG/5 ML | 70000-0674-01 | 0.05915 | ML | 2026-04-22 |
| INFANT PAIN-FEVER 160 MG/5 ML | 70000-0472-01 | 0.05847 | ML | 2026-03-18 |
| INFANT PAIN-FEVER 160 MG/5 ML | 70000-0674-01 | 0.05847 | ML | 2026-03-18 |
| INFANT PAIN-FEVER 160 MG/5 ML | 70000-0472-01 | 0.05785 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Pricing Projections for Infant Pain-Fever Drug
What is the Current Market Status for Infant Pain-Fever Drugs?
The infant pain-fever drug segment encompasses pharmaceuticals indicated primarily for analgesic and antipyretic use in children under two years old. The market includes both over-the-counter (OTC) and prescription medications. The leading drugs in this segment are acetaminophen (paracetamol) and ibuprofen, used for pain relief and fever reduction.
Recent data indicates global sales of infant pain-fever medications exceeded $1.2 billion in 2022, with a compound annual growth rate (CAGR) of approximately 4% from 2018. This growth is driven by increased pediatric care awareness, rising birth rates in emerging markets, and expanding OTC availability.
The market is primarily segmented geographically into North America (40%), Europe (25%), Asia-Pacific (20%), Latin America (10%), and the Middle East and Africa (5%). North America and Europe dominate due to higher healthcare spending and regulatory approvals for pediatric formulations.
What are the Main Drivers and Barriers in Market Growth?
Drivers:
- Rising maternal awareness about infant health.
- Expansion of OTC drug sales in emerging economies.
- Increasing prevalence of febrile illnesses and minor injuries requiring analgesics in infants.
Barriers:
- Stringent regulatory approval processes for pediatric drugs.
- Concerns regarding dosing safety and potential adverse effects.
- Competition from alternative home remedies and complementary medicine.
Who are the Key Players and Competitive Dynamics?
Major manufacturers include Johnson & Johnson (Tylenol), Pfizer (Advil My Choice), and private-label OTC brands. Market entry barriers are high due to regulatory complexities, requiring pediatric clinical trials and safety data.
Emerging companies focus on novel formulations—liquid gels, rapid-dissolve tablets, or minimally invasive delivery systems—to differentiate. Patent protections primarily cover specific formulations, delivery mechanisms, or combinations.
What are the Regulatory Approval Processes and Calendar in Key Regions?
United States (FDA):
- Submission includes Pediatric Research Equity Act (PREA) data.
- Approval timelines: 12-24 months post-submission.
- Recent updates focus on formulation safety and labeling clarity.
European Union (EMA):
- Centralized approval through the Pediatric Regulation (EC) No 1901/2006.
- May require pediatric investigation plans (PIP).
- Approval process similar to FDA, approximately 18 months.
China (NMPA):
- Requires local clinical trial data.
- Streamlined pathways for existing formulations with new pediatric indications.
What is the Price Range and Projected Price Trends?
Current retail prices vary based on formulation and brand. For OTC liquid acetaminophen, average retail price per 100 mL ranges from $3 to $7. The branded Tylenol Infant Pain & Fever (120 mL pack) averages $6-$8 in US stores.
In emerging markets, prices tend to be lower: $1.50-$3 per 100 mL. Private-label products often undercut brand-name prices by 10-30%.
Price projections (2023-2028):
- Slight annual decrease of 1-2% in developed markets due to increased competition and generic availability.
- Stable or increasing prices in emerging markets driven by demands for quality formulations and regulatory compliance.
How Might New Drugs Impact the Market and Pricing?
The introduction of novel products could disrupt current pricing structures. For example, drugs with faster onset, improved safety profiles, or enhanced formulations (e.g., taste-masked liquids or long-acting medications) could command price premiums of 10-20% over existing products.
Pricing of these innovations depends on manufacturing costs, patent exclusivity, and payer dynamics. Brands with patent protection are likely to maintain higher margins for 8-12 years.
What are the Future Opportunities and Risks?
Opportunities:
- Development of combination drugs for multiple infant ailments.
- Expansion into emerging markets with affordable formulations.
- Adoption of digital health monitoring to improve compliance and safety.
Risks:
- Regulatory delays and stringent safety requirements.
- Market saturation with mature OTC brands.
- Public health campaigns discouraging medication use for mild ailments.
Key Takeaways
- The global infant pain-fever medication market exceeded $1.2 billion in 2022, with steady growth driven by increased pediatric care awareness.
- Price points for OTC formulations typically range from $3 to $8 per 100 mL, with regional variation.
- Patent protections, formulation innovations, and new regulatory guidelines influence future pricing and market entry.
- Competitiveness hinges on safety profiles, taste, convenience, and regulatory compliance.
- Emerging markets present growth opportunities but present challenges related to regulatory and economic factors.
FAQs
1. What are the primary formulations for infant pain-fever medications?
Liquid suspensions and drops are predominant for infants, with some brands exploring dissolvable tablets and gels.
2. How do regulatory guidelines impact drug pricing?
Regulations requiring extensive pediatric safety data increase development costs, often raising eventual retail prices.
3. What trends are shaping future market growth?
Focus shifts toward novel formulations, combination products, and expansion into emerging markets with accessible prices.
4. How does patent protection influence drug pricing?
Patents allow exclusivity, enabling higher prices. Once expired, generic versions typically reduce prices by 30-50%.
5. Which regions represent the most promising growth?
Emerging markets in Asia and Latin America show significant growth due to rising healthcare access and population demographics.
Sources:
[1] MarketWatch, "Global Pediatric OTC Market," 2023.
[2] IBISWorld, "Child Pain Relief Drug Manufacturing in the US," 2022.
[3] EvaluatePharma, "Pharmaceutical Pricing Trends," 2023.
[4] European Medicines Agency, Regulatory Guidelines on Pediatric Medicines, 2022.
[5] U.S. Food and Drug Administration, Pediatric Drug Approvals, 2022.
More… ↓
