Share This Page
Drug Price Trends for HYOSCYAMINE ER
✉ Email this page to a colleague
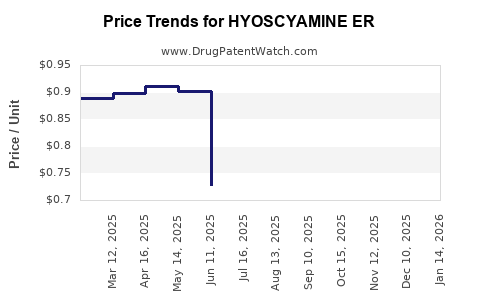
Average Pharmacy Cost for HYOSCYAMINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYOSCYAMINE ER 0.375 MG TAB | 62559-0423-01 | 0.49508 | EACH | 2026-05-20 |
| HYOSCYAMINE ER 0.375 MG TAB | 62559-0423-01 | 0.50334 | EACH | 2026-04-22 |
| HYOSCYAMINE ER 0.375 MG TAB | 62559-0423-01 | 0.53349 | EACH | 2026-03-18 |
| HYOSCYAMINE ER 0.375 MG TAB | 62559-0423-01 | 0.54864 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Hyoscyamine ER Market Analysis and Price Projections
Executive Summary
Hyoscyamine ER, an extended-release formulation of the anticholinergic drug hyoscyamine, is primarily prescribed for the symptomatic relief of gastrointestinal disorders such as irritable bowel syndrome (IBS) and peptic ulcers. Its mechanism of action involves reducing smooth muscle spasms in the gastrointestinal tract. The market for Hyoscyamine ER is characterized by established generic competition, a stable but mature demand profile, and pricing influenced by manufacturing costs, regulatory hurdles for new entrants, and payer negotiations. Projected market growth is modest, with price stability anticipated in the near to medium term, barring significant shifts in manufacturing efficiency or novel therapeutic alternatives.
What is Hyoscyamine ER and What is Its Primary Therapeutic Use?
Hyoscyamine ER is an extended-release oral dosage form of hyoscyamine, a naturally occurring tropane alkaloid. It functions as an antimuscarinic agent, meaning it blocks the action of acetylcholine at muscarinic receptors. In the gastrointestinal tract, acetylcholine mediates smooth muscle contraction. By inhibiting these actions, hyoscyamine ER reduces gastrointestinal motility and spasms [1].
The primary therapeutic indication for Hyoscyamine ER is the symptomatic treatment of functional gastrointestinal disorders, including:
- Irritable Bowel Syndrome (IBS): To alleviate abdominal cramping and pain associated with IBS [1].
- Peptic Ulcers: To reduce gastrointestinal secretions and spasms, aiding in pain management and ulcer healing [2].
- Biliary and Genitourinary Colic: To relieve pain caused by smooth muscle spasms in the bile ducts and urinary tract [1].
What is the Current Market Size and Growth Trajectory for Hyoscyamine ER?
The global market for Hyoscyamine ER is a segment within the broader antispasmodic and gastrointestinal drug market. Precise, publicly disclosed market size figures specifically for Hyoscyamine ER are limited, as it is largely a genericized product. However, industry reports on the antispasmodic market provide a proxy. The global antispasmodics market was valued at approximately USD 21.4 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% to 5.5% through 2030 [3]. Hyoscyamine ER constitutes a sub-segment of this larger market, serving a specific niche for visceral pain management.
Growth for Hyoscyamine ER is expected to be more subdued than the overall antispasmodics market, reflecting its mature status and competition from other therapeutic classes for IBS and GI disorders, such as anticholinergics with different pharmacokinetics or newer drug classes like serotonin receptor antagonists.
Who are the Key Manufacturers and Competitors in the Hyoscyamine ER Market?
The Hyoscyamine ER market is primarily populated by generic drug manufacturers. Key players that have historically manufactured or currently market Hyoscyamine ER products (often under various brand names or as generic formulations) include:
- Major Generic Pharmaceutical Companies: These companies typically have broad portfolios and robust generic manufacturing capabilities. Examples of companies that may produce or have produced hyoscyamine products include Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma [4].
- Specialty Pharmaceutical Companies: Smaller or niche companies may also focus on specific therapeutic areas, including gastrointestinal agents.
- Contract Manufacturing Organizations (CMOs): Some companies may utilize CMOs for the production of their Hyoscyamine ER formulations.
Competition is driven by:
- Price: Generic competition leads to significant price pressure.
- Distribution Networks: Access to pharmacies and healthcare providers is crucial.
- Formulation Development: While Hyoscyamine ER is established, minor improvements in release profiles or tablet characteristics can offer competitive advantages.
- Regulatory Compliance: Maintaining Good Manufacturing Practices (GMP) and meeting FDA or equivalent regulatory standards is a baseline requirement.
The market is characterized by a fragmented landscape of generic suppliers rather than a few dominant brand players.
What are the Regulatory Pathways and Hurdles for Hyoscyamine ER Products?
For new generic versions of Hyoscyamine ER to enter the market, manufacturers must navigate specific regulatory pathways and overcome certain hurdles. In the United States, this primarily involves submitting an Abbreviated New Drug Application (ANDA) to the Food and Drug Administration (FDA) [5].
Key aspects of the regulatory process include:
- Bioequivalence: The most critical requirement for an ANDA is demonstrating bioequivalence to the reference listed drug (RLD). This means proving that the generic product performs in the body in the same way as the approved brand-name drug, with similar rates and extents of absorption. This is typically demonstrated through comparative pharmacokinetic studies [5].
- Manufacturing Standards: Facilities involved in the manufacturing, processing, packaging, and holding of Hyoscyamine ER must comply with current Good Manufacturing Practices (cGMP) as outlined by the FDA [6]. Inspections by the FDA are routine.
- Quality Control: Manufacturers must implement robust quality control systems to ensure the identity, strength, quality, and purity of the drug product. This includes testing of raw materials, in-process controls, and finished product testing.
- Labeling: The labeling of generic products must be substantially the same as the labeling of the RLD, with certain permissible differences for generic drug labeling [5].
- Patent Expiry: Generic entry is contingent upon the expiry of patents and exclusivities held by the innovator drug. The Hatch-Waxman Act provides mechanisms for generic companies to challenge existing patents [7].
Hurdles include the cost and time associated with clinical studies (bioequivalence trials), securing API (Active Pharmaceutical Ingredient) suppliers, navigating FDA review processes, and potential patent litigation if challenging existing patents.
What Factors Influence the Pricing of Hyoscyamine ER?
The pricing of Hyoscyamine ER is influenced by a confluence of economic, regulatory, and market-specific factors. As a widely available generic medication, its price is generally lower than that of branded or novel therapies.
Key pricing determinants include:
- Manufacturing Costs: This is a primary driver for generic drugs. Costs include raw material sourcing (hyoscyamine API), formulation, packaging, labor, energy, and overhead. Economies of scale in production significantly impact per-unit costs.
- Generic Competition: The presence of multiple generic manufacturers competing for market share creates downward pressure on prices. The number of approved ANDAs for a particular product directly correlates with competitive pricing.
- Payer Negotiations and Reimbursement: Pharmacy benefit managers (PBMs) and insurance companies negotiate prices with manufacturers based on formulary placement, rebate programs, and expected patient volume. These negotiations can significantly impact the net price received by the manufacturer.
- Wholesale Acquisition Cost (WAC) and Average Wholesale Price (AWP): These are benchmark prices that influence actual transaction prices paid by pharmacies and distributors, though actual net prices are often lower due to rebates and discounts.
- Regulatory Compliance Costs: Maintaining cGMP standards, undergoing FDA inspections, and submitting regulatory filings represent ongoing costs that are factored into pricing.
- Supply Chain Dynamics: Disruptions in API supply, transportation, or distribution can temporarily affect pricing and availability.
- Demand Stability: The demand for Hyoscyamine ER, while relatively stable for its indications, is not rapidly growing. This mature demand profile prevents significant price increases driven by surging demand.
- Therapeutic Alternatives: The availability of other treatments for IBS and GI disorders (e.g., other antispasmodics, antidiarrheals, antacids, neuromodulators) influences pricing by limiting pricing power.
What are the Projected Price Trends for Hyoscyamine ER?
Given the established generic nature of Hyoscyamine ER and the factors influencing its pricing, the projected price trends are anticipated to be relatively stable with modest fluctuations.
Near-Term (1-3 Years):
- Stability with Minor Declines: Expect continued price stability, with potential for minor price erosion due to ongoing competition among generic manufacturers. Rebate competition and payer pressure will likely keep prices in check.
- Impact of Generic Entry: If new generic entrants emerge or if existing players expand production capacity, this could exert slight downward pressure.
- API Cost Fluctuations: Minor fluctuations in the cost of hyoscyamine API or excipients could lead to small, temporary price adjustments.
Medium-Term (3-5 Years):
- Continued Stability: The market is unlikely to see significant price increases unless there is a substantial disruption in supply or a withdrawal of key manufacturers.
- Consolidation: Potential for market consolidation among smaller generic players could lead to slightly less intense price competition, but the overall impact on pricing is likely to remain marginal.
- Cost Pressures: Increasing manufacturing and regulatory compliance costs could exert slight upward pressure, but these are expected to be absorbed by efficiency gains or minimal price adjustments.
Long-Term (5+ Years):
- Uncertainty Driven by Innovation: Long-term price trends are more susceptible to unforeseen developments. The introduction of a fundamentally new, more effective, or safer treatment for IBS and GI disorders that displaces hyoscyamine's role could lead to a decline in demand and, consequently, pricing.
- Shifts in Healthcare Policy: Changes in reimbursement policies or regulatory frameworks could influence pricing dynamics.
- Manufacturing Technology: Advances in manufacturing efficiency or API synthesis could further reduce production costs, potentially leading to lower prices.
Overall Projection: Hyoscyamine ER is expected to remain a cost-effective generic medication. Its price is unlikely to experience significant growth and will likely remain subject to competitive market forces and payer negotiations. The average wholesale price for Hyoscyamine ER (e.g., 0.375 mg tablets) has historically ranged from approximately $0.20 to $0.80 per tablet, depending on the manufacturer, quantity, and contract terms [8]. This range is anticipated to persist, with net prices after rebates being even lower.
What are the Key Market Drivers and Restraints for Hyoscyamine ER?
Market Drivers:
- Prevalence of Gastrointestinal Disorders: Conditions like IBS are highly prevalent globally, creating a consistent demand for symptomatic relief [9].
- Established Efficacy and Safety Profile: Hyoscyamine has a long history of use, and its efficacy in reducing gastrointestinal spasms is well-documented [1].
- Cost-Effectiveness: As a generic drug, Hyoscyamine ER offers a lower-cost treatment option compared to branded medications or newer therapies, making it attractive to payers and patients with cost sensitivities.
- Physician Prescribing Habits: Physicians familiar with the drug's benefits and tolerability are likely to continue prescribing it for appropriate indications.
- Availability in Multiple Formulations: Extended-release formulations improve patient compliance by reducing dosing frequency compared to immediate-release versions.
Market Restraints:
- Availability of Therapeutic Alternatives: The market for IBS and GI disorders is crowded with numerous treatment options, including other antispasmodics, antidiarrheals, laxatives, dietary interventions, and behavioral therapies [9]. Newer drug classes and biologics for specific GI conditions also compete.
- Side Effect Profile: Anticholinergic side effects, such as dry mouth, blurred vision, constipation, and urinary retention, can limit its use in certain patient populations, particularly the elderly [1].
- Generic Market Saturation: The market for established generics is highly competitive, leading to price erosion and limited opportunities for significant revenue growth.
- Lack of Novel Innovation: There is limited ongoing research and development for new Hyoscyamine ER formulations or significant improvements, as it is a mature drug.
- Stricter Regulatory Scrutiny: Ongoing regulatory oversight for GMP and drug quality can increase compliance costs for manufacturers.
Key Takeaways
- Hyoscyamine ER is a mature generic drug primarily used for gastrointestinal spasms, with stable but modest market growth potential.
- The market is characterized by intense competition among generic manufacturers, driving price stability with potential for minor erosion.
- Regulatory approval for new generic entrants hinges on demonstrating bioequivalence and adhering to strict cGMP standards.
- Pricing is predominantly influenced by manufacturing costs, generic competition, and payer negotiations.
- Future price trends are projected to remain stable, with significant increases unlikely unless supply chain disruptions or major shifts in the therapeutic landscape occur.
Frequently Asked Questions
-
What is the typical dosage range for Hyoscyamine ER in the treatment of IBS? The typical dosage of Hyoscyamine ER for IBS symptoms can range from 0.375 mg to 0.75 mg taken every 12 hours, depending on physician assessment and patient response. Dosing adjustments are common to manage individual symptom severity [1].
-
Are there any significant side effects associated with Hyoscyamine ER that might limit its use? Common side effects are related to its anticholinergic properties and include dry mouth, blurred vision, photophobia, urinary hesitancy, decreased sweating, and constipation. Patients with glaucoma, obstructive uropathy, or severe ulcerative colitis may have contraindications for its use [1].
-
How does Hyoscyamine ER differ from immediate-release hyoscyamine formulations? Hyoscyamine ER is designed for sustained release of the active ingredient over a longer period, typically 12 hours. This contrasts with immediate-release formulations, which provide a rapid onset of action but require more frequent dosing (e.g., every 4-6 hours), potentially leading to less consistent symptom control and compliance issues [1].
-
What is the role of Pharmacy Benefit Managers (PBMs) in the pricing of Hyoscyamine ER? PBMs play a significant role by negotiating drug prices and rebates with manufacturers on behalf of health insurance plans. They influence formulary placement, which impacts the volume of prescriptions for Hyoscyamine ER and ultimately the net price realized by manufacturers after discounts and rebates are applied.
-
Can Hyoscyamine ER be used to treat acute, severe abdominal pain from non-GI sources? While Hyoscyamine ER is effective in reducing smooth muscle spasms, its primary indication is for functional gastrointestinal disorders and related genitourinary or biliary colic. It is not typically recommended for acute, severe abdominal pain of unknown origin, as it may mask symptoms of more serious conditions requiring urgent diagnosis and treatment.
Citations
[1] U.S. Food & Drug Administration. (n.d.). DailyMed. Retrieved from https://dailymed.nlm.nih.gov/dailymed/ (Specific drug labels accessed via search for "Hyoscyamine ER")
[2] Klaber, B. R. (2015). The Medical Information Pocketbook. Radcliffe Publishing.
[3] Grand View Research. (2023). Antispasmodics Market Size, Share & Trends Analysis Report.
[4] F. Hoffmann-La Roche AG. (n.d.). Company Reports & Investor Relations. (Information on historical product lines and competitors often found in annual reports and investor presentations for major pharmaceutical entities).
[5] U.S. Food & Drug Administration. (2017). Guidance for Industry: ANDA Submissions - Refuse-to-Receive Criteria.
[6] U.S. Food & Drug Administration. (2023). Current Good Manufacturing Practice (cGMP) for Pharmaceuticals.
[7] U.S. Food & Drug Administration. (2012). Hatch-Waxman Act - Generic Drug Provisions.
[8] First Databank, Inc. (n.d.). Drug Pricing Data. (Accessed via subscription-based pharmaceutical pricing databases).
[9] National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Irritable Bowel Syndrome. National Institutes of Health.
More… ↓
