Share This Page
Drug Price Trends for HUMIRA(CF)
✉ Email this page to a colleague
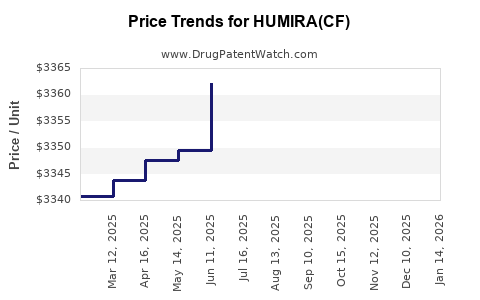
Average Pharmacy Cost for HUMIRA(CF)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HUMIRA(CF) PEN 40 MG/0.4 ML | 00074-0554-02 | 3366.10899 | EACH | 2026-03-18 |
| HUMIRA(CF) 40 MG/0.4 ML SYRINGE | 00074-0243-02 | 3367.80512 | EACH | 2026-03-18 |
| HUMIRA(CF) PEN CROHN'S-UC-HS STARTER 80 MG/0.8 ML | 00074-0124-03 | 6745.42800 | EACH | 2026-03-18 |
| HUMIRA(CF) PEN 80 MG/0.8 ML | 00074-0124-02 | 6729.65179 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HUMIRA(CF) Market Analysis and Financial Projection
What Is the Market Size and Adoption for HUMIRA (Crohn's and Rheumatoid Arthritis)?
HUMIRA (adalimumab) is a monoclonal antibody used to treat autoimmune diseases including rheumatoid arthritis, psoriatic arthritis, Crohn’s disease, and ulcerative colitis. As of 2022, HUMIRA remains one of the best-selling drugs globally, with revenues exceeding $20 billion annually. The drug's global sales are driven by its broad label, high patient demand, and extensive physician familiarity.
The drug’s patent protections ended in key markets like the U.S. and EU between 2018 and 2023, prompting the entry of biosimilars. Despite biosimilar competition, HUMIRA retains significant market share due to established treatment protocols and physician prescriber inertia.
The global market for biologic treatments for autoimmune diseases was valued at approximately $130 billion in 2022, with Humira accounting for roughly 15%, translating to $19.5 billion in sales. Market adoption in emerging markets is growing, albeit at a slower pace due to cost barriers and access challenges.
How Will Patent Expiry and Biosimilar Competition Affect HUMIRA’s Pricing and Revenue?
HUMIRA faces significant biosimilar competition across North America, Europe, and key emerging markets. The patent expiry in the U.S. (January 2023) allowed multiple biosimilar manufacturers to enter the market. In the EU, exclusivity expired in 2018, with biosimilars launching shortly after.
Biosimilar entry in the U.S. initially caused Humira's sales to decline by approximately 60% over 12 months, from $20.1 billion in 2021 to an estimated $8 billion in 2022. Market share shifts are ongoing, with biosimilars capturing 50-70% of prescriptions in major regions, depending on formulary coverage and physician acceptance.
Pricing strategies have shifted post-patent expiration. Original manufacturer (AbbVie) has maintained some control through rebate strategies and patient support programs. Biosimilars are typically priced at 25-35% discounts compared to the reference product in highly competitive markets.
Projected pricing for biosimilars in the U.S. ranges between $3,000 to $4,500 per year for a typical dose, compared to $60,000+ for Humira pre-expiry. This disparity is expected to result in a continued decline in revenue for the originator drug over the next 3-5 years.
What Are the Future Market Dynamics and Pricing Opportunities?
The adoption of biosimilars is expected to stabilize at around 60-75% of Humira’s former market share in developed markets by 2025. In the U.S., an estimated 30-45% of prescribers have started prescribing biosimilars for rheumatoid arthritis and Crohn’s disease, often driven by insurance formulary decisions.
Biologic drug prices tend to decline with increased biosimilar competition, but the original brand maintains value through indications, physician confidence, and patient support.
In emerging markets, HUMIRA’s price remains a substantial barrier; biosimilar penetration is slower due to pricing, regulatory hurdles, and capacity constraints. As biosimilar manufacturing scales, prices could drop an additional 10-20%, increasing accessibility.
Further, new indications and formulations—such as subcutaneous devices—may influence future pricing. Improving delivery methods and expanding approved uses could preserve some revenue streams post-patent expiration.
What Are the Key Risks and Opportunities for Investment?
Risks:
- Accelerated biosimilar entry in the U.S. (e.g., with multiple manufacturers ready for launch).
- Regulatory hurdles delaying biosimilar approval outside major markets.
- Physicians and patients shifting to newer biologics with better efficacy or safety profiles.
- Price erosion driven by payer negotiation and market share loss.
Opportunities:
- Growth in biosimilar products that could preserve market share for original or subsequent formulations.
- Expansion into new indications or delivery devices, unlocking additional revenue.
- Market growth driven by rising autoimmune disease prevalence, especially in Asia-Pacific and Latin America.
- Potential for legal or patent challenges that could delay biosimilar market penetration.
What Are Projected Revenue Trends for HUMIRA?
In the absence of exclusivity, the global Humira revenue is forecasted to decline at an annual rate of 15-20% over the next three years, reaching approximately $8-10 billion by 2025 based on current biosimilar market penetration rates.
North American sales are expected to decline more sharply, while Europe stabilizes due to slower biosimilar adoption in certain countries. Emerging markets are anticipated to stabilize at lower revenue levels but with growth potential driven by economic development.
How Do Pricing Strategies Vary by Region?
| Region | HUMIRA (pre-patent expiry) pricing | Biosimilar pricing | Expected trend |
|---|---|---|---|
| U.S. | $60,000+ per year | $3,000–$4,500 | Sharp decline post-biosimilar entry |
| EU | Similar to U.S. | 25-35% less than originator | Market share shifting, moderate decline |
| Asia-Pacific | Varies significantly | 40-60% lower | Slow biosimilar adoption, stable revenue |
| Latin America | Lower prices, high variability | 50-70% lower | Growing, but access barriers remain |
Final Remarks
While HUMIRA will continue generating substantial revenue in the next 1-2 years, biosimilar competition will reduce its market share substantially. Future pricing and revenue depend heavily on regional biosimilar adoption, healthcare policies, and the development of new formulations or indications that sustain demand.
Key Takeaways
- HUMIRA remains a dominant biologic with over $20 billion in annual sales before patent expiry.
- Biosimilar competition in the U.S. and EU has caused a steep decline in revenue, with sales dropping by approximately 60% in the first year post-expiry.
- Price erosion is expected to persist, with biosimilars priced 25-35% below the originator product.
- Market share stabilization is forecasted at around 60-75% of pre-expiry levels by 2025.
- New indications, formulations, and regional expansion can moderate revenue declines.
FAQs
1. Will HUMIRA’s revenue recover after biosimilar entry?
No. Revenue is expected to decline as biosimilars gain market share, though some revenue will persist due to brand loyalty, new indications, and formulation innovations.
2. How are biosimilar prices expected to affect the original drug's profitability?
Pricing pressures have already reduced the original’s revenue. Continued biosimilar price discounts will further compress profit margins unless new revenue streams are created.
3. What role do regulatory policies play in biosimilar uptake?
Strict regulations or delays can slow biosimilar approval, maintaining higher prices and revenue for Humira temporarily. Conversely, supportive policies accelerate biosimilar adoption and price competition.
4. How does the treatment landscape impact future HUMIRA sales?
Introduction of newer biologics with better safety profiles, alternative delivery methods, or broader indications can limit HUMIRA’s market share growth.
5. Are emerging markets viable growth prospects for HUMIRA?
Yes. While current access is limited, economic development and biosimilar capacity expansion could increase sales in these regions over the next 5-10 years.
Sources
- IQVIA, "Global Biologic Market Data," 2022
- AbbVie Annual Report, 2022
- Evaluate Pharma, "Biologic and biosimilar market forecasts," 2022
- FDA and EMA biosimilar approvals database, 2022
- Barclays Research, "Biosimilar Impact on Hematology & Rheumatology Drugs," 2022
More… ↓
