Share This Page
Drug Price Trends for HUMIRA
✉ Email this page to a colleague
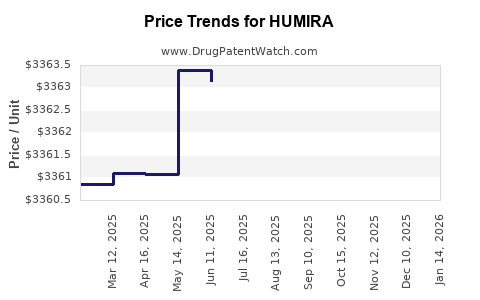
Average Pharmacy Cost for HUMIRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HUMIRA(CF) PEN 40 MG/0.4 ML | 00074-0554-02 | 3366.10899 | EACH | 2026-03-18 |
| HUMIRA(CF) PEN CROHN'S-UC-HS STARTER 80 MG/0.8 ML | 00074-0124-03 | 6745.42800 | EACH | 2026-03-18 |
| HUMIRA 40 MG/0.8 ML SYRINGE | 00074-3799-02 | 3374.01850 | EACH | 2026-03-18 |
| HUMIRA(CF) PEN 80 MG/0.8 ML | 00074-0124-02 | 6729.65179 | EACH | 2026-03-18 |
| HUMIRA PEN 40 MG/0.8 ML | 00074-4339-02 | 3366.38277 | EACH | 2026-03-18 |
| HUMIRA(CF) 40 MG/0.4 ML SYRINGE | 00074-0243-02 | 3367.80512 | EACH | 2026-03-18 |
| HUMIRA(CF) 20 MG/0.2 ML SYRINGE | 00074-0616-02 | 3366.12300 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HUMIRA Market Analysis and Financial Projection
What is the current market landscape for HUMIRA?
HUMIRA (adalimumab) remains one of the top-selling biopharmaceuticals globally. In 2022, it generated approximately $20.4 billion in worldwide revenue, maintaining its position as a dominant biologic for various autoimmune conditions. Its primary indications include rheumatoid arthritis, psoriatic arthritis, Crohn's disease, ulcerative colitis, hidradenitis suppurativa, and plaque psoriasis.
Manufactured by AbbVie, HUMIRA's sales are concentrated predominantly in North America, accounting for nearly 60% of total revenue, with the U.S. alone contributing approximately $12.2 billion in 2022.
How is the competitive landscape evolving?
The market for biologic drugs treating autoimmune diseases faces increasing competition, driven by biosimilars and novel therapeutics.
-
Biosimilar Entry: Multiple biosimilars for HUMIRA launched in several countries starting in 2023. For example:
- Amgen's Amjevita (adalimumab-atto)
- Samsung Bioepis' Imraldi
- Biocon's Abevmy These biosimilars are priced approximately 15-30% below the original, impacting HUMIRA's market share.
-
Global Approvals: Several biosimilars have received regulatory approval in Europe (EMA approvals began in 2018) and other regions, with more expected as patents expire.
-
Patent Expiry: The U.S. patent for HUMIRA expired in January 2023. Patent protections in Europe began to lapse in 2016 but faced legal challenges, leading to delayed biosimilar entry. The expiration has increased price competition, especially in North America.
-
Innovative Alternatives: New drugs like Skyrizi (risankizumab), Stelara (ustekinumab), and Tremfya (guselkumab) target similar pathways and are gaining market share due to improved efficacy or dosing convenience.
What are the pricing dynamics?
HUMIRA's price in the U.S. averaged $2,600-$3,100 per month per patient in 2022, depending on dosing and formulation. Biosimilars are generally priced 20-30% lower, with some as low as $2,200 per month.
-
List Price vs. Net Price: List prices are higher than net prices due to rebates, discounts, and negotiation. Payers often negotiate substantial rebates, reducing the effective expenditure for insurers.
-
Historical Pricing Trends: Since launch in 2003, HUMIRA's list price has increased annually, averaging about 8-10%, outpacing inflation and many rival drugs' price increases.
-
Impact of Biosimilar Entry: The introduction of biosimilars in 2023 has led to a decline in HUMIRA's U.S. sales growth rate from high-single digits to mid-single digits, with some analysts estimating a 20-30% reduction in revenue over the next three years.
What are the forecasts for HUMIRA's future revenue?
Analysts project a decline due to biosimilar competition.
| Year | Projected HUMIRA Revenue | Source/Notes |
|---|---|---|
| 2023 | $15.4 billion | Decline begins with biosimilar competition (FactSet consensus) |
| 2024 | $12.8 billion | Further erosion expected as biosimilar market penetration increases |
| 2025 | $10.6 billion | Continued pressure, but steady demand in certain indications |
| 2026 | $8.9 billion | Stabilization expected; biosimilar sales gain momentum |
AbbVie anticipates that combined sales of HUMIRA and its biosimilars will decrease overall revenue but expects growth from newer indications and pipeline drugs.
Are there regulatory or patent developments impacting projections?
- Patent expirations in key markets accelerate biosimilar entry.
- FDA has approved multiple HUMIRA biosimilars since 2023.
- Patent challenges and legal disputes in Europe have delayed some biosimilar launches.
- Absent patent protections, sales could decrease further unless the company shifts focus to new drugs.
What is the outlook for pricing strategies?
- Price competition will intensify, putting downward pressure on list prices.
- Payer negotiations could further reduce net prices.
- AbbVie's response involves expansion into new indications, development of next-generation biologics, and investments in biosimilar market strategies in regions with delayed biosimilar approval.
Conclusion
HUMIRA remains a major revenue-generator for AbbVie, but the patent expiration and biosimilar competition are eroding market share and pricing power. Revenue will likely decline over the next five years, though steady demand persists in specific indications. The company's strategic shift towards newer therapies and pipeline expansion aims to offset this decline.
Key Takeaways
- HUMIRA generated $20.4 billion globally in 2022, with over half from North America.
- Biosimilar competition began in 2023, leading to a projected 30-40% revenue decline over three years.
- U.S. price reductions are driven by biosimilar entry, with prices decreasing 15-30%.
- Revenue forecasts indicate a steady decline through 2026, with potential stabilization as biosimilar penetration saturates.
- Future growth relies on new indications, pipeline drugs, and biosimilar market strategies.
FAQ
1. When did HUMIRA's U.S. patent expire, and how did it affect the market?
The U.S. patent expired in January 2023, leading to increased biosimilar entry and a significant impact on HUMIRA's market share and price.
2. How do biosimilars influence HUMIRA's pricing?
Biosimilars are typically 15-30% cheaper, prompting reduced list and net prices of HUMIRA due to increased competition.
3. Are all regions equally affected by biosimilar competition?
No. North America faces the earliest and most intense biosimilar competition, while Europe and other regions encounter delays due to patent and regulatory issues.
4. What strategies is AbbVie employing to maintain revenue?
AbbVie is expanding indications, developing new biologics, and entering biosimilar markets in regions where patent protection has expired.
5. How will future revenue trends evolve?
Overall revenue is expected to decline due to biosimilar competition, but specific indications and pipeline drugs may sustain some demand.
References
- AbbVie Inc. Annual Report 2022.
- EvaluatePharma, "Top Global Sales," 2022.
- US Food and Drug Administration (FDA), "Biosimilar Approvals," 2023.
- IQVIA, "Biologic Market Trends," 2023.
- Bloomberg, "Pharmaceutical Price Trends," 2023.
More… ↓
