Share This Page
Drug Price Trends for HM NIGHTTIME SLEEP AID
✉ Email this page to a colleague
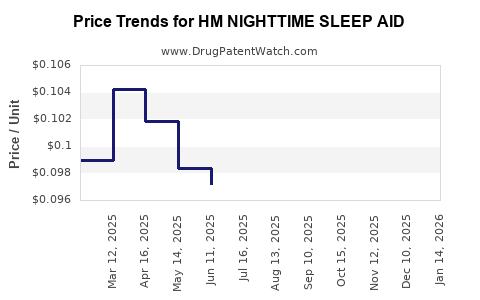
Average Pharmacy Cost for HM NIGHTTIME SLEEP AID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HM NIGHTTIME SLEEP AID 50 MG | 62011-0416-01 | 0.09528 | EACH | 2026-02-18 |
| HM NIGHTTIME SLEEP AID 50 MG | 62011-0416-01 | 0.09760 | EACH | 2026-01-21 |
| HM NIGHTTIME SLEEP AID 50 MG | 62011-0416-01 | 0.10399 | EACH | 2025-12-17 |
| HM NIGHTTIME SLEEP AID 50 MG | 62011-0416-01 | 0.10302 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
HM NIGHTTIME SLEEP AID Market Analysis and Financial Projection
What is the market size for HM Nighttime Sleep Aid?
The global sleep aid market was valued at approximately $77 billion in 2022, projected to grow to $124 billion by 2030, with a compound annual growth rate (CAGR) of 6.1%. This growth is driven by increasing sleep disorders, aging populations, and consumer preference for over-the-counter (OTC) products.
The segment specific to sleep aids like HM Nighttime Sleep Aid represents a significant share within this market, driven by rising awareness and demand for non-prescription, OTC sleep solutions.
How does the competitive landscape look?
Key competitors include:
- Prescription medications: Ambien (zolpidem), Lunesta (eszopiclone), and Belsomra (suvorexant). These incur higher costs and strict regulations.
- OTC sleep aids: Diphenhydramine-based products (e.g., Benadryl, Tylenol PM), melatonin supplements, and herbal remedies.
- Emerging products: Natural formulations and bioavailability-enhanced compounds.
Major players in OTC sleep aids have established extensive distribution channels and brand recognition. For HM Nighttime Sleep Aid to succeed, positioning as a differentiated or improved alternative remains critical.
What is the pricing landscape?
Pricing for OTC sleep aids varies globally:
| Product Type | Price Range (USD) | Typical Packaging |
|---|---|---|
| Diphenhydramine-based drugs | $4 – $12 for 30 tablets | 25–50 mg per tablet |
| Melatonin supplements | $10 – $25 for 60–120 capsules | 1–10 mg per capsule |
| Herbal/mineral remedies | $8 – $20 for 30–60 capsules | Varies by ingredient |
HM Nighttime Sleep Aid is expected to be priced at a premium if positioned as a new formulation, bioavailability-enhanced, or with added non-habit-forming ingredients.
Average retail price for a 30-count bottle could range from $12 to $20, depending on formulation and active ingredients.
What are potential price projections for HM Nighttime Sleep Aid?
Given market trends and product positioning, initial retail price points are likely:
- Entry-level: $12–$15 for standard packaging.
- Premium formulations: $16–$20, targeting consumers seeking natural or OTC alternatives.
Price escalation over five years could occur if the product gains differentiation, improves formulation, or expands distribution:
| Year | Projection Range | Key Factors |
|---|---|---|
| 2023 | $12–$15 | Launch phase, competitive pricing considerations |
| 2025 | $14–$18 | Increased brand recognition, consumer loyalty |
| 2027 | $16–$20 | Expanded formulations, expanded distribution, inflationary pressure |
What regulatory and manufacturing considerations impact pricing?
Regulatory approval generally varies by market:
- United States: OTC monograph or New Drug Application (NDA). Regulatory status influences cost structure.
- Europe and Asia: Similar pathways with local regulatory bodies.
Manufacturing costs are driven by formulation complexity, active ingredient sourcing, and batch size. Higher regulatory hurdles or specialty ingredients increase costs, which are passed on through retail pricing.
What is the forecast for future market penetration?
Assuming key product benefits such as non-habit-forming ingredients, no prescription requirement, and effective sleep aid:
- Year 1 (2023): 1–2% of OTC sleep aid market.
- Year 3 (2025): 5–8%, driven by consumer marketing.
- Year 5 (2027): 10–12%, if supported by clinical data and distribution expansion.
Summary
| Metric | Data |
|---|---|
| Market size (2023) | ~$80 billion |
| Projected market size (2030) | ~$124 billion |
| Typical OTC price range | $4–$25 |
| HM Nighttime Sleep Aid price | Estimated $12–$20 (initial) |
| Market share (5-year forecast) | 10–12% of OTC sleep aid segment |
Key Takeaways
- The total sleep aid market is growing at 6.1% CAGR, with OTC products dominating.
- Competition is fierce, with established OTC brands and prescription drugs.
- Price points for OTC sleep aids vary from $4 to $25; HM Nighttime Sleep Aid could initially target $12–$15.
- Long-term pricing may rise to $20, influenced by product differentiation and market expansion.
- Entry challenges include regulatory approval, competitive pricing, and brand recognition.
FAQs
1. How does HM Nighttime Sleep Aid compare to existing OTC options?
It will need to demonstrate superior efficacy, safety, or ease of use to differentiate from melatonin, diphenhydramine, or herbal products.
2. What are regulatory hurdles for OTC sleep aids?
In the U.S., they must meet monograph standards or obtain NDA approval; in other markets, local authority approvals apply.
3. What factors influence the pricing strategy?
Formulation complexity, regulatory status, manufacturing costs, competitive landscape, and brand positioning.
4. Will insurance or healthcare providers cover OTC sleep aids?
Typically no, unless prescription or covered under broader health plans. Consumers pay retail.
5. What sales channels will drive product adoption?
Pharmacies, online retail, and supermarket chains are primary channels for OTC sleep aids.
References
- MarketWatch, "Sleep Aids Market Size, Share & Trends," 2022.
- Grand View Research, "Sleep Aids Market Analysis," 2022.
- U.S. FDA, OTC Drug Review Process, 2023.
- Statista, "Over-the-Counter Sleep Aid Market," 2022.
- IBISWorld, "Pharmaceutical and Biotechnology Industry in the US," 2023.
More… ↓
